Abstract
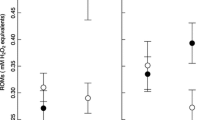
Oxidative stress has been suggested as a mechanism underlying the costs of reproduction and life history trade-offs. Reproductive activities may lead to high production of pro-oxidants, whose activity can generate oxidative damage when not countered by adequate antioxidant defenses. Because inter-individual differences in the efficiency of the antioxidant system are influenced by an individual’s diet, food availability experienced during reproduction may affect the females’ antioxidant status and, in birds, their ability to transfer antioxidants into their eggs. Moreover, a female’s ability to cope with oxidative stress has been suggested to influence pigment deposition in the eggshell, suggesting a possible signaling function of eggshell maculation. Here we performed a food supplementation experiment in a natural population of great tits (Parus major) in order to investigate how nutritional conditions experienced during the egg laying period affect the female’s oxidative status and egg investment and how maternal oxidative status and egg antioxidant protection relate to eggshell pigmentation. We show that food-supplemented females had lower oxidative damage levels (ROMs) than non-food-supplemented females. Furthermore, a female’s ROMs levels were negatively associated with the levels of yolk antioxidant protection in her eggs, but this negative association was only significant in non-food-supplemented females. This suggests that oxidative stress experienced during reproduction influences the allocation of antioxidants into the eggs. Moreover, we observed a positive relationship between eggshell pigment distribution and maternal and yolk antioxidant protection, suggesting that eggshell pigmentation is a cue of female (and offspring) quality.



Similar content being viewed by others
References
Afonso S, Vanore G, Batlle A (1999) Protoporphyrin IX and oxidative stress. Free Radic Res 31:161–170
Alan RR, McWilliams SR (2013) Oxidative stress, circulating antioxidants, and dietary preferences in songbirds. Comp Biochem Physiol B 164:185–193
Alberti A, Bolognini L, Macciantelli D, Caratelli M (2000) The radical cation of N, N-diethyl-para-paraphenylendiamine: a possible indicator of oxidative stress in biological samples. Res Chem Intermed 26:253–267
Alonso-Alvarez C, Bertrand S, Devevey G, Prost J, Faivre B, Sorci G (2004) Increased susceptibility to oxidative stress as a proximate cost of reproduction. Ecol Lett 7:363–368
Arthur JR (2000) The glutathione peroxidases. Cell Mol Life Sci 57:1825–1835
Berthouly A, Helfenstein F, Richner H (2007) Cellular immune response, stress resistance and competitiveness in nestling great tits in relation to maternally transmitted carotenoids. Funct Ecol 21:335–343
Bindoli A, Fukuto JM, Forman HJ (2008) Thiol chemistry in peroxidase catalysis and redox signaling. Antioxid Redox Signal 10:1549–1564
Blount JD, Surai PF, Nager RG, Houston DC, Møller AP, Trewby ML, Kennedy MW (2002) Carotenoids and egg quality in the lesser black-backed gull Larus fuscus: a supplemental feeding study of maternal effects. Proc R Soc Lond B 269:29–36
Bryant DM (1997) Energy expenditure in wild birds. Proc Nutr Soc 56:1025–1039
Catoni C, Peters A, Schaefer HM (2008) Life history trade-offs are influenced by the diversity, availability and interactions of dietary antioxidants. Anim Behav 76:1107–1119
Christe P, Glaizot O, Strepparava N, Devevey G, Fumagalli L (2012) Twofold cost of reproduction: an increase in parental effort leads to higher malarial parasitaemia and to a decrease in resistance to oxidative stress. Proc R Soc Lond B 279:1142–1149
Cohen AA, McGraw KJ, Robinson WD (2009) Serum antioxidant levels in wild birds vary in relation to diet, season, life history strategy, and species. Oecologia 161:673–683
Costantini D (2008) Oxidative stress in ecology and evolution: lessons from avian studies. Ecol Lett 11:1238–1251
Costantini D (2010) Complex trade-offs in the pigeon (Columba livia): egg antioxidant capacity and female serum oxidative status in relation to diet quality. J Comp Physiol B 180:731–739
Costantini D, Monaghan P, Metcalfe NB (2011) Biochemical integration of blood redox state in captive zebra finches (Taeniopygia guttata). J Exp Biol 214:1148–1152
Costantini D, Meillere A, Carravieri A et al (2014) Oxidative stress in relation to reproduction, contaminants, gender and age in a long-lived seabird. Oecologia 175:1107–1116
