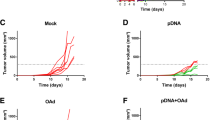
Abstract
Survivin is overexpressed in major types of cancer and is considered an ideal “universal” tumor-associated antigen that can be targeted by immunotherapeutic vaccines. However, its anti-apoptosis function raises certain safety concerns. Here, a new truncated human survivin, devoid of the anti-apoptosis function, was generated as a candidate tumor vaccine. Interleukin 2 (IL-2) has been widely used as an adjuvant for vaccination against various diseases. Meanwhile, the DNA prime and recombinant adenovirus (rAd) boost heterologous immunization strategy has been proven to be highly effective in enhancing immune responses. Therefore, the efficacy of a new cancer vaccine based on a truncated form of survivin, combined with IL-2, DNA prime, and rAd boost, was tested. As prophylaxis, immunization with the DNA vaccine alone resulted in a weak immune response and modest anti-tumor effect, whereas the tumor inhibition ratio with the DNA vaccine administered with IL-2 increased to 89 % and was further increased to nearly 100 % by rAd boosting. Moreover, complete tumor rejection was observed in 5 of 15 mice. Efficacy of the vaccine administered therapeutically was enhanced by nearly 300 % when combined with carboplatin. These results indicated that vaccination with a truncated survivin vaccine using DNA prime–rAd boost combined with IL-2 adjuvant and carboplatin represents an attractive strategy to overcoming immune tolerance to tumors and has potential therapeutic benefits in melanoma cancer.






Similar content being viewed by others
References
Jemal A, Bray F, Center MM et al (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Bolhassani A, Safaiyan S, Rafati S (2011) Improvement of different vaccine delivery systems for cancer therapy. Mol Cancer 10:3
Altieri DC (2008) Survivin, cancer networks and pathway-directed drug discovery. Nat Rev Cancer 8:61–70
Ishizaki H, Manuel ER, Song G-Y et al (2010) Modified vaccinia Ankara expressing survivin combined with gemcitabine generates specific antitumor effects in a murine pancreatic carcinoma model. Cancer Immunol Immunother 60:99–109
Kim HS, Kim CH, Park MY et al (2010) Efficient co-transduction of adenoviral vectors encoding carcinoembryonic antigen and survivin into dendritic cells by the CAR-TAT adaptor molecule enhance anti-tumor immunity in a murine colorectal cancer model. Immunol Lett 131:73–80
Lladser A, Ljungberg K, Tufvesson H et al (2009) Intradermal DNA electroporation induces survivin-specific CTLs, suppresses angiogenesis and confers protection against mouse melanoma. Cancer Immunol Immunother 59:81–92
Zwirner NW, Domaica CI (2010) Cytokine regulation of natural killer cell effector functions. BioFactors 36:274–288
Decker WK, Qiu J, Farhangfar F et al (2006) A retrogen plasmid-based vaccine generates high titer antibody responses against the autologous cancer antigen survivin and demonstrates anti-tumor efficacy. Cancer Lett 237:45–55
Lladser A, Parraga M, Quevedo L et al (2006) Naked DNA immunization as an approach to target the generic tumor antigen survivin induces humoral and cellular immune responses in mice. Immunobiology 211:11–27
Zhu K, Qin H, Cha SC et al (2007) Survivin DNA vaccine generated specific antitumor effects in pancreatic carcinoma and lymphoma mouse models. Vaccine 25:7955–7961
Barouch DH, Nabel GJ (2005) Adenovirus vector-based vaccines for human immunodeficiency virus type 1. Hum Gene Ther 16:149–156
Schulte R, Suh YS, Sauermann U et al (2009) Mucosal prior to systemic application of recombinant adenovirus boosting is more immunogenic than systemic application twice but confers similar protection against SIV-challenge in DNA vaccine-primed macaques. Virology 383:300–309
Andersen MH, Sorensen RB, Schrama D et al (2008) Cancer treatment: the combination of vaccination with other therapies. Cancer Immunol Immunother 57:1735–1743
Yu B, Zhang Y, Zhan Y et al (2011) Co-expression of herpes simplex virus thymidine kinase and Escherichia coli nitroreductase by an hTERT-driven adenovirus vector in breast cancer cells results in additive anti-tumor effects. Oncol Rep 26:255–264
Herrmann M, Lorenz HM, Voll R et al (1994) A rapid and simple method for the isolation of apoptotic DNA fragments. Nucleic Acids Res 22:5506–5507
Nagaraj S, Pisarev V, Kinarsky L et al (2007) Dendritic cell-based full-length survivin vaccine in treatment of experimental tumors. J Immunother 30:169–179
Zhang S, Zhang H, Shi H et al (2008) Induction of immune response and anti-tumor activities in mice with a DNA vaccine encoding human mucin 1 variable-number tandem repeats. Hum Immunol 69:250–258
Li F, Ackermann EJ, Bennett CF et al (1999) Pleiotropic cell-division defects and apoptosis induced by interference with survivin function. Nat Cell Biol 1:461–466
