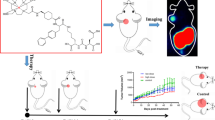
Abstract
Purpose
Prostate-specific membrane antigen (PSMA) is a promising target for prostate cancer imaging and therapy. The most commonly used scaffold incorporates a glutamate-urea (Glu-Urea) function. We recently developed oxalyldiaminopropionic acid-urea (ODAP-Urea) PSMA ligands in an attempt to improve upon the pharmacokinetic properties of existing agents. Here, we report the synthesis of an optimized 68Ga-labeled ODAP-Urea-based ligand, [68Ga]Ga-P137, and first-in-human results.
Methods
Twelve ODAP-Urea-based ligands were synthesized and radiolabeled with 68Ga in high radiochemical yield and purity. Their PSMA inhibitory capacities were determined using the NAALADase assay. Radioligands were evaluated in mice-bearing 22Rv1 prostate tumors by microPET. Lead compound [68Ga]Ga-P137 was evaluated for stability, cell uptake, and biodistribution. PET imaging of [68Ga]Ga-P137 was performed in three patients head-to-head compared to [68Ga]Ga-PSMA-617.
Results
Ligands were synthesized in 11.1-44.4% yield and > 95% purity. They have high affinity to PSMA (Ki of 0.13 to 5.47 nM). [68Ga]Ga-P137 was stable and hydrophilic. [68Ga]Ga-P137 showed higher uptake than [68Ga]Ga-PSMA-617 in tumor-bearing mice at 6.43 ± 0.98%IA/g vs 3.41 ± 1.31%IA/g at 60-min post-injection. In human studies, the normal organ biodistribution of [68Ga]Ga-P137 was grossly equivalent to that of [68Ga]Ga-PSMA-617 except for within the urinary tract, in which [68Ga]Ga-P137 demonstrated lower uptake.
Conclusion
The optimized ODAP-Urea-based ligand [68Ga]Ga-P137 can image PSMA in xenograft models and humans, with lower bladder accumulation to the Glu-Urea-based agent, [68Ga]Ga-PSMA-617, in a preliminary, first-in-human study.
Trial registration
ClinicalTrials.gov Identifier: NCT04560725, Registered 23 September 2020. https://clinicaltrials.gov/ct2/show/NCT04560725






Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Ross JS, Sheehan CE, Fisher HA, et al. Correlation of primary tumor prostate-specific membrane antigen expression with disease recurrence in prostate cancer. Clin Cancer Res. 2003;9:6357–62.
Rowe SP, Gorin MA, Pomper MG. Imaging of prostate-specific membrane antigen with small-molecule PET radiotracers: from the bench to advanced clinical applications. Annu Rev Med. 2019;70:461–77.
Sheikhbahaei S, Afshar-Oromieh A, Eiber M, et al. Pearls and pitfalls in clinical interpretation of prostate-specific membrane antigen (PSMA)-targeted PET imaging. Eur J Nucl Med Mol Imaging. 2017;44:2117–36.
Von Eyben FE, Picchio M, Von Eyben R, Rhee H, Bauman G. 68Ga-labeled prostate-specific membrane antigen ligand positron emission tomography/computed tomography for prostate cancer: a systematic review and meta-analysis. Eur Urol Focus. 2016;4:686–93.
Wester HJ, Schottelius M. PSMA-targeted radiopharmaceuticals for imaging and therapy. Semin Nucl Med. 2019;49:302–12.
Mease RC, Dusich CL, Foss CA, et al. N-[N-[(S)-1,3-Dicarboxypropyl]carbamoyl]-4-[18F]fluorobenzyl-L-cysteine, [18F]DCFBC: a new imaging probe for prostate cancer. Clin Cancer Res. 2008;14:3036–43.
Chen Y, Pullambhatla M, Foss CA, et al. 2-(3-{1-Carboxy-5-[(6-[18F]fluoro-pyridine-3-carbonyl)-amino]-pentyl}-ureido)-pentanedioic acid, [18F]DCFPyL, a PSMA-based PET imaging agent for prostate cancer. Clin Cancer Res. 2011;17:7645–53.
Cardinale J, Schäfer M, Beneˇsová M, et al. Preclinical evaluation of 18F-PSMA-1007, a new prostate-specific membrane antigen ligand for prostate cancer imaging. J Nucl Med. 2017;58:425–31.
Eder M, Schafer M, Bauder-Wust U, et al. 68Ga-complex lipophilicity and the targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem. 2012;23:688–97.
Benešová M, Schäfer M, Bauder-Wüst U, et al. Preclinical evaluation of a tailor-made DOTA-conjugated PSMA inhibitor with optimized linker moiety for imaging and endoradiotherapy of prostate cancer. J Nucl Med. 2015;56:914–20.
