Abstract
Purpose
The aim of this study was to develop a quantitative measurement of perfusion reduction using color-coded digital subtraction angiography (ccDSA) to monitor intra-procedural arterial stasis during TACE.
Materials and methods
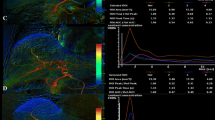
A total number of 35 patients with hepatocellular carcinoma who had undergone TACE were enrolled into the study. Pre- and post-two-dimensional digital subtraction angiography scans were conducted with same protocol and post-processed with ccDSA prototype software. Time-contrast-intensity (CI[t]) curve was obtained by region-of-interest (ROI) measurement on the generated ccDSA image. Quantitative 2D perfusion parameters time to peak, area under the curve (AUC), maximum upslope, and contrast intensity peak (CI-Peak) derived from the ROI-based CI[t] curve for pre- and post-TACE were evaluated to assess the reduction of antegrade blood flow and tumor blush. Relationships between 2D perfusion parameters, subjective angiographic chemoembolization endpoint (SACE) scale, and clinical outcomes were analyzed.
Results
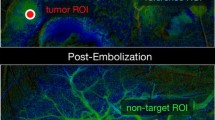
Area normalized AUC and CI-Peak revealed significant reduction after the TACE (P < 0.0001). AUCnorm decreased from pre-procedure of 0.867 ± 0.242 to 0.421 ± 0.171 (P < 0.001) after completion of TACE. CI-Peaknorm was 0.739 ± 0.221 before TACE and 0.421 ± 0.174 (P < 0.001) after TACE. Tumor blood supply time slowed down obviously after embolization. A perfusion reduction either from AUCnorm or CI-Peaknorm ranging from 30% to 40% was associated with SACE level III and a reduction ranging from 60% to 70% was equivalent to SACE level IV. For intermediate reduction (SACE level III), better tumor response was found after TACE rather than a higher reduction (SACE level IV).
Conclusion
ccDSA application provides an objective approach to quantify the perfusion reduction and subjectively evaluate the arterial stasis of antegrade blood flow and tumor blush caused by TACE.




Similar content being viewed by others
References
Bosch FX, Ribes J, Diaz M, et al. (2004) Primary liver cancer: worldwide incidence and trends. Gastroenterology 127:S5–S16
Bruix J, Sala M, Llovet JM (2004) Chemoembolization for hepatocellular carcinoma. Gastroenterology 127:S179–S188
Llovet JM, Real MI, Montana X, et al. (2002) Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet 359:1734–1739
Brown DB, Pilgram TK, Darcy MD, et al. (2005) Hepatic arterial chemoembolization for hepatocellular carcinoma: comparison of survival rates with different embolic agents. J Vasc Interv Radiol 16:1661–1666
Coldwell DM, Stokes KR, Yakes WF (1994) Embolotherapy: agents, clinical applications, and techniques. Radiographics 14(623–643):645–646
Ikeda M, Maeda S, Shibata J, et al. (2004) Transcatheter arterial chemotherapy with and without embolization in patients with hepatocellular carcinoma. Oncology 66:24–31
Maeda S, Fujiyama S, Tanaka M, et al. (2002) Survival and local recurrence rates of hepatocellular carcinoma patients treated by transarterial chemolipiodolization with and without embolization. Hepatol Res 23:202–210
Geschwind JF, Ramsey DE, Cleffken B, et al. (2003) Transcatheter arterial chemoembolization of liver tumors: effects of embolization protocol on injectable volume of chemotherapy and subsequent arterial patency. Cardiovasc Intervent Radiol 26:111–117
Bruix J, Sherman M, Llovet JM, et al. (2001) Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol 35:421–430
Lewandowski RJ, Wang D, Gehl J, et al. (2007) A comparison of chemoembolization endpoints using angiographic versus transcatheter intraarterial perfusion/MR imaging monitoring. J Vasc Interv Radiol 18:1249–1257
Wang D, Bangash AK, Rhee TK, et al. (2007) Liver tumors: monitoring embolization in rabbits with VX2 tumors–transcatheter intraarterial first-pass perfusion MR imaging. Radiology 245:130–139
Struffert T, Doerfler A (2009) Flat-detector computed tomography in diagnostic and interventional neuroradiology. Radiologe 49:820–829
Gupta R, Cheung AC, Bartling SH, et al. (2008) Flat-panel volume CT: fundamental principles, technology, and applications. Radiographics 28:2009–2022
Golitz P, Struffert T, Lucking H, et al. (2013) Parametric color coding of digital subtraction angiography in the evaluation of carotid cavernous fistulas. Clin Neuroradiol 23:113–120
Strother CM, Bender F, Deuerling-Zheng Y, et al. (2010) Parametric color coding of digital subtraction angiography. AJNR Am J Neuroradiol 31:919–924
Struffert T, Ott S, Kowarschik M, et al. (2013) Measurement of quantifiable parameters by time-density curves in the elastase-induced aneurysm model: first results in the comparison of a flow diverter and a conventional aneurysm stent. Eur Radiol 23:521–527
Zhang XB, Zhuang ZG, Ye H, et al. (2013) Objective assessment of transcatheter arterial chemoembolization angiographic endpoints: preliminary study of quantitative digital subtraction angiography. J Vasc Interv Radiol 24:667–671
Brown DB, Cardella JF, Sacks D, et al. (2006) Quality improvement guidelines for transhepatic arterial chemoembolization, embolization, and chemotherapeutic infusion for hepatic malignancy. J Vasc Interv Radiol 17:225–232
Materne R, Van Beers BE, Smith AM, et al. (2000) Non-invasive quantification of liver perfusion with dynamic computed tomography and a dual-input one-compartmental model. Clin Sci (Lond) 99:517–525
Fenchel M, Helber U, Simonetti OP, et al. (2004) Multislice first-pass myocardial perfusion imaging: Comparison of saturation recovery (SR)-TrueFISP-two-dimensional (2D) and SR-TurboFLASH-2D pulse sequences. J Magn Reson Imaging 19:555–563
Ahmed AS, Deuerling-Zheng Y, Strother CM, et al. (2009) Impact of intra-arterial injection parameters on arterial, capillary, and venous time-concentration curves in a canine model. AJNR Am J Neuroradiol 30:1337–1341
Jin B, Wang D, Lewandowski RJ, et al. (2011) Chemoembolization endpoints: effect on survival among patients with hepatocellular carcinoma. AJR Am J Roentgenol 196:919–928
Simonetti RG, Camma C, Fiorello F, et al. (1991) Hepatocellular carcinoma. A worldwide problem and the major risk factors. Dig Dis Sci 36:962–972
Acknowledgment
The authors acknowledge that the present research was supported by National Natural Science Foundation of China (Grant No. 81201172). Prototype software DSA-Analyzer (Siemens Healthcare, Forchheim, Germany) is not commercially available.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, J., Cheng, JJ., Huang, KY. et al. Quantitative assessment of angiographic perfusion reduction using color-coded digital subtraction angiography during transarterial chemoembolization. Abdom Radiol 41, 545–552 (2016). https://doi.org/10.1007/s00261-015-0622-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-015-0622-6