Abstract
Objective
To evaluate the efficacy and safety of an injectable lyophilized articular cartilage matrix (LACM) which is in the form of cartilage powder that may increase the convenience of use and become unaffected by donor site morbidity as a potential filler.
Methods
The safety test was conducted using 20 rabbits with an intravascular embolization model. Commonly used commercial hyaluronic acid (HA), acellular dermal matrix (ADM), polymethyl methacrylate (PMMA), and polylactic acid (PLA) products were selected for comparison of efficacy and adverse effect of LACM. The efficacy test was performed using 30 mice subcutaneous buttock-injection model. Gross assessments of the changes in size of injected materials and histological examinations were conducted.
Results
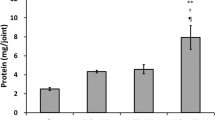
Regarding the safety test, in one rabbit in the HA group, complete blurring occurred in the left eye, and necrosis of the posterior auricular artery was observed in one rabbit in the ADM group. In the ADM, severe ecchymosis around the injection sites was observed, and the bruises lasted longer than in the other groups. The LACM exhibited faster bruise resolution. Comparing the changes in size between weeks 4 and 12 in the mouse model, no significant changes in size were observed in the LACM (p = 0.359), HA (p = 0.421), and ADM (p = 0.097), but a significant change was observed in the PMMA (p = 0.001). In the histological examination, LACM exhibited well-demarcated margins with good biocompatibility.
Conclusion
The LACM shows potential as a facial filler according to the in vivo safety and efficacy tests
Level of Evidence V
This journal requires that authors assign a level of evidence to each submission to which Evidence-Based Medicine rankings are applicable. This excludes Review Articles, Book Reviews, and manuscripts that concern Basic Science, Animal Studies, Cadaver Studies, and Experimental Studies. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266





Similar content being viewed by others
References
Liu MH, Beynet DP, Gharavi NM (2019) Overview of deep dermal fillers. Fac Plast Surg 35:224–229
Moon HJ (2018) Injection rhinoplasty using filler. Fac Plast Surg Clin North Am 26:323–330
Attenello NH, Maas CS (2015) Injectable fillers: review of material and properties. Fac Plast Surg 31:29–34
Na HG, Jang YJ (2020) Use of nasal implants and dorsal modification when treating the East Asian nose. Otolaryngol Clin North Am 53:255–266
Lee W, Koh IS, Oh W, Yang EJ (2020) Ocular complications of soft tissue filler injections: a review of literature. . 19:772–781
Kapoor KM, Kapoor P, Heydenrych I, Bertossi D (2020) Vision loss associated with hyaluronic acid fillers: a systematic review of literature. Aesthet Plast Surg 44:929–944
Lee KH (2015) Infection in the nasal tip caused by acellular dermal matrix. Plast Reconstr Surg Glob Op 3:e581
Kim SW, Dobratz EJ, Ballert JA, Voglewede AT, Park SS (2009) Subcutaneous implants coated with tissue-engineered cartilage. Laryngoscope 119:62–66
Bullocks JM, Echo A, Guerra G, Stal S, Yuksel E (2011) A novel autologous scaffold for diced-cartilage grafts in dorsal augmentation rhinoplasty. Aesthet Plast Surg 35:569–579
Suh MK, Ahn ES, Kim HR, Dhong ES (2013) A 2-year follow-up of irradiated homologous costal cartilage used as a septal extension graft for the correction of contracted nose in Asians. Ann Plast Surg 71:45–49
Cerkes N, Basaran K (2016) Diced cartilage grafts wrapped in rectus abdominis fascia for nasal dorsum augmentation. Plast Reconstr Surg 137:43–51
Gordon CR, Alghoul M, Goldberg JS, Habal MB, Papay F (2011) Diced cartilage grafts wrapped in AlloDerm for dorsal nasal augmentation. J Craniofac Surg 22:1196–1199
Jang YJ, Song HM, Yoon YJ, Sykes JM (2009) Combined use of crushed cartilage and processed fascia lata for dorsal augmentation in rhinoplasty for Asians. Laryngoscope 119:1088–1092
