Abstract
Purpose
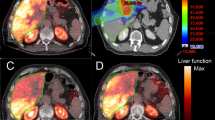
To define a threshold radiation dose to non-tumoral liver from 90Y radioembolization that results in hepatic toxicity using pair-production PET.
Materials and Methods
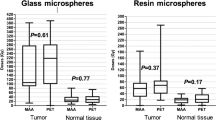
This prospective single-arm study enrolled 35 patients undergoing radioembolization. A total of 34 patients (27 with HCC and 7 with liver metastases) were included in the final analysis. Of 27 patients with underlying cirrhosis, 22 and 5 patients were Child–Pugh A and B, respectively. Glass and resin microspheres were used in 32 (94%) and 2 (6%) patients, respectively. Lobar and segmental treatment was done in 26 (76%) and 8 (24%) patients, respectively. Volumetric analysis was performed on post-radioembolization time-of-flight PET imaging to determine non-tumoral parenchymal dose. Hepatic toxicity was evaluated up to 120 days post-treatment, with CTCAE grade ≤ 1 compared to grade ≥ 2.
Results
The median dose delivered to the non-tumoral liver in the treated lobe was 49 Gy (range 0–133). A total of 15 patients had grade ≤ 1 hepatic toxicity, and 19 patients had grade ≥ 2 toxicity. Patients with a grade ≥ 2 change in composite toxicity (70.7 vs. 43.8 Gy), bilirubin (74.1 vs. 43.3 Gy), albumin (84.2 vs. 43.8 Gy), and AST (94.5 vs. 47.1 Gy) have significantly higher non-tumoral parenchymal doses than those with grade ≤ 1. Liver parenchymal dose and Child–Pugh status predicted grade ≥ 2 toxicity, observed above a dose threshold of 54 Gy.
Conclusion
Increasing delivered 90Y dose to non-tumoral liver measured by internal pair-production PET correlates with post-treatment hepatic toxicity. The likelihood of toxicity exceeds 50% at a dose threshold of 54 Gy.
ClinicalTrials.gov identifier: NCT02848638.


Similar content being viewed by others
References
Dancey JE, Shepherd FA, Paul K, et al. Treatment of nonresectable hepatocellular carcinoma with intrahepatic 90Y-microspheres. J Nucl Med. 2000;41:1673–81.
Garin E, Rolland Y, Edeline J, et al. Personalized dosimetry and intensification concept with 90Y-loaded glass microsphere radioembolization induce prolonged overall survival in hepatocellular carcinoma patients with portal vein thrombosis. J Nucl Med. 2015;56:339–46.
Song YS, Paeng JC, Kim HC, et al. PET/CT-based dosimetry in 90Y-microsphere selective internal radiation therapy: single cohort comparison with pretreatment planning on (99m)Tc-MAA imaging and correlation with treatment efficacy. Medicine. 2015;94(23):e945.
Gil-Alzugaray B, Chopitea A, Iñarrairaegui M, et al. Prognostic factors and prevention of radioembolization-induced liver disease. Hepatology. 2013;57:1078–87.
Riaz A, Lewandowski RJ, Kulik LM, et al. Complications following radioembolization with yttrium-90 microspheres: a comprehensive literature review. J Vasc Interv Radiol. 2009;20:1121–30.
Piana PE, Gonsalves CF, Sato T, et al. Toxicities after radioembolization with yttrium-90 SIR-Spheres: incidence and contributing risk factors at a single center. J Vasc Interv Radiol. 2011;22:1373–9.
Lam MG, Abdelmaksoud MH, Chang DT, et al. Safety of 90Y radioembolization in patients who have undergone previous external beam radiation therapy. Int J Radiat Oncol Biol Phys. 2013;87(2):323–9.
Gabrielson A, Miller A, Banovac F, Kim A, He AR, Unger K. Outcomes and predictors of toxicity after selective internal radiation therapy using yttrium-90 resin microspheres for unresectable hepatocellular carcinoma. Front Oncol. 2015;5:292.
Son SH, Jang HS, Jo IY, et al. Significance of an increase in the Child–Pugh score after radiotherapy in patients with unresectable hepatocellular carcinoma. Radiat Oncol. 2014;9:101.
Young JY, Rhee TK, Atassi B, et al. Radiation dose limits and liver toxicities resulting from multiple yttrium-90 radioembolization treatments for hepatocellular carcinoma. J Vasc Interv Radiol. 2007;18:1375–82.
Lau WY, Leung WT, Ho S, et al. Treatment of inoperable hepatocellular carcinoma with intrahepatic arterial yttrium-90 microspheres: a phase I and II study. Br J Cancer. 1994;70:994–9.
Goin JE, Salem R, Carr BI, et al. Treatment of unresectable hepatocellular carcinoma with intrahepatic yttrium 90 microspheres: factors associated with liver toxicities. J Vasc Interv Radiol. 2005;16:205–13.
