Abstract
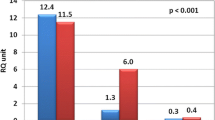
To investigate the role of Notch signaling pathway in immune thrombocytopenic purpura (ITP), we measured the expression of 11 Notch pathway molecules in ITP patients and evaluated their clinical relevance. Real-time reverse transcriptase polymerase chain reaction results showed there was aberrant expression of some Notch molecules in ITP. Notch1 and Notch3 expression elevated, while Notch2 decreased statistically in ITP patients. As for Notch ligands, only DLL1 was found downregulated in ITP. The expression of Notch target gene, Hes1, was also upregulated. In accordance with the mRNA level, Notch1 and Hes1 protein expression was also found elevated by Western blot. Immunocytochemistry showed that Notch1 expressed highly in the cytomembrane, cytoplasm, and part of cellular nucleus for ITP while weak in cytomembrane for controls, and Hes1 of ITP was found expressed higher in cellular nucleus than that of controls. Our findings suggest that the aberrant expression profile of Notch pathway may be involved in ITP, and blockage of Notch1 pathway is likely a promising therapeutic concept.


Similar content being viewed by others
References
Amsen D, Blander JM, Lee GR, Tanigaki K, Honjo T, Flavell RA (2004) Instruction of distinct CD4 T helper cell fates by different notch ligands on antigen-presenting cells. Cell 117:515–526. doi:10.1016/S0092-8674(04)00451-9
Ansel KM, Djuretic I, Tanasa B, Rao A (2006) Regulation of Th2 differentiation and Il4 locus accessibility. Annu Rev Immunol 24:607–656. doi:10.1146/annurev.immunol.23.021704.115821
Bhanot U, Köhntop R, Hasel C, Möller P (2008) Evidence of Notch pathway activation in the ectatic ducts of chronic pancreatitis. J Pathol 214:312–319. doi:10.1002/path.2293
Cines DB, Blanchette VS (2002) Immune thrombocytopenic purpura. N Engl J Med 346:995–1008. doi:10.1056/NEJMra010501
Coopamah MD, Freedman J, Semple JW (2003) Anti-D initially stimulates an Fc-dependent leukocyte oxidative burst and subsequently suppresses erythrophagocytosis via interleukin-1 receptor antagonist. Blood 102:2862–2867. doi:10.1182/blood-2003-04-1029
Cooper N, Bussel J (2006) The pathogenesis of immune thrombocytopaenic purpura. Br J Haematol 133:364–374. doi:10.1111/j.1365-2141.2006.06024.x
Ehebauer M, Hayward P, Arias AM (2006) Notch, a universal arbiter of cell fate decisions. Science 314:1414–1415. doi:10.1126/science.1134042
Elyaman W, Bradshaw EM, Wang Y, Oukka M, Kivisäkk P, Chiba S, Yagita H, Khoury SJ (2007) JAGGED1 and delta1 differentially regulate the outcome of experimental autoimmune encephalomyelitis. J Immunol 179:5990–5998
Fleming RJ (1998) Structural conservation of Notch receptors and ligands. Semin Cell Dev Biol 9:599–607. doi:10.1006/scdb.1998.0260
George JN, Woolf SH, Raskob GE, Wasser JS, Aledort LM, Ballem PJ, Blanchette VS, Bussel JB, Cines DB, Kelton JG, Lichtin AE, McMillan R, Okerbloom JA, Regan DH, Warrier I (1996) Idiopathic thrombocytopenic purpura: a practice guideline developed by explicit methods for the American Society of Hematology. Blood 88:3–40
Hou M, Lv B, He Q, Lu L, Shi Y, Ji X, Ma D, Zhang M (2003) Both splenic CD5(+) B and CD5(−) B cells produce platelet glycoprotein-specific autoantibodies in chronic ITP. Thromb Res 110:1–5. doi:10.1016/S0049-3848(03)00244-5
Jurynczyk M, Jurewicz A, Raine CS, Selmaj K (2008) Notch3 inhibition in myelin-reactive T cells down-regulates protein kinase C theta and attenuates experimental autoimmune encephalomyelitis. J Immunol 180:2634–2640
Kostianovsky AM, Maier LM, Baecher-Allan C, Anderson AC, Anderson DE (2007) Up-regulation of gene related to anergy in lymphocytes is associated with Notch-mediated human T cell suppression. J Immunol 178:6158–6163
Lackner KJ, Peetz D, von Landenberg P (2006) Revision of the Sapporo criteria for the antiphospholipid syndrome: coming to grips with evidence and Thomas Bayes? Thromb Haemost 95:917–919
Maekawa Y, Tsukumo S, Chiba S, Hirai H, Hayashi Y, Okada H, Kishihara K, Yasutomo K (2003) Delta1–Notch3 interactions bias the functional differentiation of activated CD4+ T cells. Immunity 19:549–559. doi:10.1016/S1074-7613(03)00270-X
