Abstract
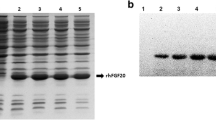
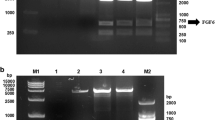
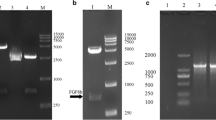
In recent years, an increasing number of studies have shown that fibroblast growth factor 12 (FGF12) plays important roles in regulating neural development and function. Importantly, changes of FGF12 expression are thought to be related to the pathophysiology of many neurological diseases. However, little research has been performed to explore the protective effect of FGF12 on nerve damage. This study aims to explore its neuroprotective effects using our recombinant humanized FGF12 (rhFGF12). The hFGF12 gene was cloned and ligated into an expression vector to construct a recombinant plasmid pET-3a-hFGF12. Single colonies were screened to obtain high expression engineering strains, and fermentation and purification protocols for rhFGF12 were designed and optimized. The biological activities and related mechanisms of rhFGF12 were investigated by MTT assay using NIH3T3 and PC12 cell lines. The in vitro neurotoxicity model of H2O2-induced oxidative injury in PC12 cells was established to explore the protective effects of rhFGF12. The results indicate that the beneficial effects of rhFGF12 were most likely achieved by promoting cell proliferation and reducing apoptosis. Moreover, a transgenic zebrafish (islet) with strong GFP fluorescence in the motor neurons of the hindbrain was used to establish a central injury model caused by mycophenolate mofetil (MMF). The results suggested that rhFGF12 could ameliorate central injury induced by MMF in zebrafish. In conclusion, we have established an efficient method to express and purify active rhFGF12 using an Escherichia coli expression system. Besides, rhFGF12 plays a protective effect of on nerve damage, and it provides a promising therapeutic approach for nerve injury.
Key points
• Effective expression and purification of bioactive rhFGF12 protein in E. coli.
• ERK/MAPK pathway is involved in rhFGF12-stimulated proliferation on PC12 cells.
• The rhFGF12 has the neuroprotective effects by inhibiting apoptosis.





Similar content being viewed by others
References
Alibolandi M, Mirzahoseini H (2011) Purification and refolding of overexpressed human basic fibroblast growth factor in Escherichia coli. Biotechnol Res Int 2011:973741–973746. https://doi.org/10.4061/2011/973741
Al-Mehmadi S, Splitt M, Ramesh V, DeBrosse S, Dessoffy K, Xia F, Minassian BA (2016) FHF1 (FGF12) epileptic encephalopathy. Neurol Genet 2:e115. https://doi.org/10.1212/nxg.0000000000000115
Beenken A, Mohammadi M (2009) The FGF family: biology, pathophysiology and therapy. Nat Rev Drug Discov 8:235–253. https://doi.org/10.1038/nrd2792
Brennan CH (2011) Zebrafish behavioural assays of translational relevance for the study of psychiatric disease. Rev Neurosci 22:37–48. https://doi.org/10.1515/rns.2011.006
Burgess HA, Granato M (2007) Sensorimotor gating in larval zebrafish. J Neurosci 27:4984–4994. https://doi.org/10.1523/jneurosci.0615-07.2007
Castellanos-Mendoza A, Castro-Acosta RM, Olvera A, Zavala G, Mendoza-Vera M, García-Hernández E, Valdez-Cruz NA (2014) Influence of pH control in the formation of inclusion bodies during production of recombinant sphingomyelinase-D in Escherichia coli. Microb Cell Factories 13:137. https://doi.org/10.1186/s12934-014-0137-9
Chen L, Wu X, Shen T, Wang X, Wang S, Wang J, Ren D (2019) Protective effects of ethyl gallate on H2O2-induced mitochondrial dysfunction in PC12 cells. Metab Brain Dis 34:545–555. https://doi.org/10.1007/s11011-019-0382-z