Elschot M, Vermolen BJ, Lam MG, Keizer B, Van den Bosch MA, De Jong HW. Quantitative comparison of PET and Bremsstrahlung SPECT for imaging the in vivo yttrium-90 microsphere distribution after liver radioembolization. PLoS ONE. 2013;8(2):e55742.
Padia SA, Alessio A, Kwan SW, Lewis DJ, Vaidya S, Minoshima S. Comparison of positron emission tomography and bremsstrahlung imaging to detect particle distribution in patients undergoing yttrium-90 radioembolization for large hepatocellular carcinomas or associated portal vein thrombosis. J Vasc Interv Radiol. 2013;24(8):1147–53.
Salem R, Thurston KG. Radioembolization with 90Yttrium microspheres: a state-of-the-art brachytherapy treatment for primary and secondary liver malignancies. Part 1: technical and methodologic considerations. J Vasc Interv Radiol. 2006;17:1251–78.
Salem R, Thurston KG. Radioembolization with 90Yttrium microspheres: a state-of-the-art brachytherapy treatment for primary and secondary liver malignancies. Part 2: special topics. J Vasc Interv Radiol. 2006;17:1425–39.
Padia SA, Kwan SW, Roudsari B, Monsky WL, Coveler A, Harris WP. Superselective yttrium-90 radioembolization for hepatocellular carcinoma yields high response rates with minimal toxicity. J Vasc Interv Radiol. 2014;25:1067–73.
Lea WB, Tapp KN, Tann M, Hutchins GD, Fletcher JW, Johnson MS. Microsphere localization and dose quantification using positron emission tomography/CT following hepatic intraarterial radioembolization with yttrium-90 in patients with advanced hepatocellular carcinoma. J Vasc Interv Radiol. 2014;25(10):1595–603.
Pan CC, Kavanagh BD, Dawson LA, et al. Radiation-associated liver injury. Int J Radiat Oncol Biol Phys. 2010;76(3 Suppl):S94–100.
National Cancer Institute. Common Terminology Criteria for Adverse Events v4.0. NCI, NIH, DHHS. May 29, 2009. NIH publication # 09-7473.
Roberson JD, McDonald AM, Baden CJ, Lin CP, Jacob R, Burnett OL. Factors associated with increased incidence of severe toxicities following yttrium-90 resin microspheres in the treatment of hepatic malignancies. World J Gastroenterol. 2016;22(10):3006–14.
Strigari L, Sciuto R, Rea S, et al. Efficacy and toxicity related to treatment of hepatocellular carcinoma with 90Y-SIR spheres: radiobiologic considerations. J Nucl Med. 2010;51:1377–85.
Smits ML. Clinical and laboratory toxicity after intra-arterial radioembolization with 90Y-microspheres for unresectable liver metastases. PLoS ONE. 2013;8(10):10.
Kennedy AS, McNeillie P, Dezarn WA, et al. Treatment parameters and outcome in 680 treatments of internal radiation with resin 90Y-microspheres for unresectable hepatic tumors. Int J Radiat Oncol Biol Phys. 2009;74(5):1494–500.
Lau WY, Kennedy AS, Kim YH, et al. Patient selection and activity planning guide for selective internal radiotherapy with yttrium-90 resin microspheres. Int J Radiat Oncol Biol Phys. 2012;82(1):401–7.
Hickey R, Lewandowski RJ, Prudhomme T, et al. 90Y radioembolization of colorectal hepatic metastases using glass microspheres: safety and survival outcomes from a 531-patient multicenter study. J Nucl Med. 2016;57(5):665–71.
Srinivas S, Natarajan N, Kuroiwa J, et al. Determination of radiation absorbed dose to primary liver tumors and normal liver tissue using post-radioembolization 90Y PET. Front Oncol. 2014;13(4):255.
Lam MG, Louie JD, Iagaru AH, Goris ML, Sze DY. Safety of repeated yttrium-90 radioembolization. Cardiovasc Interv Radiol. 2013;36(5):1320–8.
Guha C, Kavanagh BD. Hepatic radiation toxicity: avoidance and amelioration. Semin Radiat Oncol. 2011;21(4):256.
Acknowledgements
Grants for this study: Philips Healthcare. IRB statement: This study was performed under IRB approval at the University of Washington.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S. Padia: Consultant for BTG International and the other authors have no conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Chan, K.T., Alessio, A.M., Johnson, G.E. et al. Hepatotoxic Dose Thresholds by Positron-Emission Tomography After Yttrium-90 Radioembolization of Liver Tumors: A Prospective Single-Arm Observational Study. Cardiovasc Intervent Radiol 41, 1363–1372 (2018). https://doi.org/10.1007/s00270-018-1949-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-018-1949-5