De Coster G, De Neve L, Lens L (2013) Intra-clutch variation in avian eggshell pigmentation covaries with female quality. J Ornithol 154:1057–1065
de Heij ME, van der Graaf AJ, Hafner D, Tinbergen JM (2007) Metabolic rate of nocturnal incubation in female great tits, Parus major, in relation to clutch size measured in a natural environment. J Exp Biol 210:2006–2012
Fechner A, Bohme CC, Gromer S, Funk M, Schirmer RH, Becker K (2001) Antioxidant status and nitric oxide in the malnutrition syndrome kwashiorkor. Pediatr Res 49:237–243
Feoli AM, Siqueira IR, Almeida L, Tramontina AC, Vanzella C, Sbaraini S, Schweigert ID, Netto CA, Perry MLS, Goncalves CA (2006) Effects of protein malnutrition on oxidative status in rat brain. Nutrition 22:160–165
Gibb J (1957) Food requirements and other observations on captive tits. Bird Study 4:207–215
Giordano M, Groothuis TGG, Tschirren B (2014) Interactions between prenatal maternal effects and posthatching conditions in a wild bird population. Behav Ecol 25:1459–1466
Giordano M, Costantini D, Tschirren B (2015) Sex-specific effects of prenatal and postnatal nutritional conditions on the oxidative status of great tit nestlings. Oecologia 177:123–131
Gosler AG, Barnett PR, Reynolds SJ (2000) Inheritance and variation in eggshell patterning in the great tit Parus major. Proc R Soc Lond B 267:2469–2473
Gosler AG, Higham JP, Reynolds SJ (2005) Why are birds’ eggs speckled? Ecol Lett 8:1105–1113
Halliwell B, Gutteridge JMC (2007) Free radicals in biology and medicine, 4th edn. Oxford University Press, Oxford
Hanley D, Heiber G, Dearborn DC (2008) Testing an assumption of the sexual-signaling hypothesis: does blue-green egg reflect maternal antioxidant capacity? Condor 110:767–771
Hansen M, Flatt T, Aguilaniu H (2013) Reproduction, fat metabolism, and life span: what is the connection? Cell Metab 17:10–19
Harshman LG, Zera AJ (2007) The cost of reproduction: the devil in the details. Trends Ecol Evol 22:80–86
Hulbert AJ, Pamplona R, Buffenstein R, Buttemer WA (2007) Life and death: metabolic rate, membrane composition, and life span of animals. Physiol Rev 87:1175–1213
Isaksson C, Johansson A, Andersson S (2008) Egg yolk carotenoids in relation to habitat and reproductive investment in the great tit Parus major. Physiol Biochem Zool 81:112–118
Kaur H, Hughes MN, Green CJ, Naughton P, Foresti R, Motterlini R (2003) Interaction of bilirubin and biliverdin with reactive nitrogen species. FEBS Lett 543:113–119
Kennedy GY, Vevers HG (1976) Survey of avian eggshell pigments. Comp Biochem Physiol B 55:117–123
Li J, Wang H, Stoner GD, Bray TM (2002) Dietary supplementation with cysteine prodrugs selectively restores tissue glutathione levels and redox status in protein-malnourished mice. J Nutr Biochem 13:625–633
Marques de Sá JP (2007) Applied statistics. Springer, Berlin
Marri V, Richner H (2014) Yolk carotenoids increase fledging success in great tit nestlings. Oecologia 176:371–377
Martínez-de la Puente J, Merino S, Moreno J, Tomás G, Morales J, Lobato E, García-Fraile S, Martínez J (2007) Are eggshell spottiness and colour indicators of health and condition in blue tits Cyanistes caeruleus? J Avian Biol 38:377–384
McGraw KJ, Adkins-Regan E, Parker RS (2005) Maternally derived carotenoid pigments affect offspring survival, sex ratio, and sexual attractiveness in a colorful songbird. Naturwissenschaften 92:375–380
Metcalfe NB, Alonso-Alvarez C (2010) Oxidative stress as a life-history constraint: the role of reactive oxygen species in shaping phenotypes from conception to death. Funct Ecol 24:984–996
Metcalfe NB, Monaghan P (2013) Does reproduction cause oxidative stress? An open question. Trends Ecol Evol 28:347–350
Morales J, Velando A, Moreno J (2008) Pigment allocation to eggs decreases plasma antioxidants in a songbird. Behav Ecol Sociobiol 63:227–233
Moreno J, Osorno JL (2003) Avian egg colour and sexual selection: does eggshell pigmentation reflect female condition and genetic quality? Ecol Lett 6:803–806
Moreno J, Sanz JJ (1994) The relationship between energy-expenditure during incubation and clutch size in the pied flycatcher Ficedula hypoleuca. J Avian Biol 25:125–130
Nikolaidis MG, Mougios V (2004) Effects of exercise on the fatty-acid composition of blood and tissue lipids. Sports Med 34:1051–1076
Nilsson JA, Råberg L (2001) The resting metabolic cost of egg laying and nestling feeding in great tits. Oecologia 128:187–192
