Abstract
Purpose
Stimulated thyroglobulin (Tg) and 131I total-body scan (TBS) have been the mainstays of differentiated thyroid carcinoma (DTC), but now diagnostic TBS has been eliminated from some follow-up protocols. Nevertheless, Tg can be negative in the presence of thyroid tissue, and moderately elevated Tg poses management problems. The purpose of this study was to check how many patients have Tg negative but visible thyroid tissue and if diagnostic TBS could be clinically useful.
Material and methods
Retrospective review of 317 exams (stimulated Tg and TBS) of 128 patients with DTC. Patients with high-grade criteria and/or positive autoantibodies are excluded. Tg is considered positive if higher than the sensitivity level of the technique. TBS is positive if thyroid remnant, lymphatic node or metastases are detected. Results are classified in Tg and TBS concordant or discordant.
Results
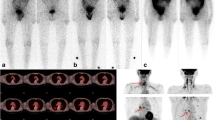
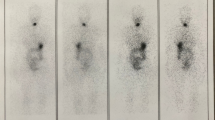
Discordant 131I TBS and Tg were found in 74/317 studies (23.3%), 48 (65%) being TBS positive and Tg negative. None of 128 patients had a first post-surgery negative scan, but 13 (10%) were Tg negative, even two patients with lymph node uptake. One year after radioiodine treatment, 26 out of 115 patients (initially Tg and TBS concordant positive) showed Tg negative and TBS still positive due to cervical remnants (20 patients), lymph node uptake (one) and metastasis (five); 20 patients remained Tg and TBS positive, but 14 of them only showed thyroid remnants.
Conclusions
We propose to perform periodic diagnostic TBS to improve Tg specificity, at least while neck thyroid activity is detected, even if Tg is negative.






Similar content being viewed by others
References
Mazzaferri EL, Kloos RT. Current approaches to primary therapy for papillary and follicular thyroid cancer. J Clin Endocrinol Metab 2001;86(4):1447–63.
National Comprehensive Cancer Network (NCCN). Thyroid carcinoma. Clinical practice guidelines in oncology, version 1. Fort Washington: NCCN; 2005.
Robbins RJ, Schlumberger MJ. The evolving role of 131I for the treatment of differentiated thyroid carcinoma. J Nucl Med 2005;46S:28S–37S.
Schlumberger M, Berg G, Cohen O, Duntas L, Jamar F, Jarzab B, et al. Follow up of low-risk patients with differentiated thyroid carcinoma: an European Perspective. Eur J Endocrinol 2004;150:105–12.
Mazzaferri EL, Robbins RJ, Spencer CA, Braverman LE, Pacini F, Wartofsky L, et al. A consensus report of the role of serum thyroglobulin as a monitoring method for low-risk patients with papillary thyroid carcinoma. J Clin Endocrinol Metab 2003;88:1433–41.
Pacini F, Molinaro E, Castagna MG, Agate L, Elisei R, Ceccarelli F, et al. Recombinant human thyrotropin-stimulated serum thyroglobulin combined with neck ultrasonography has the highest sensitivity in monitoring differentiated thyroid carcinoma. J Clin Endocrinol Metab 2003;88:3668–73.
Freitas JE. Changing concepts in the management of thyroid cancer. In: Freeman LM, editor. Nuclear Medicine Annual 2003. Philadelphia: Lippincott Williams and Wilkins; 2003. p. 101–29.
Ma C, Xie J, Kuang A. Is empiric 131I therapy justified for patients with positive thyroglobulin and negative 131I whole-body scanning results? J Nucl Med 2005;46:1164–70.
Ozata M, Suzuki S, Miyamoto T, Tsuan liu R, Fierro-Renoy F, Degroot L. Serum thyroglobulin in the follow up of patients with treated differentiated thyroid cancer. J Clin Endocrinol Metab 1994;79:98–105.
Bal C, Padhy R, Jana S, Pant GS, Basu AK. Prospective randomized clinical trial to evaluate the optimal dose of 131I for remnant ablation in patients with differentiated thyroid carcinoma. Cancer 1996;77:2574–80.