Park P, Jin HR (2016) Diced cartilage in fascia for major nasal dorsal augmentation in Asians: a review of 15 consecutive cases. Aesthet Plast Surg 40:832–839
Tan O, Algan S, Cinal H, Barin EZ, Kara M, Inaloz A (2016) Management of saddle nose deformity using dermal fat and costal cartilage “Sandwich” graft: a problem-oriented approach and anthropometric evaluation. J Oral Maxillofac Surg 74:1848.e1841-1848.e1814
Kim DW, Yoon ES, Ji YH, Park SH, Lee BI, Dhong ES (2011) Vascular complications of hyaluronic acid fillers and the role of hyaluronidase in management. J Plast Reconstr Aesthet Surg 64:1590–1595
Nie F, Xie H, Wang G, An Y (2019) Risk comparison of filler embolism between polymethyl methacrylate (PMMA) and hyaluronic acid (HA). Aesthet Plast Surg 43:853–860
Yildirim G, Alpak H, Sayin I, Onar V (2013) Biocompatibility of high-density porous polyethylene covered with fascia lata in dorsal nasal augmentation-an experimental study in rabbits. Turk J Vet Anim Sci 8:326–328
Park MK, Kim HK, Park KY, Li K, Seo SJ, Hong CK (2011) Complications following BellaGenTM injection. Ann Dermatol 23:S306-309
Read-Fuller AM, Yates DM, Radwan A, Schrodt AM, Finn RA (2018) The use of allogeneic cartilage for grafting in functional and reconstructive rhinoplasty. J Oral Maxillofac Surg 76:1560.e1561-1560.e1567
Vila PM, Jeanpierre LM, Rizzi CJ, Yaeger LH, Chi JJ (2020) Comparison of autologous vs homologous costal cartilage grafts in dorsal augmentation rhinoplasty: a systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg. https://doi.org/10.1001/jamaoto.2019.4787
Kon E, Filardo G, Di Matteo B, Perdisa F, Marcacci M (2013) Matrix assisted autologous chondrocyte transplantation for cartilage treatment: a systematic review. Bone Jt Res 2:18–25
Tohyama H, Yasuda K, Minami A, Majima T, Iwasaki N, Muneta T, Sekiya I, Yagishita K, Takahashi S, Kurokouchi K, Uchio Y, Iwasa J, Deie M, Adachi N, Sugawara K, Ochi M (2009) Atelocollagen-associated autologous chondrocyte implantation for the repair of chondral defects of the knee: a prospective multicenter clinical trial in Japan. J Orthop Sci 14:579–588
Sito G, Manzoni V, Sommariva R (2019) Vascular complications after facial filler injection: a literature review and meta-analysis. J clin aesthet dermatol 12:E65–E72
Lee KH (2016) Infection in the nasal tip caused by acellular dermal matrix. Plast reconstr surg Glob op 3:e581–e581
Sherris DA, Oriel BS (2011) Human acellular dermal matrix grafts for rhinoplasty. Aesthet Surg J 31:95s–100s
La Padula S, Hersant B, Helynck P, Mezi SidAhmed M, Meningaud JP (2020) Proposal of a noninvasive method to reduce injection-related bruising in aesthetic medicine: transillumination. Aesthet Plast Surg 44:530–534
Donald PJ (1986) Cartilage grafting in facial reconstruction with special consideration of irradiated grafts. Laryngoscope 96:786–807
Quatela VC, Chow J (2008) Synthetic facial implants. Fac Plast Surg Clin North Am 16(1–10):v
Gelfer A, Carruthers A, Carruthers J, Jang F, Bernstein SC (2007) The natural history of polymethylmethacrylate microspheres granulomas. Dermatol Surg 33:614–620
Limongi RM, Tao J, Borba A, Pereira F, Pimentel AR, Akaishi P, Velasco e Cruz AA, (2016) Complications and management of polymethylmethacrylate (PMMA) injections to the midface. Aesthet Surg J 36:132–135
Funding
None
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human or Animal Rights
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Informed Consent
Informed consent is not applicable to this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hwang, S.H., Kim, D.H., Nam, B.M. et al. Efficacy and Safety of Lyophilized Articular Cartilage Matrix as an Injectable Facial Filler. Aesth Plast Surg 45, 1266–1272 (2021). https://doi.org/10.1007/s00266-020-02017-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-020-02017-1