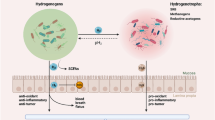
Abstract
Exposure to ionizing radiation (IR) tends to cause serious health concerns. Thus, radioprotective agents are vital for the population exposed to radiation. As microorganisms have the advantages of fast reproduction and no geographical restrictions, direct microbe-based and environmental induction compounds are thriving radioprotectants resources. Oxidative system and oxidase in Acetobacter pasteurianus are unique and intriguing, the radioprotective effect of the cell-free extract from A. pasteurianus (APE) and 60Coγ-treated extract (IRE) were comparatively investigated in the present study. The survival rate of A. pasteurianus with IRE addition was 149.1% in H2O2 damage test, while that with APE was only 10.4%. The viability of 60Coγ-treated AML-12 cells was increased by 18.8% with IRE addition, yet APE showed no significant radioprotective effect. Moreover, in 60Coγ-treated mice, IRE could significantly protect the white blood cell, improve the liver index, and attenuate the injuries of immune organs in mice. Administration of IRE significantly raised the activities of superoxide dismutase (SOD) and reduced the products of lipid peroxidation. These results clarified that gavage with APE and IRE presented notable antioxidant and radioprotective efficacy. A. pasteurianus showed appealing potential to be novel radioprotective bioagents and 60Coγ treatment on microbe could be a new method for the development of better radioprotectant.
Key points
• 60Coγ induction could improve the radioprotective effect of APE.
• IRE protected white blood cell in mice under IR.
• IRE products have broad application prospects in radioprotection based on microbes.
Graphical abstract







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Achuthan AA, Duary RK, Madathil A, Panwar H, Kumar H, Batish VK, Grover S (2012) Antioxidative potential of lactobacilli isolated from the gut of Indian people. Mol Biol Rep 39(8):7887–7897. https://doi.org/10.1007/s11033-012-1633-9
Azuma Y, Hosoyama A, Matsutani M, Furuya N, Horikawa H, Harada T, Hirakawa H, Kuhara S, Matsushita K, Fujita N, Shirai M (2009) Whole-genome analyses reveal genetic instability of Acetobacter pasteurianus. Nucleic Acids Res 37(17):5768–5783. https://doi.org/10.1093/nar/gkp612
Brand M, Sommer M, Ellmann S, Wuest W, May MS, Eller A, Vogt S, Lell MM, Kuefner MA, Uder M (2015) Influence of different antioxidants on X-ray induced DNA double-strand breaks (DSBs) using γ-H2AX immunofluorescence microscopy in a preliminary study. PLoS ONE 10(5):e0127142. https://doi.org/10.1371/journal.pone.0127142
Cravens A, Payne J, Smolke CD (2019) Synthetic biology strategies for microbial biosynthesis of plant natural products. Nat Commun 10(1):2142. https://doi.org/10.1038/s41467-019-09848-w
Clutton SM, Townsend KM, Walker C, Ansell JD, Wright EG (1996) Radiation-induced genomic instability and persisting oxidative stress in primary bone marrow cultures. Carcinogenesis 17(8):1633–9. https://doi.org/10.1093/carcin/17.8.1633
Demain AL (2014) Importance of microbial natural products and the need to revitalize their discovery. J Ind Microbiol Biotechnol 41(2):185–201. https://doi.org/10.1007/s10295-013-1325-z
Fukami H, Tachimoto H, Kishi M, Kaga T, Tanaka Y (2010) Acetic acid bacterial lipids improve cognitive function in dementia model rats. J Agric Food Chem 58(7):4084–4089. https://doi.org/10.1021/jf9045842
