Abstract
Purpose
To determine the utility of 18F-FDG (FDG) PET/CT performed in an early and delayed phase during neoadjuvant chemotherapy in the prediction of lymph node histopathological response in patients with locally advanced breast cancer.
Methods
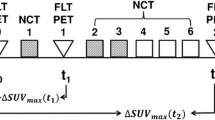
FDG PET/CT studies performed in 76 patients (mean age 53 years) at baseline (PET-1), after the second course of chemotherapy (PET-2) and after the last course of chemotherapy (PET-3) were prospectively analysed. Inclusion criteria were lymph node involvement detected by PET/CT and non-sentinel node biopsy before or after the baseline PET/CT scan. Following the recommendations of the 12th International Breast Conference (St. Gallen), the patients were divided into five subgroups in relation to biological prognostic factors by immunohistochemistry. For diagnosis visual and semiquantitative analyses was performed. Absence of detectable lymph node uptake on the PET-2 or PET-3 scan with respect to the PET-1 scan was considered metabolic complete response (mCR). Lymph nodes were histopathologically classified according the lymph node regression grade and in response groups as pathological complete response (pCR) or not pCR (type A/D or B/C of the Smith grading system, respectively). ROC analysis was performed to determine a cut-off value of Δ% SUV1–2 and SUV1–3 for prediction of nodal status after chemotherapy. An association between mCR and pCR was found (Cohen’s kappa analysis), and associations between phenotypes and metabolic behaviour and the final histopathological status were also found.
Results
Lymph node pCR was seen in 34 patients. The sensitivity, specificity, and positive and negative predictive values of PET-2 and PET-3 in establishing the final status of the axilla after chemotherapy were 52 %, 45 %, 50 % and 47 %, and 33 %, 84 %, 67 % and 56 %, respectively. No significant relationship was observed between mCR on PET-2 and PET-3 and pCR (p = 0.31 and 0.99, respectively). Lymph node metabolism on PET-1 was not able to predict the final histopathological status, whereas basal carcinomas showed a higher rate of pCR (70.6 %) than the other groups (p = 0.03).
Conclusion
FDG PET/CT seems to have limitations in both the early and delayed evaluation of lymph node status after chemotherapy, with reduced predictive values.

Similar content being viewed by others
References
Mamounas EP. Overview of national surgical adjuvant breast project neoadjuvant chemotherapy studies. Semin Oncol. 1998;25:31–5.
Rouzier R, Extra JM, Klijanienko J, Falcou MC, Asselain B, Vincent-Salomon A, et al. Incidence and prognostic significance of complete axillary downstaging after primary chemotherapy in breast cancer patients with T1 to T3 tumors and cytologically proven axillary metastatic lymph nodes. J Clin Oncol. 2002;20:1304–10.
Dalus K, Rendl G, Rettenbacher L, Pirich C. FDG PET/CT for monitoring response to neoadjuvant chemotherapy in breast cancer patients. Eur J Nucl Med Mol Imaging. 2010;37:1992–3.
Prati R, Minami CA, Gornbein JA, Debruhl N, Chung D, Chang HR. Accuracy of clinical evaluation of locally advanced breast cancer in patients receiving neoadjuvant chemotherapy. Cancer. 2009;115:1194–202.
Rousseau C, Devillers A, Sagan C, Ferrer L, Bridji B, Campion L, et al. Monitoring of early response to neoadjuvant chemotherapy in stage II and III breast cancer by [18F]fluorodeoxyglucose positron emission tomography. J Clin Oncol. 2006;24:5366–72.
Bassa P, Kim EE, Inoue T, Wong FC, Korkmaz M, Yang DJ, et al. Evaluation of preoperative chemotherapy using PET with fluorine-18-fluorodeoxyglucose in breast cancer. J Nucl Med. 1996;37:931–8.
Mankoff DA, Dunnwald LK, Gralow JR, Ellis GK, Schubert EK, Tseng J, et al. Changes in blood flow and metabolism in locally advanced breast cancer treated with neoadjuvant chemotherapy. J Nucl Med. 2003;44:1806–14.
Smith IC, Welch AE, Hutcheon AW, Miller ID, Payne S, Chilcott F, et al. Positron emission tomography using [(18)F]-fluorodeoxy- D-glucose to predict the pathologic response of breast cancer to primary chemotherapy. J Clin Oncol. 2000;18:1676–88.
Straver ME, Aukema TS, Valdes Olmos RA, Rutgers EJ, Gilhuijs KG, Schot ME, et al. Feasibility of FDGPET/CT to monitor the response of axillary lymph node metastases to neoadjuvant chemotherapy in breast cancer patients. Eur J Nucl Med Mol Imaging. 2010;37:1069–76.
García JR, Rodríguez A, Cabrera A. Tomografía por emisión de positrones de cuerpo completo (PET/TAC) con 18F-fluorodesoxiglucosa. Rev Esp Med Nucl. 2009;28:85–9.
American Joint Committee on Cancer (AJCC) 7th edition. http://www.cancerstaging.org/staging/index.html.
Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thürlimann B, Senn HJ, et al. Strategies for subtypes – dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol. 2011;22:1736–47.
Smith IC, Heys SD, Hutcheon AW, Miller ID, Payne S, Gilbert FJ, et al. Neoadjuvant chemotherapy in breast cancer: significantly enhanced response with docetaxel. J Clin Oncol. 2002;20:1456–66.
