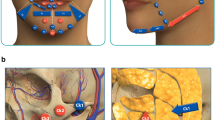
Abstract
Background
The polyacrylamide hydrogel Aquamid® has been used as a permanent filler to enhance facial soft tissue volume and correct wrinkles since 2001. Various long-term studies have proved the safety of the product. Nonetheless, if complications such as migration occur, they can be difficult to treat.
Methods
Eleven patients suffering from late-onset complications after taking Aquamid® injections in the lips underwent product removal and subsequent labial reconstruction between 2009 and 2017. The reconstruction was performed using a modified bikini reduction technique combined, in eight cases, with immediate autologous fat grafting.
Results
In all the patients, general fibrosis and a diffused distribution of the product within all three layers of the lips resulted in the need for labial reconstruction. Migration, as far as in the mucosa and perioral skin, accounted for macroscopically visible yellowish accumulations. In ten out of eleven cases, an individually modified bikini reduction technique, with or without any combination of autologous fat grafting, led to an esthetically satisfying result. One patient developed a severe upper lip necrosis.
Conclusion
Contradictory to several previous studies attesting to the lack of migration after Aquamid® application to the lips, capsule formation around the product is impaired, allowing for migration even years after the injection. Product aspiration is not possible in these cases, thus necessitating complex lip reconstruction. Bikini reduction and fat grafting are valuable tools for labial reconstruction. Product residuals within the mucosa have to be accepted. Special care has to be taken while treating smokers.
Level of Evidence IV
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.








Similar content being viewed by others
References
McCollister D, Hake C, Sadek S, Rowe V (1965) Toxicological investigations of polyacrylamides. Toxicol Appl 7:639
Smith E, Oehma F (1991) Acrylamide and polyacrylamide:a review of production, use, environmental fate and neurotoxicity. Rev Environ Health 9:215
Christensen L, Breiting V, Aasted A, Jørgensen A, Kebuladze I (2003) Long term effects of polyacrylamide hydrogel on human breast tissue. Plast Reconstr Surg 111:1883
Caulfield M, Qiao G, Solomon D (2002) Some aspects of the properties and degradation of polyacrylamides. Chem Rev 102:3067–3084
Limited SEP (2017) http://www.specialityeuropeanpharma.com/contura.php
Wolters M, Lampe H (2009) Prospective multicenter study for evaluation of safety, efficacy, and esthetic results of cross-linked polyacrylamide hydrogel in 81 patients. Dermatol Surg 35:338–343
Pallua N, Wolter T (2010) A 10-year assessment of safety and aesthetic results after facial soft-tissue augmentation with polyacrylamide hydrogel (Aquamid): a prospective multicenter study of 251 patients. Plast Reconstr Surg 125:1797–1804
Kalantar- Hormozi A, Mozafari N, Rasti M (2008) Adverse effects after use of polyacrylamide gel as a facial soft tissue filler. Aesthet Surg J 28:139–142
Nygart J, Nygart V, Borggren M, Tvede M (2014) Effect of prophylactic antibiotics on polyacrylamide gel saftey in facial augmentation. J Drugs Dermatol 13:571–573
Negredo E, Puig J, Ornelas A, Echeverría P, Bonjoch A, Estany C, Higueras C, Gonzalez-Mestre V, Clotet B (2009) Ten-year safety with polyacrylamide gel used to correct facial lipoatrophy in HIV-infected patients. AIDS Res Hum Retrovir 25:451–455
Von Buelow S, Pallua N (2005) Efficacy and safety of polyacrylamide hydrogel for facial soft tissue augmentation. Plast Reconstr Surg 116:1137–1146
Christensen L, Breitling V, Janssen M, Vuust J, Hogdall E (2005) Adverse reactions to injectable soft tissue permanent fillers. Aesthet Plast Surg 29:34–48
Wolter T, Pallua N (2010) Removal of the permanent filler polyacrylamide hydrogel (Aquamid) is possible and easy even after several years. Plast Reconstr Surg 126:138e–139e
Fanous N, Brousseau V, Yoskovitch A (2007) The bikini lip reduction: a detailed approach to hypertrophic lips. Can J Plast Surg 15:205–210
Yoskovitch A, Fanous N (2003) Correction of thin lips: a 17-year follow-up of the original technique. Plast Reconstr Surg 112:670–675
Botti G, Botti C, Cella A (2002) A simple surgical remedy for iatrogenic excessively thick lips. Plast Reconstr Surg 110:1329–1334
Bello G, Jackson I, Keskin M, Kelly C, Dajani K, Studinger R, Kim EM, Lincoln D, Silberberg B, Lee A (2007) The use of Polyacrylamide gel in soft-tissue augmentation: an experimental assessment. Plast Reconst Surg 119:1326–1336
Zarini E, Supino R, Pratesi G, Laccabue D, Tortoreto M, Scanziani E, Ghisleni G, Paltrinieri S, Tunesi G, Nava M (2004) Biocompatibility and tissue interactions of a new filler material for medical use. Plast Reconstr Surg 114:934–942
De Cassia Novaes W, Berg A (2003) Experiences with a new nonbiodegradable hydrogel (Aquamid): a pilot study. Aesthet Plast Surg 27:276–380
Cheng NX, Xu SL, Deng H, Ding XB, Zhang XM, Wu DH, Zhong H, Sun ZH (2006) Migration of implants: a problem with injectable polyacrylamide gel in aesthetic plastic surgery. Aesthet Plast Surg 30:215–225
Xu XG, Zhu ZX, Jin BY (2002) Dynamics study of polyacrylamide hydrogel in vivo. J Pract Aesthet Plast Surg 13:41–43
Christensen L (2007) Normal and pathologic tissue reactions to soft tissue gel fillers. Dermatol Surg 33:168–175
Christensen L, Breiting V, Bjarnsholt T, Eickhardt S, Høgdall E, Janssen M, Pallua N, Zaat SA (2013) Bacterial infection as a likely cause of adverse reactions to polyacrylamide hydrogel fillers in cosmetic surgery. Clin Infect Dis 56:1438–1444
El-Shafey S (2008) Complications from repeated injection or puncture of old polyacrylamide gel implant sites: case reports. Aesthet Plast Surg 32:162–165
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and Animal Participants
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee, and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
All participants gave their informed consent in writing for their inclusion in this retrospective, clinical case study.
Rights and permissions
About this article
Cite this article
Kästner, S., Gonser, P., Paprottka, F. et al. Removal of Polyacrylamide Gel (Aquamid®) from the Lip as a Solution for Late-Onset Complications: Our 8-Year Experience. Aesth Plast Surg 42, 791–797 (2018). https://doi.org/10.1007/s00266-018-1114-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-018-1114-1