Abstract
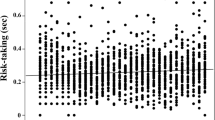
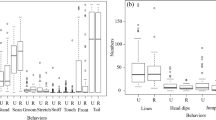
Environmental variation strongly influences individual life histories, behavior, and immunity. For many animals living in non-tropical regions, environmental variation due to season is very important and can be reliably predicted by photoperiod. By manipulating the pre- and post-natal photoperiod for cavies (Cavia aperea), we simulated different birth seasons that go along with predictable differences in life history. Offspring born into simulated spring conditions mature earlier, indicating a fast pace of life compared to offspring born into simulated fall. We investigated the development of two innate (titers of natural antibodies and complement) and one adaptive (immunoglobulin G) immune effectors across multiple life stages. In addition, we tested if three behaviors, indicating risk-taking and stress-coping, differed between seasons. Finally, we tested if behaviors and immune traits correlate with each other as predicted by the pace-of-life syndrome hypothesis. The season of birth influenced average trait expression of both immunity and behavior. Boldness, indicating risk-taking behavior, correlated positively with the titers of natural antibodies, fitting the predictions of the pace-of-life syndrome (POLS). However, fearlessness, indicating how an individual copes with a stressful situation, correlated positively with immunoglobulin G concentrations, against the predictions of the POLS. Together, our results show that behaviors and immune traits react to differences in expected life histories, but do not show the predicted covariation. We conclude that proximate mechanisms and different functions of traits need to be considered to predict the nature of behavior-immune associations.
Significance statement
Individual life histories, behavior, and immunity are strongly influenced by the environment individual’s experience during early life. Recent theory predicts correlations between behavior and immunity to arise as a consequence of different life histories. By simulating different birth seasons, we induced different life history expectations as well as a season-dependent development of immunity and behavior. However, correlations between behavior and immunity do not fit theoretical predictions, highlighting the need for more detailed theory.



Similar content being viewed by others
References
Ashley NT, Demas GE (2017) Neuroendocrine-immune circuits, phenotypes, and interactions. Horm Behav 87:25–34. https://doi.org/10.1016/j.yhbeh.2016.10.004
Barber I, Dingemanse NJ (2010) Parasitism and the evolutionary ecology of animal personality. Philos Trans R Soc B 365:4077–4088. https://doi.org/10.1098/rstb.2010.0182
Boyer N, Réale D, Marmet J, Pisanu B, Chapuis J-L (2010) Personality, space use and tick load in an introduced population of Siberian chipmunks Tamias sibiricus. J Anim Ecol 79:538–547. https://doi.org/10.1111/j.1365-2656.2010.01659.x
Castro Conde I, de Uña Álvarez J (2013) SGoF multitesting method under the Bayesian paradigm. Discussion Papers in Statistics and Operation Research, Report 13/06. Statistics and OR Department. University of Vigo
Diagne C, Ribas A, Charbonnes N et al (2016) Parasites and invasions: changes in gastrointestinal helminth assemblages in invasive and native rodents in Senegal. Int J Parasitol 46:857–869
Dosmann A, Brooks KC, Mateo JM (2015) Evidence for a mechanism of phenotypic integration of behaviour and innate immunity in a wild rodent: implications for animal personality and ecological immunology. Anim Behav 101:179–189. https://doi.org/10.1016/j.anbehav.2014.12.026
Eccard J, Herde A (2013) Seasonal variation in the behaviour of a short-lived rodent. BMC Ecol 13:43. https://doi.org/10.1186/1472-6785-13-43
Finkemeier M-A, Trillmich F, Guenther A (2016) Match-mismatch experiments using photoperiod expose developmental plasticity of personality traits. Ethology 122:80–93
Gelman A, Rubin DB (1992) Inference from iterative simulation using multiple sequences. Stat Sci 7:457e511
Gilot-Fromont E, Jégo M, Bonenfant C, Gibert P, Rannou B, Klein F, Gaillard J-M (2012) Immune phenotype and body condition in roe deer: individuals with high body condition have different, not stronger immunity. PLoS One 7:e45576
Goldman BD (2001) Mammalian photoperiodic system: formal properties and neuroendocrine mechanisms of photoperiodic time measurement. J Biol Rhythm 16:283–301. https://doi.org/10.1177/074873001129001980
