Abstract
Purpose
Irreversible electroporation (IRE) is a new potential ablation modality for small renal masses. Animal experiments have shown preservation of the urine-collecting system (UCS). The purpose of this clinical study was to perform the first evaluation and comparison of IRE’s effects on the renal UCS by using urinary cytology, magnetic-resonance imaging, and resection histology in men after IRE of pT1a renal-cell carcinoma (RCC).
Methods
Seven patients with biopsy-proven RCC pT1a cN0cM0 underwent IRE in a phase 2a pilot ablate-and-resect study (IRENE trial). A contrast-enhanced, diffusion-weighted MRI and urinary cytology was performed 1 day before and 2, 7, and 27 days after IRE. Twenty-eight days after IRE the tumour region was completely resected surgically.
Results
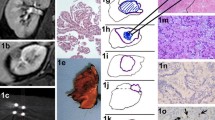
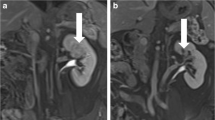
Technical feasibility was demonstrated in all patients. In all cases, MRI revealed complete coverage of the tumour area by the ablation zone with degenerative change. The urographic late venous MRI phase (urogram scans) demonstrated normal morphological appearances. Urine cytology showed a temporary vacuolisation of the cyto- and caryoplasmas after IRE. Whereas the urothelium showed signs of regeneration 28 days after IRE-ablation, the tumour and parenchyma below it showed necrosis and permanent tissue destruction.
Conclusions
Renal percutaneous IRE appears to be a safe treatment for pT1a RCC. The preservation of the UCS with unaltered normal morphology as well as urothelial regeneration and a phenomenon (new in urinary cytology) of temporary degeneration with vacuolisation of detached transitional epithelium cells were demonstrated in this clinical pilot study.


Similar content being viewed by others
References
Ljungberg B, Albiges L, Bensalah K, Bex A, Giles RH, Hora M, Kuczyk MA, Lam T, Marconi L, Merseburger AS, Powles T, Staehler M, Volpe A, Dabestani S, Fernandez-Pello Montes S, Hofmann F, Tahbaz R. EAU Guidelines on renal cell carcinoma. European Association of Urology 2017. Update March 2017. http://uroweb.org/wp-content/uploads/10-Renal-Cell-Carcinoma_2017_web.pdf. Accessed 4 July 2017.
D’Andrea D, Shariat SF, Klatte T. Update on ablative therapies of renal tumors. Curr Opin Urol. 2016;26(5):410–6. doi:10.1097/MOU.0000000000000306.
Rubinsky B (2009) Irreversible electroporation. 1st ed. Berlin: Springer; p. 328. ISBN 3642054196.
Wendler JJ, Pech M, Blaschke S, Porsch M, Janitzky A, Ulrich M, Dudeck O, Ricke J, Liehr UB. Angiography in the isolated perfused kidney: radiological evaluation of vascular protection in tissue ablation by nonthermal irreversible electroporation. Cardiovasc Interv Radiol. 2012;35(2):383–90. doi:10.1007/s00270-011-0187-x.
Wendler JJ, Pech M, Porsch M, Janitzky A, Fischbach F, Buhtz P, Vogler K, Hühne S, Borucki K, Strang C, Mahnkopf D, Ricke J, Liehr UB. Urinary tract effects after multifocal nonthermal irreversible electroporation of the kidney: acute and chronic monitoring by magnetic resonance imaging, intravenous urography and urinary cytology. Cardiovasc Interv Radiol. 2012;35(4):921–6. doi:10.1007/s00270-011-0257-0.
Wendler JJ, Porsch M, Hühne S, Baumunk D, Buhtz P, Fischbach F, Pech M, Mahnkopf D, Kropf S, Roessner A, Ricke J, Schostak M, Liehr UB. Short- and mid-term effects of irreversible electroporation on normal renal tissue: an animal model. Cardiovasc Interv Radiol. 2013;36(2):512–20. doi:10.1007/s00270-012-0452-7.
