Abstract
Purpose
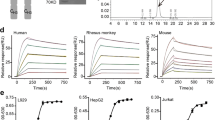
TAS266 is a novel agonistic tetravalent Nanobody® targeting the DR5 receptor. In preclinical studies, TAS266 was more potent than a cross-linked DR5 antibody or TRAIL. This first-in-human study was designed to evaluate the safety and tolerability, maximum tolerated dose, pharmacokinetics, pharmacodynamics, immunogenicity, and preliminary efficacy of TAS266.
Methods
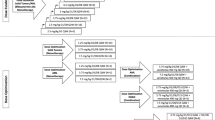
Adult patients with advanced solid tumors were to receive assigned doses of TAS266 (3, 10, 15, or 20 mg/kg) intravenously on days 1, 8, 15, and 22 of a 28-day treatment cycle.
Results
Grade ≥3 elevations in aspartate aminotransferase and/or alanine aminotransferase levels, occurring during cycle 1 in three of four patients at the 3 mg/kg dose level, were attributed to TAS266 and led to early study termination. Liver enzyme levels quickly returned to grade ≤1 following TAS266 discontinuation. Evidence of preexisting antibodies able to bind to TAS266 was found in the three patients who experienced these dose-limiting toxicities. Immunogenic responses remained elevated and strengthened at end-of-treatment (EOT). In the one patient who did not develop hepatotoxicity, no evidence of immunogenicity was observed at baseline or following administration of 4 TAS266 doses; however, incipient positive immunogenicity was observed at the EOT visit.
Conclusion
TAS266 was associated with unexpected, significant but reversible hepatotoxicity. Although the underlying mechanism is not fully elucidated, factors including the molecule’s high potency, immunogenicity to TAS266, and possibly increased DR5 expression on hepatocytes further enhancing the activity of the Nanobody® may have contributed to enhanced DR5 clustering and activation of hepatocyte apoptosis.



Similar content being viewed by others
References
Ghobrial IM, Witzig TE, Adjei AA (2005) Targeting apoptosis pathways in cancer therapy. CA Cancer J Clin 55:178–194
Ashkenazi A (2002) Targeting death and decoy receptors or the tumor-necrosis factor superfamily. Nat Rev Cancer 2:420–430
Pan G, O’Rourke K, Chinnaiyan AM et al (1997) The receptor for the cytotoxic ligand TRAIL. Science 276:111–113
Roth W, Isenmann S, Naumann U et al (1999) Locoregional Apo2L/TRAIL eradicates intracranial human malignant glioma xenografts in athymic mice in the absence of neurotoxicity. Biochem Biophys Res Commun 265:479–483
Mitsiades CS, Treon SP, Mitsiades N et al (2001) TRAIL/Apo2L ligand selectively induces apoptosis and overcomes drug resistance in multiple myeloma: therapeutic applications. Blood 98:795–804
Ashkenazi A, Pai RC, Fong S et al (1999) Safety and antitumor activity of recombinant soluble Apo2 ligand. J Clin Invest 104:155–162
Duiker WE, Mom CH, de Jong S et al (2006) The clinical of TRAIL. Eur J Cancer 42:2233–2240
Oikonomou E, Pintzas A (2013) The TRAIL of oncogenes to apoptosis. Biofactors 39:343–354
Gonzalvez F, Ashkenazi A (2010) New insights into apoptosis signaling by Apo2L/TRAIL. Oncogene 29:4752–4765
Alamasan A, Ashkenazi A (2003) Apo2L/TRAIL: apoptosis signaling, biology, and potential for cancer therapy. Cytokine Growth Factor Rev 14:337–348
Soria JC, Márk Z, Zaltoukal P et al (2011) Randomized phase II study of dulanermin in combination with paclitaxel, carboplatin, and bevacizumab in advanced non-small-cell lung cancer. J Clin Oncol 29:4442–4451
Wakelee HA, Patnaik A, Sikic BI et al (2010) Phase I and pharmacokinetic study of lexatumumab (HGS-ETR2) given every 2 weeks in patients with advanced solid tumors. Ann Oncol 21:376–381
Sharma S, de Vries EG, Infante JR et al (2014) Safety, pharmacokinetics, and pharmacodynamics of the DR5 antibody LBY135 alone an in combination with capecitabine in patients with advanced solid tumors. Invest New Drugs 32:135–144
Forero-Torres A, Shah J, Wood T et al (2010) Phase I trial of weekly tigatuzumab, an agonistic humanized monoclonal antibody targeting death receptor 5 (DR5). Cancer Biother Radiopharm 25:13–19
Paz-Ares L, Bálint B, de Boer RH et al (2013) A randomized phase 2 study of paclitaxel and carboplatin with or without conatumumab for first-line treatment of advanced non-small-cell lung cancer. J Thorac Oncol 8:329–337
Rocha Lima CM, Bayraktar S, Flores AM et al (2012) Phase Ib study of drozitumab combined with first-line mFOLFOX plus bevacizumab in patients with metastatic colorectal cancer. Cancer Invest 30:727–731
Huet HA, Growney JD, Johnson JA et al (2014) Multivalent nanobodies targeting death receptor 5 elicit superior tumor cell killing through efficient caspase induction. MAbs 6:1560–1570
Hao C, Song JH, His B et al (2004) TRAIL inhibits tumor growth but is nontoxic to human hepatocytes in chimeric mice. Cancer Res 64:8502–8506