Groothuis TGG, Maestripieri D (2013) Parental influences on offspring personality. In: Carere C, Maestripieri D (eds) Animal personalities. The University of Chicago Press, Chicago, pp 317–352
Groothuis TGG, Taborsky B (2015) Introducing biological realism into the study of developmental plasticity in behaviour. Front Zool 12:S6. https://doi.org/10.1186/1742-9994-12-S1-S6
Guenther A, Trillmich F (2013) Photoperiod influences the behavioral and physiological phenotype during ontogeny. Behav Ecol 24:402–411. https://doi.org/10.1093/beheco/ars177
Guenther A, Trillmich F (2015) Within-litter differences in personality and physiology relate to size differences among siblings in cavies. Physiol Behav 145:22–28. https://doi.org/10.1016/j.physbeh.2015.03.026
Guenther A, Finkemeier M-A, Trillmich F (2014a) The ontogeny of personality in the wild guinea pig. Anim Behav 90:131–139. https://doi.org/10.1016/j.anbehav.2014.01.032
Guenther A, Palme R, Dersen M, Kaiser S, Trillmich F (2014b) Photoperiodic effects on reproductive development in male cavies (Cavia aperea). Physiol Behav 123:142–147. https://doi.org/10.1016/j.physbeh.2013.10.017
Hadfield JD (2010) MCMC methods for multi-response generalized linear mixed models: the MCMCglmm R package. J Stat Softw 33:1–22
Hasselquist D, Tobler M, Nilsson J-Å (2012) Maternal modulation of offspring immune function in vertebrates. In: Demas GE, Nelson RJ (eds) Ecoimmunology. Oxford University Press, Oxford, pp 165–224
Heinrich SK, Hofer H, Courtiol A, Melzheimer J, Dehnhard M, Czirják GÁ, Wachter B (2017) Cheetah have a stronger constitutive innate immunity than leopards. Sci Rep 7:44837. https://doi.org/10.1038/srep44837
Horton TH (1984) Growth and reproductive development of male Microtus montanus is affected by the prenatal photoperiod. Biol Reprod 504:499–504
Jacques-Hamilton R, Hall ML, Buttemer WA, Matson KD, Gonҫalves da Silva A, Mulder RA, Peters A (2017) Personality and innate immune defenses in a wild bird: evidence for the pace-of-life hypothesis. Horm Behav 88:31–40. https://doi.org/10.1016/j.yhbeh.2016.09.005
Khokhlova IS, Spinu S, Krasnov BR, Degen AA (2004) Immune response to fleas in a wild desert rodent: effect of parasite species, parasite burden, sex of host and host parasitological experience. J Exp Biol 207:2725–2733. https://doi.org/10.1242/jeb.01090
Koolhaas JM (2008) Coping style and immunity in animals: making sense of individual variation. Brain Behav Immun 22:662–667. https://doi.org/10.1016/j.bbi.2007.11.006
Koolhaas JM, Korte SM, de Boer SF, van der Vegt B, van Reenen CG, Hopster H, de Jong IC, Ruis MAW, Blokhuis HJ (1999) Coping styles in animals: current status in behavior and stress-physiology. Neurosci Biobehav Rev 23:925–935
Koolhaas JM, de Boer SF, Coppens CM, Buwalda B (2010) Neuroendocrinology of coping styles: towards understanding the biology of individual variation. Front Neuroendocrinol 31:307–321. https://doi.org/10.1016/j.yfrne.2010.04.001
Kortet R, Hedrick AV, Vainikka A (2010) Parasitism, predation and the evolution of animal personalities. Ecol Lett 13:1449–1458. https://doi.org/10.1111/j.1461-0248.2010.01536.x
Krams I, Vrublevska J, Cirule D, Kivleniece I, Krama T (2013) Stress, behaviour and immunity in wild-caught wintering great tits (Parus major). Ethology 119:397–406. https://doi.org/10.1111/eth.12075
Martin LB, Weil ZM, Nelson RJ (2008) Seasonal changes in vertebrate immune activity: mediation by physiological trade-offs. Philos Trans R Soc B 363:321–339. https://doi.org/10.1098/rstb.2007.2142
Matson KD, Ricklefs RE, Klasing KC (2005) A hemolysis–hemagglutination assay for characterizing constitutive innate humoral immunity in wild and domestic birds. Dev Comp Immunol 29:275–286
Nelson R, Demas G (1996) Seasonal changes in immune function. Q Rev Biol 71:511–548
Nelson RJ, Demas GE (2004) Seasonal patterns of stress, disease, and sickness responses. Curr Dir Psychol Sci 13:198–201
Pap PL, Czirják GÁ, Vágási CI, Barta Z, Hasselquist D (2010a) Sexual dimorphism in immune function changes during the annual cycle in the house sparrows. Naturwissenschaften 97:891–901
Pap PL, Vágási CI, Tökölyi J, Czirják GÁ, Barta Z (2010b) Variation in haematological indices and immune function during the annual cycle in the great tit Parus major. Ardea 98:105–112