Ogston KN, Miller ID, Payne S, Hutcheon AW, Sarkar TK, Smith I, et al. A new histological grading system to assess response of breast cancers to primary chemotherapy: prognostic significance and survival. Breast. 2003;12:320–7.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics 2007. CA Cancer J Clin. 2007;57:43–66.
Bear HD, Anderson S, Smith RE, Geyer Jr CE, Mamounas EP, Fisher B, et al. Sequential preoperative or postoperative docetaxel added to preoperative doxorubicin plus cyclophosphamide for operable breast cancer: National Surgical Adjuvant Breast and Bowel Project Protocol B-27. J Clin Oncol. 2006;24:2019–27.
Martoni AA, Zamagni C, Quercia S, Rosati M, Cacciari N, Bernardi A, et al. Early 18F-2-Fluoro-2-deoxy-D-glucose positron emission tomography may identify a subset of patients with estrogen receptor-positive breast cancer who will not respond optimally to preoperative chemotherapy. Cancer. 2010;116:805–13.
Berriolo-Riedinger A, Touzery C, Riedinger JM, Toubeau M, Coudert B, Arnould L, et al. [18F]FDG-PET predicts complete pathological response of b breast cancer to neoadjuvant chemotherapy. Eur J Nucl Med Mol Imaging. 2007;34:1915–24.
Duch J, Fuster D, Muñoz M, Fernández PL, Paredes P, Fontanillas M, et al. (18)F-FDG PET/CT for early prediction of response to neoadjuvant chemotherapy in breast cancer. Eur J Nucl Med Mol Imaging. 2009;36:1551–7.
Groheux D, Glacchetti S, Espie M, Rubello D, Moretti JL, Hindié E. Early monitoring of response to neoadjuvant chemotherapy in breast cancer with 18F-FDG PET/CT: defining a clinical aim. Eur J Nucl Med Mol Imaging. 2011;38:419–25.
Rousseau C, Devillers A, Campone M, Campion L, Ferrer L, Sagan C, et al. FDG PET evaluation of early axillary lymph node response to neoadjuvant chemotherapy in stage II and III breast cancer patients. Eur J Nucl Med Mol Imaging. 2011;38:1029–36.
Gilardi L, De Cicco C, Colleoni M, Cardillo A, Montagna E, Dellapasqua S, et al. Investigation of 18F-FDG PET in the selection of patients with breast cancer as candidates for sentinel node biopsy after neoadjuvant therapy. Eur J Nucl Med Mol Imaging. 2010;37:1834–41.
McDermott GM, Welch A, Staff RT, Gilbert FJ, Schweiger L, Semple SI, et al. Monitoring primary breast cancer throughout chemotherapy using FDG-PET. Breast Cancer Res Treat. 2007;102:75–84.
Krak NC, Hoekstra OS, Lammertsma AA. Measuring response to chemotherapy in locally advanced breast cancer: methodological considerations. Eur J Nucl Med Mol Imaging. 2004;31:S103–11.
Basu S, Kwee TC, Torigian D, Saboury B, Alavi A. Suboptimal and inadequate quantification: an alarming crisis in medical application of PET. Eur J Nucl Med Mol Imaging. 2011;38:1381–2.
Dose Schwarz J, Bader M, Jenicke L, Hemminger G, Jänicke F, Avril N. Early prediction of response to chemotherapy in metastatic breast cancer using sequential 18F-FDG PET. J Nucl Med. 2005;46:1144–50.
García Vicente A, Soriano Castrejón A, Relea Calatayud F, Muñoz Madero V, Molina Garrido MJ, León Martín AA, et al. 18F-FDG semi-quantitative parameters and biological prognostic factors in locally advanced breast cancer. Rev Esp Med Nucl. 2012;31:308–14.
García Vicente AM, Soriano Castrejón A, Cruz Mora MA, González Ageitos A, Muñoz Sánchez M del M, León Martín A, et al. Semiquantitative lymph node assessment of 18F-FDG PET/CT in locally advanced breast cancer: correlation with biological prognostic factors. Eur J Nucl Med Mol Imaging. 2013;40:72–9.
Liedtke C, Mazouni C, Hess KR, André F, Tordai A, Mejia JA, et al. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26:1275–81.
Kong X, Moran MS, Zhang N, Haffty B, Yang Q. Meta-analysis confirms achieving pathological complete response after neoadjuvant chemotherapy predicts favourable prognosis for breast cancer patients. Eur J Cancer. 2011;47:2084–90.
Veronesi U, De Cicco C, Galimberti VE, Fernandez JR, Rotmensz N, Viale G, et al. A comparative study on the value of FDG-PET and sentinel node biopsy to identify occult axillary metastases. Ann Oncol. 2007;18:473–8.
García Vicente AM, Soriano Castrejón A, Cruz Mora MA, Ortega Ruiperez C, Espinosa Aunión R, León Martín A, et al. Dual time point 2-deoxy-2-[18F] fluoro-D-glucose PET/CT: nodal staging in locally advanced breast cancer. Rev Esp Med Nucl Imagen Mol. 2013. doi:10.1016/j.remn.2013.03.005
Hodgson NC, Gulenchyn KY. Is there a role for positron emission tomography in breast cancer staging? J Clin Oncol. 2008;26:712–20.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
García Vicente, A.M., Soriano Castrejón, Á., León Martín, A. et al. Early and delayed prediction of axillary lymph node neoadjuvant response by 18F-FDG PET/CT in patients with locally advanced breast cancer. Eur J Nucl Med Mol Imaging 41, 1309–1318 (2014). https://doi.org/10.1007/s00259-013-2657-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-013-2657-7