Pech M, Janitzky A, Wendler JJ, Strang C, Blaschke S, Dudeck O, Ricke J, Liehr UB. Irreversible electroporation of renal cell carcinoma: a first-in-man phase I clinical study. Cardiovasc Interv Radiol. 2011;34(1):132–8. doi:10.1007/s00270-010-9964-1.
Thomson KR, Cheung W, Ellis SJ, Federman D, Kavnoudias H, Loader-Oliver D, Roberts S, Evans P, Ball C, Haydon A. Investigation of the safety of irreversible electroporation in humans. J Vasc Interv Radiol. 2011;22(5):611–21. doi:10.1016/j.jvir.2010.12.014.
Wendler JJ, Ricke J, Pech M, Fischbach F, Jürgens J, Siedentopf S, Roessner A, Porsch M, Baumunk D, Schostak M, Köllermann J, Liehr UB. First delayed resection findings after irreversible electroporation (IRE) of human localised renal cell carcinoma (RCC) in the IRENE pilot Phase 2a trial. Cardiovasc Interv Radiol. 2016;39(2):239–50. doi:10.1007/s00270-015-1200-6.
Diehl SJ, Rathmann N, Kostrzewa M, Ritter M, Smakic A, Schoenberg SO, Kriegmair MC. Irreversible electroporation for surgical renal masses in solitary kidneys: short-term interventional and functional outcome. J Vasc Interv Radiol. 2016;27(9):1407–13. doi:10.1016/j.jvir.2016.03.044.
Canvasser NE, Sorokin I, Lay AH, Morgan MS, Ozayar A, Trimmer C, Cadeddu JA. Irreversible electroporation of small renal masses: suboptimal oncologic efficacy in an early series. World J Urol. 2017. doi:10.1007/s00345-017-2025-5.
Wendler JJ, Porsch M, Nitschke S, Köllermann J, Siedentopf S, Pech M, Fischbach F, Ricke J, Schostak M, Liehr UB. A prospective Phase 2a pilot study investigating focal percutaneous irreversible electroporation (IRE) ablation by NanoKnife in patients with localised renal cell carcinoma (RCC) with delayed interval tumour resection (IRENE trial). Contemp Clin Trials. 2015;43:10–9. doi:10.1016/j.cct.2015.05.002.
Manual NanoKnife_ System Procedure & Trouble Shouting Guide. Software version 2.2.0. AngioDynamics_, _ 2011 AngioDynamics, Inc. p. 1–159.
Dupuy DE, Gervais DA, Gianfelice D, Gillams AR, Lee FT Jr, Leen E, Lencioni R, Littrup PJ, Livraghi T, Lu DS, McGahan JP, Meloni MF, Nikolic B, Pereira PL, Liang P, Rhim H, Rose SC, Salem R, Sofocleous CT, Sofocleous CT, Solomon SB, Soulen MC, Tanaka M, Vogl TJ, Wood BJ, Goldberg SN. International Working Group on Image-Guided Tumor Ablation, Standard of Practice Committee of the Cardiovascular and Interventional Radiological Society of Europe. Image-guided tumor ablation: standardization of terminology and reporting criteria—a 10-year update. J Vasc Interv Radiol. 2014;25(11):1691–705. doi:10.1016/j.jvir.2014.08.027.
Ahmed M. Technology Assessment Committee of the Society of Interventional Radiology. Image-guided tumor ablation: standardization of terminology and reporting criteria—a 10-year update: supplement to the consensus document. J Vasc Interv Radiol. 2014;25(11):1706–8. doi:10.1016/j.jvir.2014.09.005.
Wendler JJ, Friebe B, Baumunk D, Blana A, Franiel T, Ganzer R, Hadaschik B, Henkel T, Köhrmann KU, Köllermann J, Kuru T, Machtens S, Roosen A, Salomon G, Schlemmer HP, Sentker L, Witzsch U, Liehr UB, Ricke J, Schostak M. Focal therapy for small renal masses: observation, ablation or surgery [in German]. Urologe A. 2016;55(5):594–606. doi:10.1007/s00120-016-0075-8.