Pap PL, Vágási CI, Vincze O, Osváth G, Veres-Szászka J, Czirják GÁ (2015) Physiological pace of life: the link between constitutive immunity, developmental period, and metabolic rate in European birds. Oecologia 177:147–158
Pilorz V, Jäckel M, Knudsen K, Trillmich F (2005) The cost of a specific immune response in young guinea pigs. Physiol Behav 85:205–211
Prendergast BJ, Nelson RJ, Zucker I (2002) Mammalian seasonal rhythms: behavior and neuroendocrine substrates. Horm Brain Behav 2:93–156
Pyter LM, Nelson RJ (2006) Enduring effects of photoperiod on affective behaviors in Siberian hamsters (Phodopus sungorus). Behav Neurosci 120:125–134. https://doi.org/10.1037/0735-7044.120.1.125
R Core Team (2016) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. URL https://www.R-project.org/
Réale D, Reader S, Sol D, Mcdougall PT, Dingemanse NJ (2007) Integrating animal temperament within ecology and evolution. Biol Rev 82:291–318. https://doi.org/10.1111/j.1469-185X.2007.00010.x
Réale D, Garant D, Humphries MM, Bergeron P, Careau V, Montiglio P-O (2010) Personality and the emergence of the pace-of-life syndrome concept at the population level. Philos Trans R Soc B 365:4051–4063. https://doi.org/10.1098/rstb.2010.0208
Ross PS, Pohajdak B, Bowen WD, Addison RF (1993) Immune function in free-ranging harbor seal (Phoca vitulina) mothers and their pups during lactation. J Wildl Dis 29:21–29
Rübensam K, Hribal R, Jewgenow K, Guenther A (2015) Seasonally different reproductive investment in a medium-sized rodent (Cavia aperea). Theriogenology 84:639–644. https://doi.org/10.1016/j.theriogenology.2015.04.023
Schneeberger K, Courtiol A, Czirják GÁ, Voigt CC (2014) Immune profile predicts survival and reflects senescence in a small, long-lived mammal, the greater sac-winged bat (Saccopteryx bilineata). PLoS One 9:e108268. https://doi.org/10.1371/journal.pone.0108268
Schumann K, Guenther A, Göritz F, Jewgenow K (2014) Characterization of fetal growth by repeated ultrasound measurements in the wild guinea pig (Cavia aperea). Theriogenology 82:490–494. https://doi.org/10.1016/j.theriogenology.2014.05.007
Sild E, Sepp T, Hõrak P (2011) Behavioural trait covaries with immune responsiveness in a wild passerine. Brain Behav Immun 25:1349–1354. https://doi.org/10.1016/j.bbi.2011.03.020
Thorbecke GJ (1964) Development of immune globulin formation in fetal, newborn and immature guinea pigs. Fed Proc 23:346
Trillmich F, Mueller B, Kaiser S, Krause J (2009) Puberty in female cavies (Cavia aperea) is affected by photoperiod and social conditions. Physiol Behav 96:476–480. https://doi.org/10.1016/j.physbeh.2008.11.014
Weil ZM, Pyter LM, Martin LB, Nelson RJ (2006) Perinatal photoperiod organizes adult immune responses in Siberian hamsters (Phodopus sungorus). Am J Physiol-Reg I 290:R1714–R1719. https://doi.org/10.1152/ajpregu.00869.2005
Zylberberg M, Klasing KC, Hahn TP (2014) In house finches, Haemorhous mexicanus, risk takers invest more in innate immune function. Anim Behav 89:115–122. https://doi.org/10.1016/j.anbehav.2013.12.021
Acknowledgments
We are grateful to the animal keepers who took care of the cavies and to Katja Pohle for her help during laboratory analysis. We would like to thank two anonymous referees for their valuable comments on the manuscript.
Funding
This research was supported by funds from the DFG (Deutsche Forschungsgemeinschaft (FOR1232, TR105/22-2)) and the Leibniz Institute for Zoo and Wildlife Research Berlin.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and institutional guidelines for the care and use of animals were followed. All procedures involving animals were in accordance with the ethical standards of the institution at which the studies were conducted. Animal facilities were approved (dated 18 April 2002) for keeping and breeding cavies for research purposes by the local government authority responsible for health, veterinary, and food monitoring (Gesundheits-, Veterinär-, und Lebensmittelüberwachungsamt). The experiments were performed under license 84-02.05.20.12.246 LANUV, NRW, Germany.
Additional information
Communicated by C. Soulsbury
Electronic supplementary material
ESM 1
(DOCX 72 kb)
Rights and permissions
About this article
Cite this article
Guenther, A., Trillmich, F. & Czirják, G.Á. Seasonal effects on behavior and immunity in a precocial rodent. Behav Ecol Sociobiol 72, 115 (2018). https://doi.org/10.1007/s00265-018-2513-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-018-2513-7