Gullo M, Verzelloni E, Canonico M (2014) Aerobic submerged fermentation by acetic acid bacteria for vinegar production: process and biotechnological aspects. Process Biochem 49(10):1571–1579
Guo H, Chou WC, Lai Y, Liang K, Tam JW, Brickey WJ, Chen L, Montgomery ND, Li X, Bohannon LM (2020) Multi-omics analyses of radiation survivors identify radioprotective microbes and metabolites. Science 370
Johnke RM, Sattler JA, Allison RR (2014) Radioprotective agents for radiation therapy: future trends. Future Oncol 10(15):2345–2357. https://doi.org/10.2217/fon.14.175
Kryston TB, Georgiev AB, Pissis P, Georgakilas AG (2011) Role of oxidative stress and DNA damage in human carcinogenesis. Mutat Res 711(1–2):193–201. https://doi.org/10.1016/j.mrfmmm.2010.12.016
Kuehne A, Hildebrand J, Soehle J, Wenck H, Terstegen L, Gallinat S, Knott A, Winnefeld M, Zamboni N (2017) An integrative metabolomics and transcriptomics study to identify metabolic alterations in aged skin of humans in vivo. BMC Genomics 18(1):169. https://doi.org/10.1186/s12864-017-3547-3
Lauschke VM, Hendriks DF, Bell CC, Andersson TB, Ingelman-Sundberg M (2016) Novel 3D culture systems for studies of human liver function and assessments of the hepatotoxicity of drugs and drug candidates. Chem Res Toxicol 29(12):1936–1955. https://doi.org/10.1021/acs.chemrestox.6b00150
Lim S, Jung JH, Blanchard L, de Groot A (2019) Conservation and diversity of radiation and oxidative stress resistance mechanisms in Deinococcus species. FEMS Microbiol Rev 43(1):19–52. https://doi.org/10.1093/femsre/fuy037
Maejima Y, Kuroda J, Matsushima S, Ago T, Sadoshima J (2011) Regulation of myocardial growth and death by NADPH oxidase. J Mol Cell Cardiol 50(3):408–416. https://doi.org/10.1016/j.yjmcc.2010.12.018
Matsutani M, Nishikura M, Saichana N, Hatano T, Masud-Tippayasak U, Theergool G, Yakushi T, Matsushita K (2013) Adaptive mutation of Acetobacter pasteurianus SKU1108 enhances acetic acid fermentation ability at high temperature. J Biotechnol 165(2):109–119. https://doi.org/10.1016/j.jbiotec.2013.03.006
Mishra KN, Moftah BA, Alsbeih GA (2018) Appraisal of mechanisms of radioprotection and therapeutic approaches of radiation countermeasures. Biomed Pharmacother 106:610–617. https://doi.org/10.1016/j.biopha.2018.06.150
Microbial Products (2015) Microbial products: technologies, applications and global markets (September 2015: BIO086C). BCC Research
Monzen S, Kashiwakura I (2012) Radioprotective effects of (-)-epigallocatechin-3-gallate on human erythrocyte/granulocyte lineages. Radiat Prot Dosimetry 152(1–3):224–228. https://doi.org/10.1093/rpd/ncs226
Mu G, Gao Y, Tuo Y, Li H, Zhang Y, Qian F, Jiang S (2018) Assessing and comparing antioxidant activities of lactobacilli strains by using different chemical and cellular antioxidant methods. J Dairy Sci 101(12):10792–10806. https://doi.org/10.3168/jds.2018-14989
Obe G, Durante M (2010) DNA double strand breaks and chromosomal aberrations. Cytogenet Genome Res 128(1–3):8–16. https://doi.org/10.1159/000303328
Prasad J, McJarrow P, Gopal P (2003) Heat and osmotic stress responses of probiotic Lactobacillus rhamnosus HN001 (DR20) in relation to viability after drying. Appl Environ Microbiol 69(2):917–925. https://doi.org/10.1128/aem.69.2.917-925.2003
Qiu K, Zhao Q, Wang J, Qi GH, Wu SG, Zhang HJ (2021) Effects of pyrroloquinoline quinone on lipid metabolism and antioxidative capacity in a high-fat-diet metabolic dysfunction-associated fatty liver disease chick model. Int J Mol Sci 22(3) https://doi.org/10.3390/ijms22031458
