Abstract
Purpose
The reason for the formation of an atrophic non-union is not clear and an altered vascularization as well as a deregulation of endogenous growth factors is hypothesized. To obtain more information, we analysed human non-union tissue regarding the histology and quantity of several growth factors.
Methods
Tissue from patients with an atrophic non-union (n = 44) or with a healed fracture (n = 13) was analysed. Using histological and immunohistochemical methods the tissue composition was investigated. On the protein level the amount of several growth factors important for bone healing was analysed.
Results
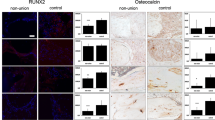
The tissue composition was very inhomogeneous containing fibrous, cartilaginous and bony tissue. Vessels were present in all investigated samples without a difference between the tissue from non-union and control patients. The growth factor BMP-2 was below the detection limit in all samples, whereas IL-6 and IGF-I were measured only in a few samples of both groups. TGF-ß1, VEGF-A and BMP-4 were detectable in the majority of the samples of both groups with a high variability in the amount but no difference between the groups. The quantity of both growth factors, BMP-7 and PDGF-AB, was significantly lower in the non-union tissue compared to the healed controls.
Conclusion
The reduced quantity of BMP-7 and PDGF-AB might be responsible for the impaired healing. Further studies analysing material from more patients and investigating the early healing phases, however, are necessary to obtain further information and consequently improve healing strategies.



Similar content being viewed by others
References
Giannoudis PV, Einhorn TA, Schmidmaier G, Marsh D (2008) The diamond concept—open questions. Injury 39(Suppl 2):S5–S8
Ferguson C, Alpern E, Miclau T, Helms JA (1999) Does adult fracture repair recapitulate embryonic skeletal formation? Mech Dev 87:57–66
Ai-Aql ZS, Alagl AS, Graves DT, Gerstenfeld LC, Einhorn TA (2008) Molecular mechanisms controlling bone formation during fracture healing and distraction osteogenesis. J Dent Res 87:107–118
Kon T, Cho TJ, Aizawa T, Yamazaki M, Nooh N, Graves D et al (2001) Expression of osteoprotegerin, receptor activator of NF-kappaB ligand (osteoprotegerin ligand) and related proinflammatory cytokines during fracture healing. J Bone Miner Res 16:1004–1014
Ruschke K, Hiepen C, Becker J, Knaus P (2012) BMPs are mediators in tissue crosstalk of the regenerating musculoskeletal system. Cell Tissue Res 347:521–544
David L, Feige JJ, Bailly S (2009) Emerging role of bone morphogenetic proteins in angiogenesis. Cytokine Growth Factor Rev 20:203–212
Wildemann B, Kadow-Romacker A, Haas NP, Schmidmaier G (2007) Quantification of various growth factors in different demineralized bone matrix preparations. J Biomed Mater Res 81:437–442
Garcia P, Histing T, Holstein JH, Klein M, Laschke MW, Matthys R et al (2013) Rodent animal models of delayed bone healing and non-union formation: a comprehensive review. Eur Cell Mater 26:1–12
Kokubu T, Hak DJ, Hazelwood SJ, Reddi AH (2003) Development of an atrophic nonunion model and comparison to a closed healing fracture in rat femur. J Orthop Res 21:503–510
Kloen P, Lauzier D, Hamdy RC (2012) Co-expression of BMPs and BMP-inhibitors in human fractures and non-unions. Bone 51:59–68
Fajardo M, Liu CJ, Egol K (2009) Levels of expression for BMP-7 and several BMP antagonists may play an integral role in a fracture nonunion: a pilot study. Clin Orthop Relat Res 467:3071–3078
Kwong FN, Hoyland JA, Freemont AJ, Evans CH (2009) Altered relative expression of BMPs and BMP inhibitors in cartilaginous areas of human fractures progressing towards nonunion. J Orthop Res 27:752–757
Kloen P, Doty SB, Gordon E, Rubel IF, Goumans MJ, Helfet DL (2002) Expression and activation of the BMP-signaling components in human fracture nonunions. J Bone Joint Surg Am 84-A:1909–1918
Niikura T, Hak DJ, Reddi AH (2006) Global gene profiling reveals a downregulation of BMP gene expression in experimental atrophic nonunions compared to standard healing fractures. J Orthop Res 24:1463–1471
Andrew J, Hoyland J, Freemont A, Marsh D (1995) Platelet-derived growth factor expression in normally healing human fractures. Bone 16:455–460
Beer HD, Longaker MT, Werner S (1997) Reduced expression of PDGF and PDGF receptors during impaired wound healing. J Invest Dermatol 109:132–138
Zeckey C, Hildebrand F, Glaubitz LM, Jurgens S, Ludwig T, Andruszkow H et al (2011) Are polymorphisms of molecules involved in bone healing correlated to aseptic femoral and tibial shaft non-unions? J Orthop Res 29:1724–1731
Wildemann B, Kadow-Romacker A, Pruss A, Haas NP, Schmidmaier G (2007) Quantification of growth factors in allogenic bone grafts extracted with three different methods. Cell Tissue Bank 8:107–114
Vukicevic S, Oppermann H, Verbanac D, Jankolija M, Popek I, Curak J et al (2014) The clinical use of bone morphogenetic proteins revisited: a novel biocompatible carrier device OSTEOGROW for bone healing. Int Orthop 38:635–647
Acknowledgments
The study was supported by a grant from Synthes, USA.
Conflict of interest
The authors have no conflict of interest related to the present study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schwabe, P., Simon, P., Kronbach, Z. et al. A pilot study investigating the histology and growth factor content of human non-union tissue. International Orthopaedics (SICOT) 38, 2623–2629 (2014). https://doi.org/10.1007/s00264-014-2496-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-014-2496-6