Abstract
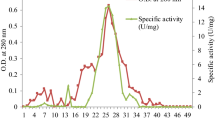
The thermostable esterase from the moderate thermophile Bacillus circulans was purified to homogeneity using a four-step procedure. Esterase activity was associated with a protein of molecular mass 95 kDa, composed of three identical subunits of 30 kDa. The esterase activity was thermostable with a maximum activity at 55 °C using initial rate assay. The half-inactivation temperature was 71 °C after a 1-h treatment, which compared favorably to that of other enzymes. Activity at temperatures of 30–37 °C was high (about half of maximum), making this new enzyme very attractive for applications in this moderate temperature range. The esterase also showed high activity at a rather alkaline pH (higher than 10). The specificity pattern showed a marked specificity for mid-chain-length fatty acids (3–8 carbon atoms), which classified the enzyme as a carboxylesterase.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 10 September 1999 / Received revision: 26 January 2000 / Accepted: 27 January 2000
Rights and permissions
About this article
Cite this article
Kademi, A., Aït-Abdelkader, N., Fakhreddine, L. et al. Purification and characterization of a thermostable esterase from the moderate thermophile Bacillus circulans . Appl Microbiol Biotechnol 54, 173–179 (2000). https://doi.org/10.1007/s002530000353
Issue Date:
DOI: https://doi.org/10.1007/s002530000353