McKenzie G, Ward G, Stallwood Y, Briend E, Papadia S, Lennard A, Turner M, Champion B, Hardingham GE (2006) Cellular Notch responsiveness is defined by phosphoinositide 3-kinase-dependent signals. BMC Cell Biol 7:10. doi:10.1186/1471-2121-7-10
Osborne BA, Minter LM (2007) Notch signaling during peripheral T-cell activation and differentiation. Nat Rev Immunol 7:64–75. doi:10.1038/nri1998
Palaga T, Miele L, Golde TE, Osborne BA (2003) TCR-mediated Notch signaling regulates proliferation and IFN-gamma production in peripheral T cells. J Immunol 171:3019–3024
Radtke F, Wilson A, Mancini SJ, MacDonald HR (2004) Notch regulation of lymphocyte development and function. Nat Immunol 5:247–253. doi:10.1038/ni1045
Rutz S, Mordmüller B, Sakano S, Scheffold A (2005) Notch ligands Delta-like1, Delta-like4 and Jagged1 differentially regulate activation of peripheral T helper cells. Eur J Immunol 35:2443–2451. doi:10.1002/eji.200526294
Seifert T, Bauer J, Weissert R, Fazekas F, Storch MK (2007) Notch1 and its ligand Jagged1 are present in remyelination in a T-cell- and antibody-mediated model of inflammatory demyelination. Acta Neuropathol 113:195–203. doi:10.1007/s00401-006-0170-9
Semple JW, Freedman J (1991) Increased antiplatelet T helper lymphocyte reactivity in patients with autoimmune thrombocytopenia. Blood 78:2619–2625
Semple JW, Milev Y, Cosgrave D, Mody M, Hornstein A, Blanchette V, Freedman J (1996) Differences in serum cytokine levels in acute and chronic autoimmune thrombocytopenic purpura: relationship to platelet phenotype and antiplatelet T-cell reactivity. Blood 87:4245–4254
Skokos D, Nussenzweig MC (2007) CD8-DCs induce IL-12-independent Th1 differentiation through Delta 4 Notch-like ligand in response to bacterial LPS. J Exp Med 204:1525–1531
Sodsai P, Hirankarn N, Avihingsanon Y, Palaga T (2008) Defects in Notch1 upregulation upon activation of T cells from patients with systemic lupus erythematosus are related to lupus disease activity. Lupus 17:645–653. doi:10.1177/0961203308089406
Tan EM, Cohen AS, Fries JF, Masi AT, McShane DJ, Rothfield NF, Schaller JG, Talal N, Winchester RJ (1982) The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 25:1271–1277. doi:10.1002/art.1780251101
Teachey DT, Seif AE, Brown VI, Bruno M, Bunte RM, Chang YJ, Choi JK, Fish JD, Hall J, Reid GS, Ryan T, Sheen C, Zweidler-McKay P, Grupp SA (2008) Targeting Notch signaling in autoimmune and lymphoproliferative disease. Blood 111:705–714. doi:10.1182/blood-2007-05-087353
Ware RE, Howard TA (1993) Phenotypic and clonal analysis of T lymphocytes in childhood immune thrombocytopenic purpura. Blood 82:2137–2142
Wilson WA, Gharavi AE, Koike T, Lockshin MD, Branch DW, Piette JC, Brey R, Derksen R, Harris EN, Hughes GR, Triplett DA, Khamashta MA (1999) International consensus statement on preliminary classification criteria for definite antiphospholipid syndrome: report of an international workshop. Arthritis Rheum 42:1309–1311. doi:10.1002/1529-0131(199907)42:7<1309::AID-ANR1>3.0.CO;2-F
Zang S, Ji C, Qu X, Dong X, Ma D, Ye J, Ma R, Dai J, Guo D (2007) A study on Notch signaling in human breast cancer. Neoplasma 54:304–310
Acknowledgments
This study was partially supported by grants from the National Natural Science Foundation (30600680, 30471941, 30770922, 30470742, 30570779, 30600259, 30628015 and 30300312), 973 Project (2006CB503800), Key Clinical Research Project of Chinese Ministry of Health (2007–2009), Research Project of National Public Fare (200802031), and the Shandong Technological Development Project (2005BS03022, 2005GG4202018 and Q2008C07), and Taishan Scholar Fundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jianjian Dai and Xiaojuan Zhu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ma, D., Dai, J., Zhu, X. et al. Aberrant expression of Notch signaling molecules in patients with immune thrombocytopenic purpura. Ann Hematol 89, 155–161 (2010). https://doi.org/10.1007/s00277-009-0790-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-009-0790-y