Reisz JA, Bansal N, Qian J, Zhao W, Furdui CM (2014) Effects of ionizing radiation on biological molecules–mechanisms of damage and emerging methods of detection. Antioxid Redox Signal 21(2):260–292. https://doi.org/10.1089/ars.2013.5489
Riehl TE, Alvarado D, Ee X, Zuckerman A, Foster L, Kapoor V, Thotala D, Ciorba MA, Stenson WF (2019) Lactobacillus rhamnosus GG protects the intestinal epithelium from radiation injury through release of lipoteichoic acid, macrophage activation and the migration of mesenchymal stem cells. Gut 68(6):1003–1013. https://doi.org/10.1136/gutjnl-2018-316226
Saichana N, Matsushita K, Adachi O, Frébort I, Frebortova J (2015) Acetic acid bacteria: a group of bacteria with versatile biotechnological applications. Biotechnol Adv 33(6 Pt 2):1260–1271. https://doi.org/10.1016/j.biotechadv.2014.12.001
Singh R, Kumar M, Mittal A, Mehta PK (2017) Microbial metabolites in nutrition, healthcare and agriculture. 3 Biotech 7(1):15. https://doi.org/10.1007/s13205-016-0586-4
Singh VK, Seed TM (2019) The efficacy and safety of amifostine for the acute radiation syndrome. Expert Opin Drug Saf 18(11):1077–1090. https://doi.org/10.1080/14740338.2019.1666104
Smith TA, Kirkpatrick DR, Smith S, Smith TK, Pearson T, Kailasam A, Herrmann KZ, Schubert J, Agrawal DK (2017) Radioprotective agents to prevent cellular damage due to ionizing radiation. J Transl Med 15(1):232. https://doi.org/10.1186/s12967-017-1338-x
Takeuchi T, Miyauchi E, Kanaya T, Kato T, Nakanishi Y, Watanabe T, Kitami T, Taida T, Sasaki T, Negishi H, Shimamoto S, Matsuyama A, Kimura I, Williams IR, Ohara O, Ohno H (2021) Acetate differentially regulates IgA reactivity to commensal bacteria. Nature 595(7868):560–564. https://doi.org/10.1038/s41586-021-03727-5
Winczura P, Jassem J (2010) Combined treatment with cytoprotective agents and radiotherapy. Cancer Treat Rev 36(3):268–275. https://doi.org/10.1016/j.ctrv.2009.12.001
Zhao L, Tao X, Qi Y, Xu L, Yin L, Peng J (2018) Protective effect of dioscin against doxorubicin-induced cardiotoxicity via adjusting microRNA-140-5p-mediated myocardial oxidative stress. Redox Biol 16:189–198. https://doi.org/10.1016/j.redox.2018.02.026
Zhou R, Si J, Zhang H, Wang Z, Li J, Zhou X, Gan L, Liu Y (2014) The effects of x-ray radiation on the eye development of zebrafish. Hum Exp Toxicol 33(10):1040–1050. https://doi.org/10.1177/0960327114522278
Funding
We are grateful for the Natural Science Foundation of Henan Province for Outstanding Youth (202300410365), the Program for Science & Technology Innovation Talents in Universities of Henan Province (22HASTIT037), the Key Scientific Research Projects of Higher Education Institutions in Henan Province (21A530006), the National Natural Science Foundation of China (81870093, 31900296), the Research Project of People’s Liberation Army (BX115C007, BFP20C006).
Author information
Authors and Affiliations
Contributions
Z.J.Q and L.J.K designed and supervised the research, analyzed the data, and revised the manuscript. G.Y.H performed the experiments, analyzed the data, and wrote the manuscript. Y.J.J, Z.C.C, K.Q.Z, H.L.M, L.L.Z and J.S.R contributed new materials or analytical tools.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gao, Y., Zhu, J., Zhao, C. et al. 60Coγ induction improves the protective effect of Acetobacter pasteurianus against ionizing radiation in mice. Appl Microbiol Biotechnol 105, 9285–9295 (2021). https://doi.org/10.1007/s00253-021-11664-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11664-6