Grossman D, McNiff JM, Li F, Altieri DC (1999) Expression and targeting of the apoptosis inhibitor, survivin, in human melanoma. J Invest Dermatol 113:1076–1081
Grossman D, McNiff JM, Li F, Altieri DC (1999) Expression of the apoptosis inhibitor, survivin, in nonmelanoma skin cancer and gene targeting in a keratinocyte cell line. Lab Invest 79:1121–1126
O’Connor DS, Grossman D, Plescia J et al (2000) Regulation of apoptosis at cell division by p34cdc2 phosphorylation of survivin. Proc Natl Acad Sci USA 97:13103–13107
O’Connor DS (2000) Regulation of apoptosis at cell division by p34cdc2 phosphorylation of survivin. Proc Nat Acad Sci 97:13103–13107
Grossman D, Kim PJ, Schechner JS, Altieri DC (2001) Inhibition of melanoma tumor growth in vivo by survivin targeting. Proc Natl Acad Sci USA 98:635–640
Yagihashi A, Ohmura T, Asanuma K et al (2005) Detectio n of autoantibodies to survivin and livin in sera from patients with breast cancer. Clin Chim Acta 362:125–130
Kameshima H, Tsuruma T, Torigoe T et al (2011) Immunogenic enhancement and clinical effect by type-I interferon of anti-apoptotic protein, survivin-derived peptide vaccine, in advanced colorectal cancer patients. Cancer Sci 102:1181–1187
Miyazaki A, Kobayashi J, Torigoe T et al (2011) Phase I clinical trial of survivin-derived peptide vaccine therapy for patients with advanced or recurrent oral cancer. Cancer Sci 102:324–329
Honma I, Kitamura H, Torigoe T et al (2009) Phase I clinical study of anti-apoptosis protein survivin-derived peptide vaccination for patients with advanced or recurrent urothelial cancer. Cancer Immunol Immunother 58:1801–1807
Tsuruma T, Torigoe T, Hata F et al (2004) Anti-apoptosis protein, survivin-2B-derived peptide vaccine therapy. Gan To Kagaku Ryoho 31:1634–1636
Tsuruma T, Hata F, Torigoe T et al (2004) Phase I clinical study of anti-apoptosis protein, survivin-derived peptide vaccine therapy for patients with advanced or recurrent colorectal cancer. J Transl Med 2:19
Trepiakas R, Berntsen A, Hadrup SR et al (2010) Vaccination with autologous dendritic cells pulsed with multiple tumor antigens for treatment of patients with malignant melanoma: results from a phase I/II trial. Cytotherapy 12:721–734
Hirschowitz EA, Foody T, Hidalgo GE, Yannelli JR (2007) Immunization of NSCLC patients with antigen-pulsed immature autologous dendritic cells. Lung Cancer 57:365–372
Pisarev V, Yu B, Salup R et al (2003) Full-length dominant-negative survivin for cancer immunotherapy. Clin Cancer Res 9:6523–6533
Fuessel S, Meye A, Schmitz M et al (2006) Vaccination of hormone-refractory prostate cancer patients with peptide cocktail-loaded dendritic cells: results of a phase I clinical trial. Prostate 66:811–821
Xiang R, Mizutani N, Luo Y et al (2005) A DNA vaccine targeting survivin combines apoptosis with suppression of angiogenesis in lung tumor eradication. Cancer Res 65:553–561
Verdecia MA, Huang H, Dutil E et al (2000) Structure of the human anti-apoptotic protein survivin reveals a dimeric arrangement. Nat Struct Biol 7:602–608
Chantalat L, Skoufias DA, Kleman JP et al (2000) Crystal structure of human survivin reveals a bow tie-shaped dimer with two unusual alpha-helical extensions. Mol Cell 6:183–189
Shi Y (2000) Survivin structure: crystal unclear. Nat Struct Biol 7:620–623
Bachinsky MM, Guillen DE, Patel SR et al (2005) Mapping and binding analysis of peptides derived from the tumor-associated antigen survivin for eight HLA alleles. Cancer Immunol 5:6
Andersen MH, Pedersen LO, Becker JC, Straten PT (2001) Identification of a cytotoxic T lymphocyte response to the apoptosis inhibitor protein survivin in cancer patients. Cancer Res 61:869–872
Andersen MH, Pedersen LO, Capeller B et al (2001) Spontaneous cytotoxic T-cell responses against survivin-derived MHC class I-restricted T-cell epitopes in situ as well as ex vivo in cancer patients. Cancer Res 61:5964–5968
Pitisuttithum P, Rerks-Ngarm S, Bussaratid V et al (2011) Safety and reactogenicity of canarypox ALVAC-HIV (vCP1521) and HIV-1 gp120 AIDSVAX B/E vaccination in an efficacy trial in Thailand. PLoS ONE 6:e27837
Gabrilovich DI (2007) Combination of chemotherapy and immunotherapy for cancer: a paradigm revisited. Lancet Oncol 8:2–3
Acknowledgments
The authors thank the staff of the National Engineering Laboratory of AIDS Vaccine for reagent ordering and instrument management. The present work was supported by the Central University Basic Research Foundation of China (No. 200903255, 201103179).
Conflict of interest
The authors declare that there are no conflicts of interest in regard to this work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, H., Wang, Y., Liu, C. et al. DNA and adenovirus tumor vaccine expressing truncated survivin generates specific immune responses and anti-tumor effects in a murine melanoma model. Cancer Immunol Immunother 61, 1857–1867 (2012). https://doi.org/10.1007/s00262-012-1296-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-012-1296-3