Kabasakal L, AbuQbeitah M, Aygun A, et al. Pre-therapeutic dosimetry of normal organs and tissues of 177Lu-PSMA-617 prostate-specific membrane antigen (PSMA) inhibitor in patients with castration-resistant prostate cancer. Eur J Nucl Med Mol Imaging. 2015;42:1976–83.
Martina W, Margret S, Jakub S, et al. 68Ga- and 177Lu-labeled PSMA I&T: optimization of a PSMA-targeted theranostic concept and first proof-of-concept human studies. J Nucl Med. 2015;56:1169–76.
Graham K, Lesche R, Gromov AV, et al. Radiofluorinated derivatives of 2-(phosphonomethyl)pentanedioic acid as inhibitors of prostate specific membrane antigen (PSMA) for the imaging of prostate cancer. J Med Chem. 2012;55:9510–20.
Yang X, Mease RC, Pullambhatla M, et al. [18F]Fluorobenzoyllysinepentanedioic acid carbamates: new scaffolds for positron emission tomography (PET) imaging of prostate-specific membrane antigen (PSMA). J Med Chem. 2016;59:206–18.
Duan X, Liu F, Kwon H, et al. (S)-3-(Carboxyformamido)-2-(3-(carboxymethyl)ureido)propanoic acid as a novel PSMA targeting scaffold for prostate cancer imaging. J Med Chem. 2020;63:3563–76.
Thisgaard H, Kumlin J, Langkjaer N, et al. Multi-curie production of gallium-68 on a biomedical cyclotron and automated radiolabelling of PSMA-11 and DOTATATE. EJNMMI Radiopharm Chem. 2021;6:1–11.
Benešová M, Bauderwüst U, Schäfer M, et al. Linker modification strategies to control the prostate-specific membrane antigen (PSMA)-targeting and pharmacokinetic properties of DOTA-conjugated PSMA inhibitors. J Med Chem. 2016;59:1761–75.
Olszewski RT, Bukhari N, Zhou J, et al. NAAG peptidase inhibition reduces locomotor activity and some stereotypes in the PCP model of schizophrenia via group II mGluR. J Neurochem. 2004;89:876–85.
Liu T, Liu C, Xu X, et al. Preclinical evaluation and pilot clinical study of Al18F-PSMA-BCH for prostate cancer imaging. J Nucl Med. 2019;60:1284–92.
Acknowledgements
We gratefully appreciate all the chemists, nurses, and technicians from the Department of Nuclear Medicine, Peking University Cancer Hospital for their contributions to imaging studies.
Funding
This work was supported by the National Natural Science Foundation of China (21877004, 92059101), the Clinical Medicine Plus X—Young Scholars Project of Peking University (PKU2020LCXQ029), the Wu Jieping Medical Foundation (320.6750.2020–6-25), the Beijing Science and Technology Project (Z181100001618017), and the Science Foundation of Peking University First Hospital (2019SF08), the Found For Fostering Young Scholars of Peking University Health Science Center (BMU2021PYB024) .
Author information
Authors and Affiliations
Contributions
Xing Yang, Yan Fan, and Zhi Yang conceived and designed the experiments. Xiaojiang Duan and Futao Liu performed the chemical synthesis of the PSMA-targeting ligands. Xiaojiang Duan, Hua Zhu, and Zhen Cao performed radiosynthesis, binding affinity, cellular experiments, and animal model preparation. Xiaojiang Duan, Xiaojun Zhang, Jinming Zhang, and Zhen Cao performed preclinical imaging experiments. Chen Liu, Hua Zhu, Ya'nan Ren, Xiaoyi Guo, and Xiaojiang Duan performed the clinical translation imaging preparation and experiments. Xing Yang, Xiaojiang Duan, and Xuekang Cai co-wrote the paper. Zhen Xi and Martin G. Pomper helped the data analysis with constructive discussions. All authors discussed the results and commented on manuscript.
Corresponding authors
Ethics declarations
Ethics approval
All procedures involving human participants were carried out in accordance with the Ethics Committee of Peking University Cancer Hospital (2020KT107) and registered in NIH (NCT04560725). All animal studies were performed according to a protocol approved by the Peking University First Hospital Animal Care and Use Committee.
Informed consent
Written informed consents were obtained from all participants included in the study.
Consent to participate
Written informed consents were obtained from all participants included in the study.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiaojiang Duan, Zhen Cao and Hua Zhu contribute equally to this work.
This article is part of the Topical Collection on Radiopharmacy
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Duan, X., Cao, Z., Zhu, H. et al. 68Ga-labeled ODAP-Urea-based PSMA agents in prostate cancer: first-in-human imaging of an optimized agent. Eur J Nucl Med Mol Imaging 49, 1030–1040 (2022). https://doi.org/10.1007/s00259-021-05486-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-021-05486-x