Nord A, Sandell MI, Nilsson JA (2010) Female zebra finches compromise clutch temperature in energetically demanding incubation conditions. Funct Ecol 24:1031–1036
Olsson M, Healey M, Perrin C, Wilson M, Tobler M (2012) Sex-specific SOD levels and DNA damage in painted dragon lizards (Ctenophorus pictus). Oecologia 170:917–924
Paglia DE, Valentine WN (1967) Studies on quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Perrins CM (1996) Eggs, egg formation and the timing of breeding. Ibis 138:2–15
Porter NA, Caldwell SE, Mills KA (1995) Mechanisms of free-radical oxidation of unsaturated lipids. Lipids 30:277–290
Postma E, Siitari H, Schwabl H, Richner H, Tschirren B (2014) The multivariate egg: quantifying within- and among-clutch correlations between maternally derived yolk immunoglobulins and yolk androgens using multivariate mixed models. Oecologia 174:631–638
Powers SK, Jackson MJ (2008) Exercise-induced oxidative stress: cellular mechanisms and impact on muscle force production. Physiol Rev 88:1243–1276
Remeš V, Matysiokova B, Klejdus B (2011) Egg yolk antioxidant deposition as a function of parental ornamentation, age, and environment in great tits Parus major. J Avian Biol 42:387–396
Reynolds SJ, Martin GR, Cassey P (2009) Is sexual selection blurring the functional significance of eggshell coloration hypotheses? Anim Behav 78:209–215
Rindler PM, Plafker SM, Szweda LI, Kinter M (2013) High dietary fat selectively increases catalase expression within cardiac mitochondria. J Biol Chem 288:1979–1990
Saino N, Bertacche V, Ferrari RP, Martinelli R, Møller AP, Stradi R (2002) Carotenoid concentration in barn swallow eggs is influenced by laying order, maternal infection and paternal ornamentation. Proc R Soc Lond B 269:1729–1733
Sanz JJ, García-Navas V (2009) Eggshell pigmentation pattern in relation to breeding performance of blue tits Cyanistes caeruleus. J Anim Ecol 78:31–41
SAS Institute (1989–2007) JMP, Version 10. Cary, NC
Sies H (1991) The role of reactive oxygen species in biological processes. Klin Wochenschr 69:965–968
Stearns SC (1992) The evolution of life histories. Oxford University Press, New York
Surai PF (2002) Natural antioxidants in avian nutrition and reproduction. Nottingham University Press, Nottingham
Surai PF, Noble RC, Speake BK (1996) Tissue-specific differences in antioxidant distribution and susceptibility to lipid peroxidation during development of the chick embryo. Biochim Biophys Acta 1304:1–10
Török J, Hargitai R, Hegyi G, Matus Z, Michl G, Peczely P, Rosivall B, Toth G (2007) Carotenoids in the egg yolks of collared flycatchers (Ficedula albicollis) in relation to parental quality, environmental factors and laying order. Behav Ecol Sociobiol 61:541–550
Travers M, Clinchy M, Zanette L, Boonstra R, Williams TD (2010) Indirect predator effects on clutch size and the cost of egg production. Ecol Lett 13:980–988
Tschirren B, Richner H, Schwabl H (2004) Ectoparasite-modulated deposition of maternal androgens in great tit eggs. Proc R Soc Lond B 271:1371–1375
van de Crommenacker J, Horrocks NPC, Versteegh MA, Komdeur J, Tieleman BI, Matson KD (2010) Effects of immune supplementation and immune challenge on oxidative status and physiology in a model bird: implications for ecologists. J Exp Biol 213:3527–3535
Wiersma P, Selman C, Speakman JR, Verhulst S (2004) Birds sacrifice oxidative protection for reproduction. Proc R Soc Lond B 271:S360–S363
Williams TD (2005) Mechanisms underlying the costs of egg production. Bioscience 55:39–48
Wilson HR (1997) Effects of maternal nutrition on hatchability. Poult Sci 76:134–143
Acknowledgments
We thank Martina Keller, Kathrin Näpflin and Jamal Hanafi for field assistance and help in the lab and three anonymous reviewers for comments on the manuscript. This research was financially supported by the Swiss National Science Foundation (grant PP00P3_128386 to BT).
Ethical standards
All procedures were conducted under license from the Veterinary Office of the Canton of Zurich, Switzerland (195/2010) and the Federal Office for the Environment, Switzerland (324).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K. McGraw
Rights and permissions
About this article
Cite this article
Giordano, M., Costantini, D., Pick, J.L. et al. Female oxidative status, egg antioxidant protection and eggshell pigmentation: a supplemental feeding experiment in great tits. Behav Ecol Sociobiol 69, 777–785 (2015). https://doi.org/10.1007/s00265-015-1893-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-015-1893-1