Cunliffe VT (2016) Building a zebrafish toolkit for investigating the pathobiology of epilepsy and identifying new treatments for epileptic seizures. J Neurosci Methods 260:91–95. https://doi.org/10.1016/j.jneumeth.2015.07.015
Das KP, Freudenrich TM, Mundy WR (2004) Assessment of PC12 cell differentiation and neurite growth: a comparison of morphological and neurochemical measures. Neurotoxicol Teratol 26:397–406. https://doi.org/10.1016/j.ntt.2004.02.006
Fontana BD, Mezzomo NJ, Kalueff AV, Rosemberg DB (2018) The developing utility of zebrafish models of neurological and neuropsychiatric disorders: a critical review. Exp Neurol 299:157–171. https://doi.org/10.1016/j.expneurol.2017.10.004
Goldfarb M (2005) Fibroblast growth factor homologous factors: evolution, structure, and function. Cytokine Growth Factor Rev 16:215–220. https://doi.org/10.1016/j.cytogfr.2005.02.002
Goldfarb M, Schoorlemmer J, Williams A, Diwakar S, Wang Q, Huang X, D'Angelo E (2007) Fibroblast growth factor homologous factors control neuronal excitability through modulation of voltage-gated sodium channels. Neuron 55:449–463. https://doi.org/10.1016/j.neuron.2007.07.006
Goshen-Lago T, Melamed D, Admon A, Engelberg D (2017) Isolation and characterization of intrinsically active (MEK-independent) mutants of Mpk1/ERK. Methods Mol Biol 1487:65–88. https://doi.org/10.1007/978-1-4939-6424-6_5
Guella I, Huh L, McKenzie MB, Toyota EB, Bebin EM, Thompson ML, Demos M (2016) De novo FGF12 mutation in 2 patients with neonatal-onset epilepsy. Neurol Genet 2:e120. https://doi.org/10.1212/nxg.0000000000000120
Hanada Y, Nakamura Y, Ozono Y, Ishida Y, Takimoto Y, Taniguchi M, Shimada S (2018) Fibroblast growth factor 12 is expressed in spiral and vestibular ganglia and necessary for auditory and equilibrium function. Sci Rep 8:11491. https://doi.org/10.1038/s41598-018-28618-0
Harada M, Murakami H, Okawa A, Okimoto N, Hiraoka S, Nakahara T, Koseki H (2009) FGF9 monomer-dimer equilibrium regulates extracellular matrix affinity and tissue diffusion. Nat Genet 41:289–298. https://doi.org/10.1038/ng.316
Hartung H, Feldman B, Lovec H, Coulier F, Birnbaum D, Goldfarb M (1997) Murine FGF-12 and FGF-13: expression in embryonic nervous system, connective tissue and heart. Mech Dev 64:31–39. https://doi.org/10.1016/s0925-4773(97)00042-7
Hecht HJ, Adar R, Hofmann B, Bogin O, Weich H, Yayon A (2001) Structure of fibroblast growth factor 9 shows a symmetric dimer with unique receptor- and heparin-binding interfaces. Acta Crystallogr D Biol Crystallogr 57:378–384. https://doi.org/10.1107/s0907444900020813
Hennessey JA, Wei EQ, Pitt GS (2013) Fibroblast growth factor homologous factors modulate cardiac calcium channels. Circ Res 113:381–388. https://doi.org/10.1161/circresaha.113.301215
Huang K, Shen L, Niu T, Zhao Y, Fu J, Cao Y (2019) Naomaitai ameliorated brain damage in rats with vascular dementia by PI3K/PDK1/AKT signaling pathway. Evid Based Complement Alternat Med 2019:2702068–2702017. https://doi.org/10.1155/2019/2702068
Hui Q, Jin Z, Li X, Liu C, Wang X (2018) FGF family: from drug development to clinical application. Int J Mol Sci 19. https://doi.org/10.3390/ijms19071875
Kalinina J, Byron SA, Makarenkova HP, Olsen SK, Eliseenkova AV, Larochelle WJ, Mohammadi M (2009) Homodimerization controls the fibroblast growth factor 9 subfamily’s receptor binding and heparan sulfate-dependent diffusion in the extracellular matrix. Mol Cell Biol 29:4663–4678. https://doi.org/10.1128/mcb.01780-08