Holland MC, Wurthner JU, Morley PJ et al (2013) Autoantibodies to variable heavy (VH) chain Ig sequences in humans impact the safety and clinical pharmacology of a VH domain antibody antagonist of TNF-α receptor 1. J Clin Immunol 33:1192–1203
Xue L, Rup B (2013) Evaluation of pre-existing antibody presence as a risk factor for posttreatment anti-drug antibody induction: analysis of human clinical study data for multiple biotherapeutics. AAPS J 15:893–896
Xue L, Fiscella M, Rajadhyaksha M et al (2013) Pre-existing biotherapeutic-reactive antibodies: survey results within the American Association of Pharmaceutical Scientists. AAPS J 15:852–855
Neuenschwander B, Branson M, Gsponer T (2008) Critical aspects of the Bayesian approach to phase I cancer trials. Stat Med 27:2420–2439
Jo M, Kim TH, Seol DW et al (2000) Apoptosis induced in normal human hepatocytes by tumor necrosis factor-related apoptosis-inducing ligand. Nat Med 6:564–567
Volkmann X, Fischer U, Bahr MJ et al (2007) Increased hepatotoxicity of tumor necrosis factor-related apoptosis-inducing ligand in diseased human liver. Hepatology 46:1498–1508
Plummer R, Attard G, Pacey S et al (2007) Phase 1 and pharmacokinetic study of lexatumumab in patients with advanced cancers. Clin Cancer Res 13:6187–6194
Camidge DR, Herbst RS, Gordon MS et al (2010) A phase I safety and pharmacokinetic study of the death receptor 5 agonistic antibody PRO95780 in patients with advanced malignancies. Clin Cancer Res 16:1256–1263
Demetri GD, Le Cesne A, Chawla SP et al (2012) First-line treatment of metastatic or locally advanced unresectable soft tissue sarcomas with conatumumab in combination with doxorubicin or doxorubicin alone: a phase I/II open-label and double-blind study. Eur J Cancer 48:547–563
Herbst RS, Eckhardt SG, Kurzrock R et al (2010) Phase I dose-escalation study of recombinant human Apo2L/TRAIL, a dual proapoptotic receptor agonist, in patients with advanced cancer. J Clin Oncol 28:2839–2846
Bartunek J, Barbato E, Heyndrickx G et al (2013) Novel antiplatelet agents: ALX-0081, a Nanobody directed towards von Willebrand factor. J Cardiovasc Transl Res 6:363–366
Holz JB, Sargentini-Maier L, De Bruyn S et al (2013) Twenty-four weeks of treatment with a novels anti-IL-6 receptor Nanobody® (ALX-0061) resulted in 84% ACR20 improvement and 58% DAS28 remission in a phase I/II study in RA. Ann Rheum Dis 72(suppl 3):64
Schoen P, Jacobs S, Verschueren K et al (2013) Anti-RANKL Nanobody ALX-0141 shows sustained biomarker inhibition in a phase I study in healthy postmenopausal women. Bone Abstr. doi:10.1530/boneabs.1.PP135
Fleischmann Roy M, Bruyn De et al (2012) A novel individualized treatment approach in open-label extension study of ozoralizumab (ATN-103) in subjects with rheumatoid arthritis on a background of methotrexate. Arthritis Rheum 64(Suppl 10):1311
Corazza N, Kassahn D, Jakob S et al (2009) TRAIL-induced apoptosis: between tumor therapy and immunopathology. Ann N Y Acad Sci 1171:50–58
Mundt B, Wirth T, Zender L et al (2005) Tumour necrosis factor related apoptosis inducing ligand (TRAIL) induces hepatic steatosis in viral hepatitis and after alcohol intake. Gut 54:1590–1596
Pilgrim CH, Thomson BN, Banting S et al (2012) The developing clinical problem of chemotherapy-induced hepatic injury. ANZ J Surg 82:23–29
Ryan P, Nanji S, Pollett A et al (2010) Chemotherapy-induced liver injury in metastatic colorectal cancer: semiquantitative histologic analysis of 334 resected liver specimens shows that vascular injury but not steatohepatitis is associated with preoperative chemotherapy. Am J Surg Pathol 34:784–791
Acknowledgments
The authors thank Syntaxx Communications, Inc who provided manuscript development and medical writing services, with the support of Novartis Pharmaceuticals Corporation and to Glenda Chambers, the TAS266 study coordinator at South Texas Accelerated Research Therapeutics.
Conflict of interest
SB and ZT are employees of Novartis Pharmaceuticals Corporation. RI, NK, HH, KK, SB, and MH are employees of Novartis Institutes for Biomedical Research. RI, NK, HH, and ZT own stock in Novartis Pharmaceuticals Corporation. START received funding from Novartis Pharmaceuticals Corporation for conduct of this clinical trial by KP and DR. JC and AM have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papadopoulos, K.P., Isaacs, R., Bilic, S. et al. Unexpected hepatotoxicity in a phase I study of TAS266, a novel tetravalent agonistic Nanobody® targeting the DR5 receptor. Cancer Chemother Pharmacol 75, 887–895 (2015). https://doi.org/10.1007/s00280-015-2712-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-015-2712-0