Samuel AM, Rajashekharrao B. Radioiodine therapy for well-differentiated thyroid cancer: a quantitative dosimetric evaluation for remnant thyroid ablation after surgery. J Nucl Med 1994;35:1944–50.
Pacini F, Ladenson PW, Schlumberger M, Driedger A, Luster M, Kloos RT, et al. Radioiodine ablation of thyroid remnants after preparation with recombinant thyrotropin in differentiated thyroid carcinoma: results of an international, randomized, controlled study. J Clin Endocrinol Metab 2006;91(3):926–32.
Meier DA, Brill DR, Becker DV, Clarke SEM, Silberstein EB, Royal HD, et al. Procedure guidelines for therapy of thyroid disease with 131Iodine. J Nucl Med 2002;43:856–61.
Abós Olivares MD, González P. Controversias en el seguimiento del carcinoma diferenciado de tiroides. Nuevas respuestas a viejas cuestiones. Rev Esp Med Nucl 2005;24(3):207–15.
Ma C, Kuang A, Xie J, Ma T. Possible explanations for patients with discordant findings of serum thyroglobulin and 131I whole-body scanning. J Nucl Med 2005;46(9):1473–80.
Sawka AM, Thephamongkhol K, Brouwers M, Thabane L, Browman G, Gerstein HC. A systematic review and metaanalysis of the effectiveness of radioactive iodine remnant ablation for well-differentiated thyroid cancer. J Clin Endocrinol Metab 2004;89:3668–76.
Brendel AJ, Lambert B, Guyot M, Jeandot R, Dubourg H, Roger P, et al. Low levels of serum thyroglobulin after withdrawal of thyroid suppression therapy in the follow up of differentiated thyroid carcinoma. Eur J Nucl Med 1990;16:35–8.
Schlumberger MJ. Papillary and follicular thyroid carcinoma. N Engl J Med 1998;338(5):297–306.
Bushnell DL, Boles MA, Kaufman GE, Wadas MA, Barnes WE. Complications, sequela and dosimetry of iodine-131 therapy for thyroid carcinoma. J Nucl Med 1992;33:2214–21.
Wartofsky L, Sherman SI, Gopal J, Schlumberger M, Hay ID. The use of radioactive iodine in patients with papillary and follicular thyroid cancer. J Clin Endocrinol and Metab 1998;83(12):4195–203.
Hay ID, Klee GG. Thyroid cancer diagnosis and management. Clin Lab Med 1993;13(3):725–34.
Hay ID, Thompson GB, Grant CS, Bergstralh EJ, Dvorak CE, Gorman CA, et al. Papillary thyroid carcinoma managed at the Mayo Clinic during six decades (1940–1999): temporal trends in initial therapy and long-term outcome in 2444 consecutive treated patients. World J Surg 2002;26:879–85.
Pace L, Klain M, Albanese C, Salvatore B, Storto G, Soricelli A, et al. Short term outcome of differentiated thyroid cancer patients receiving a second iodine-131 therapy on the basis of a detectable serum thyroglobulin level after initial treatment. Eur J Nucl Med Mol Imaging 2006;33:179–83.
Pacini F, Agate L, Elisei R, Capezzone M, Ceccarelli C, Lippi F, et al. Outcome of differentiated thyroid cancer with detectable serum Tg and negative diagnostic 131I whole body scan: comparison of patients treated with high 131I activities versus untreated patients. J Clin Endocrinol Metab 2001;86:4092–97.
Pacini F, Schlumberger M, Dralle H, Elisei R, Smit JWA, Wiersinga W, et al. European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur J Endocrinol 2006;154:787–803.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Caballero-Calabuig, E., Cano-Terol, C., Sopena-Monforte, R. et al. Influence of the thyroid remnant in the elevation of the serum thyroglobulin after thyroidectomy in differentiated thyroid carcinoma. Importance of the diagnostic iodine total-body scanning. Eur J Nucl Med Mol Imaging 35, 1449–1456 (2008). https://doi.org/10.1007/s00259-008-0789-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-008-0789-y