Kim YH, Lee Y, Kim D, Jung MW, Lee CJ (2010) Scopolamine-induced learning impairment reversed by physostigmine in zebrafish. Neurosci Res 67:156–161. https://doi.org/10.1016/j.neures.2010.03.003
Lin H, Lu P, Zhou M, Wu F, Weng L, Meng K, Tian H (2019) Purification of recombinant human fibroblast growth factor 13 in E. coli and its molecular mechanism of mitogenesis. Appl Microbiol Biotechnol 103:7017–7027. https://doi.org/10.1007/s00253-019-09973-y
Liu CJ, Dib-Hajj SD, Renganathan M, Cummins TR, Waxman SG (2003) Modulation of the cardiac sodium channel Nav1.5 by fibroblast growth factor homologous factor 1B. J Biol Chem 278:1029–1036. https://doi.org/10.1074/jbc.M207074200
Manu TM, Anand T, Pandareesh MD, Kumar PB, Khanum F (2019) Terminalia arjuna extract and arjunic acid mitigate cobalt chloride-induced hypoxia stress-mediated apoptosis in H9c2 cells. Naunyn Schmiedeberg's Arch Pharmacol 392:1107–1119. https://doi.org/10.1007/s00210-019-01654-x
Marcon M, Herrmann AP, Mocelin R, Rambo CL, Koakoski G, Piato AL (2016) Prevention of unpredictable chronic stress-related phenomena in zebrafish exposed to bromazepam, fluoxetine and nortriptyline. Psychopharmacology 233:3815–3824. https://doi.org/10.1007/s00213-016-4408-5
Moldovan GL, Pfander B, Jentsch S (2007) PCNA, the maestro of the replication fork. Cell 129:665–679. https://doi.org/10.1016/j.cell.2007.05.003
Nakayama F, Yasuda T, Umeda S, Asada M, Imamura T, Meineke V, Akashi M (2011) Fibroblast growth factor- 12 (FGF12) translocation into intestinal epithelial cells is dependent on a novel cell-penetrating peptide domain: involvement of internalization in the in vivo role of exogenous FGF12. J Biol Chem 286:25823–25834. https://doi.org/10.1074/jbc.M110.198267
Nybakken K, Perrimon N (2002) Heparan sulfate proteoglycan modulation of developmental signaling in drosophila. Biochim Biophys Acta 1573:280–291. https://doi.org/10.1016/s0304-4165(02)00395-1
Olsen SK, Garbi M, Zampieri N, Eliseenkova AV, Ornitz DM, Goldfarb M, Mohammadi M (2003) Fibroblast growth factor (FGF) homologous factors share structural but not functional homology with FGFs. J Biol Chem 278:34226–34236. https://doi.org/10.1074/jbc.M303183200
Ornitz DM (2000) FGFs, heparan sulfate and FGFRs: complex interactions essential for development. Bioessays 22:108–112. https://doi.org/10.1002/(sici)1521-1878(200002)22:2<108:Aid-bies2>3.0.Co;2-m
Shin M, Franks CE, Hsu KL (2018) Isoform-selective activity-based profiling of ERK signaling. Chem Sci 9:2419–2431. https://doi.org/10.1039/c8sc00043c
Siekierska A, Isrie M, Liu Y, Scheldeman C, Vanthillo N, Lagae L, Buyse GM (2016) Gain-of-function FHF1 mutation causes early-onset epileptic encephalopathy with cerebellar atrophy. Neurology 86:2162–2170. https://doi.org/10.1212/wnl.0000000000002752
Singh SM, Eshwari AN, Garg LC, Panda AK (2005) Isolation, solubilization, refolding, and chromatographic purification of human growth hormone from inclusion bodies of Escherichia coli cells: a case study. Methods Mol Biol 308:163–176. https://doi.org/10.1385/1-59259-922-2:163
Smallwood PM, Munoz-Sanjuan I, Tong P, Macke JP, Hendry SH, Gilbert DJ, Nathans J (1996) Fibroblast growth factor (FGF) homologous factors: new members of the FGF family implicated in nervous system development. Proc Natl Acad Sci U S A 93:9850–9857. https://doi.org/10.1073/pnas.93.18.9850
Stewart A, Gaikwad S, Kyzar E, Green J, Roth A, Kalueff AV (2012) Modeling anxiety using adult zebrafish: a conceptual review. Neuropharmacology 62:135–143. https://doi.org/10.1016/j.neuropharm.2011.07.037