Zondervan PJ, Wagstaff PG, Desai MM, de Bruin DM, Fraga AF, Hadaschik BA, Köllermann J, Liehr UB, Pahernik SA, Schlemmer HP, Wendler JJ, Algaba F, de la Rosette JJ, Laguna Pes MP. Follow-up after focal therapy in renal masses: an international multidisciplinary Delphi consensus project. World J Urol. 2016;34(12):1657–65.
Wendler JJ, Ricke J, Pech M, Fischbach F, Jürgens J, Porsch M, Janitzky A, Baumunk D, Siedentopf S, Köllermann J, Schostak M, Liehr U-B. Initial assessment of clinical feasibility, safety and efficacy of NanoKnife irreversible electroporation (IRE) in the focal treatment of localized renal cell carcinoma (RCC) with delayed interval tumor resection (IRENE trial). Eur Urol Suppl. 2017;16(3):e102–3. doi:10.1016/S1569-9056(17)30129-X.
Wendler JJ, Fischbach K, Ricke J, Jürgens J, Fischbach F, Köllermann J, Porsch M, Baumunk D, Schostak M, Liehr UB, Pech M. Irreversible electroporation (IRE): standardization of terminology and reporting criteria for analysis and comparison. Pol J Radiol. 2016;81:54–64. doi:10.12659/PJR.896034.
Deodhar A, Monette S, Single GW Jr, Hamilton WC Jr, Thornton R, Maybody M, Coleman JA, Solomon SB. Renal tissue ablation with irreversible electroporation: preliminary results in a porcine model. Urology. 2011;77(3):754–60. doi:10.1016/j.urology.2010.08.036.
Tracy CR, Kabbani W, Cadeddu JA. Irreversible electroporation (IRE): a novel method for renal tissue ablation. BJU Int. 2011;107(12):1982–7. doi:10.1111/j.1464-410X.2010.09797.x.
Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. 8th ed. New York: Wiley; 2017.
Wittekind C, Compton CC, Brierley J, Sobin LH. TNM supplement. A commentary on uniform use. 4th ed. New York: Wiley; 2012.
Srimathveeravalli G, Silk M, Wimmer T, Monette S, Kimm S, Maybody M, Solomon SB, Coleman J, Durack JC. Feasibility of catheter-directed intraluminal irreversible electroporation of porcine ureter and acute outcomes in response to increasing energy delivery. J Vasc Interv Radiol. 2015;26(7):1059–66. doi:10.1016/j.jvir.2015.01.020.
Srimathveeravalli G, Cornelis F, Wimmer T, Monette S, Kimm SY, Maybody M, Solomon SB, Coleman JA, Durack JC. Normal porcine ureter retains lumen wall integrity but not patency following catheter-directed irreversible electroporation: imaging and histologic assessment over 28 days. J Vasc Interv Radiol. 2017;28(6):913–919.e1. doi:10.1016/j.jvir.2017.02.032.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed and the conduct of the study as a whole was in accordance with the 1964 Helsinki declaration and its later amendments and comparable ethical standards. Approval was obtained for the study by the national research committee (BfArM) and the institutional ethics committee.
Informed Consent
Informed consent was obtained from all subjects in this study. Additional informed consent for identifying information was not required.
Additional information
M. Böhm: Working group urine cytology and urine-based markers of the German Society of Urology (DGU).
Study group IRENE trial featured by AKFM-DGU (J. J. Wendler, J. Köllermann, D. Schindele, D. Baumunk, M. Schostak, U. B. Liehr) and DAfMT (J. J. Wendler, M. Pech, B. Friebe, J. Jürgens, F. Fischbach, J. Ricke, U. B. Liehr), Germany.
Rights and permissions
About this article
Cite this article
Wendler, J.J., Pech, M., Köllermann, J. et al. Upper-Urinary-Tract Effects After Irreversible Electroporation (IRE) of Human Localised Renal-Cell Carcinoma (RCC) in the IRENE Pilot Phase 2a Ablate-and-Resect Study. Cardiovasc Intervent Radiol 41, 466–476 (2018). https://doi.org/10.1007/s00270-017-1795-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-017-1795-x