Stewart AM, Ullmann JF, Norton WH, Parker MO, Brennan CH, Gerlai R, Kalueff AV (2015) Molecular psychiatry of zebrafish. Mol Psychiatry 20:2–17. https://doi.org/10.1038/mp.2014.128
Takeguchi R, Haginoya K, Uchiyama Y, Fujita A, Nagura M, Takeshita E, Sasaki M (2018) Two Japanese cases of epileptic encephalopathy associated with an FGF12 mutation. Brain and Development 40:728–732. https://doi.org/10.1016/j.braindev.2018.04.002
Thomadaki H, Scorilas A (2006) BCL2 family of apoptosis-related genes: functions and clinical implications in cancer. Crit Rev Clin Lab Sci 43:1–67. https://doi.org/10.1080/10408360500295626
Tian H, Zhao Y, Chen N, Wu M, Gong W, Zheng J, Jiang C (2016) High production in E. coli of biologically active recombinant human fibroblast growth factor 20 and its neuroprotective effects. Appl Microbiol Biotechnol 100:3023–3034. https://doi.org/10.1007/s00253-015-7168-y
Wang C, Wang C, Hoch EG, Pitt GS (2011) Identification of novel interaction sites that determine specificity between fibroblast growth factor homologous factors and voltage-gated sodium channels. J Biol Chem 286:24253–24263. https://doi.org/10.1074/jbc.M111.245803
Wang S, Lin H, Zhao T, Huang S, Fernig DG, Xu N, Tian H (2017) Expression and purification of an FGF9 fusion protein in E. coli, and the effects of the FGF9 subfamily on human hepatocellular carcinoma cell proliferation and migration. Appl Microbiol Biotechnol 101:7823–7835. https://doi.org/10.1007/s00253-017-8468-1
Wang Z, Zhou F, Dou Y, Tian X, Liu C, Li H, Chen G (2018) Melatonin alleviates intracerebral hemorrhage-induced secondary brain injury in rats via suppressing apoptosis, inflammation, oxidative stress, DNA damage, and mitochondria injury. Transl Stroke Res 9:74–91. https://doi.org/10.1007/s12975-017-0559-x
Wei X, Ao Q, Meng L, Xu Y, Lu C, Tang S, Li X (2020) Expression, purification and functional assessment of asprosin inclusion body. Nan Fang Yi Ke Da Xue Xue Bao 40: 67-72. doi: 10.12122/j.issn.1673-4254.2020.01.11
Wu M, Song N, Cheng J, Zhao Y, Chen N, Ma J, Wang H (2017) Increased production of human fibroblast growth factor 17 in Escherichia coli and proliferative activity in NIH3T3 cells. Mol Med Rep 16:447–452. https://doi.org/10.3892/mmr.2017.6575
Zhang X, Bao L, Yang L, Wu Q, Li S (2012) Roles of intracellular fibroblast growth factors in neural development and functions. Sci China Life Sci 55:1038–1044. https://doi.org/10.1007/s11427-012-4412-x
Zhao M, Tao XY, Wang FQ, Ren YH, Wei DZ (2018) Establishment of a low-dosage-IPTG inducible expression system construction method in Escherichia coli. J Basic Microbiol 58:806–810. https://doi.org/10.1002/jobm.201800160
Data availability statement
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
Funding
This work was funded by the Natural Science Foundation of Zhejiang Province (No. LY19H160029) and the Zhejiang Provincial Department of Education (No. Y201839837).
Author information
Authors and Affiliations
Contributions
HT and XL conceived and designed research. MZ, KM, YZ, MZ, PL, YF, and MH conducted experiments. JC and QD contributed new reagents or analytical tools. MZ and KM analyzed data. MZ and KM wrote the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, M., Chen, J., Meng, K. et al. Production of bioactive recombinant human fibroblast growth factor 12 using a new transient expression vector in E. coli and its neuroprotective effects. Appl Microbiol Biotechnol 105, 5419–5431 (2021). https://doi.org/10.1007/s00253-021-11430-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11430-8