Abstract
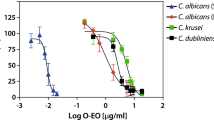
Candida albicans invasion is one of the most serious fungal infections in clinical history. In recent years, because of the widespread use of immunosuppressive drugs, chemotherapy drugs, glucocorticoids, and broad-spectrum antibiotics, serious drug resistance has been reported; therefore, a new type of antifungal drug needs to be developed. In this study, we found that Nerol (NEL) had strong antimicrobial activity and 0.77 μL/mL NEL was the minimum inhibitory concentration (MIC) effective against C. albicans. We determined the change of the growth curve of NEL for C. albicans, to identify the trend of NEL activity against C. albicans. Through the determination of the ergosterol content and glucose-induced extracellular fluid acidification of NEL on C. albicans, we found that NEL inhibits the growth of C. albicans by destroying cell membranes. This finding was also supported by the expression of SAP (secreted aspartyl proteinase) involved in cell membrane synthesis. Finally, demonstrations of phenotype investigation, colony-forming unit (CFU) counts, and PAS (periodic acid-Schiff) staining were conducted to prove that NEL had the ability to treated mouse oral C. albicans infection and vaginal C. albicans infection. This research may help us to investigate new antimicrobial agents for treating C. albicans infections.
Key Points
• NEL can inhibit the growth of C. albicans.
• NEL destroys the cell membrane formation and permeability of C. albicans.
• NEL can treat vulvovaginal candidiasis and oropharyngeal candidiasis in mice.
• NEL could be used as a possible antifungal agent.










Similar content being viewed by others
References
Adriana GR, Eva GTM, Francisco P, Francisco LM (2012) Anti-nociceptive and anti-inflammatory activities of the Agastache mexicana extracts by using several experimental models in rodents. J Ethnopharmacol 142(3):700–705. https://doi.org/10.1016/j.jep.2012.05.044
Aoki W, Kitahara N, Miura N, Morisaka H, Yamamoto Y, Kuroda K, Ueda M (2011) Comprehensive characterization of secreted aspartic proteases encoded by a virulence gene family in Candida albicans. Biochem 150(4):431–438. https://doi.org/10.1093/jb/mvr073
Aviles P, Falcoz C, San Roman R, Gargallo-Viola D (2000) Pharmacokinetics-pharmacodynamics of a sordarin derivative (GM 237354) in a murine model of lethal candidiasis. Antimicrob Agents Chemother 44(9):2333–2340. https://doi.org/10.1128/aac.44.9.2333-2340.2000
Baddley JW, Pappas PG (2005) Antifungal combination therapy: clinical potential. Drugs 65(11):1461–1480. https://doi.org/10.2165/00003495-200565110-00002
Barchiesi F, Schimizzi AM, Najvar LK, Bocanegra R, Caselli F, Di Cesare S, Giannini D, Di Francesco LF, Giacometti A, Carle F, Scalise G, Graybill JR (2001) Interactions of posaconazole and flucytosine against Cryptococcus neoformans. Antimicrob Agents Chemother 45(5):1355–1359. https://doi.org/10.1128/AAC.45.5.1355-1359.2001
Benmansour W, Boucherit-Otmani Z, Boucherit K (2014) Dormancy of Candida albicans ATCC10231 in the presence of amphotericin B. Investigation using the scanning electron microscope (SEM). Mycol Med 24(3):e93–e100. https://doi.org/10.1016/j.mycmed.2014.02.007
Berman J, Sudbery PE (2002) Candida albicans: a molecular revolution built on lessons from budding yeast. Nat Rev Genet 3(12):918–930. https://doi.org/10.1038/nrg948
Brown GD, Denning DW, Gow NA, Levitz SM, Netea MG, White TC (2012) Hidden killers: human fungal infections. Sci Transl Med 4(165):165rv13. https://doi.org/10.1126/scitranslmed.3004404
Chen L, Wang Z, Liu L, Qu S, Mao Y, Peng X, Li YX, Tian J (2019) Cinnamaldehyde inhibits Candida albicans growth by causing apoptosis and its treatment on vulvovaginal candidiasis and oropharyngeal candidiasis. Appl Microbiol Biotechnol 103(21–22):9037–9055. https://doi.org/10.1007/s00253-019-10119-3
Cox SD, Mann CM, Markham JL (2001) Interactions between components of the essential oil of Melaleuca alternifolia. J Appl Microbiol 91(3):492–497. https://doi.org/10.1046/j.1365-2672.2001.01406.x
Dang S, Atiq M, Saccente M, Olden KW, Aduli F (2009) Isolated tuberculosis of the pancreas: a case report. JOP 10(1):64–66. https://doi.org/10.5580/206e
Feng W, Yang J, Pan Y, Xi Z, Qiao Z, Ma Y (2016) The correlation of virulence, pathogenicity, and itraconazole resistance with SAP activity in Candida albicans strains. Can J Microbiol 62(2):173–178. https://doi.org/10.1139/cjm-2015-0457
Gao M, Wang H, Zhu LJCP (2016) Biochemistry BIJoECP, Pharmacology quercetin assists fluconazole to inhibit biofilm formations of fluconazole-resistant Candida albicans in in vitro and in vivo antifungal managements of vulvovaginal candidiasis. Cell Physiol Biochem 40(3–4):727–742. https://doi.org/10.1159/000453134
Gonzalez-Ramirez AE, Gonzalez-Trujano ME, Orozco-Suarez SA, Alvarado-Vasquez N, Lopez-Munoz FJ (2016) Nerol alleviates pathologic markers in the oxazolone-induced colitis model. Eur J Pharmacol 776:81–89. https://doi.org/10.1016/j.ejphar.2016.02.036
Hong E, Dixit S, Fidel PL, Bradford J, Fischer G (2014) Vulvovaginal candidiasis as a chronic disease: diagnostic criteria and definition. J Low Genit Tract Dis 18(1):31–38. https://doi.org/10.1097/LGT.0b013e318287aced
Hornbach A, Heyken A, Schild L, Hube B, Loffler J, Kurzai O (2009) The glycosylphosphatidylinositol-anchored protease Sap9 modulates the interaction of Candida albicans with human neutrophils. Infect Immun 77(12):5216–5224. https://doi.org/10.1128/IAI.00723-09
Jirovetz L, Buchbauer G, Schmidt E, Stoyanova AS, Denkova Z, Nikolova R, Geissler M (2007) Purity, antimicrobial activities and olfactoric evaluations of geraniol/nerol and various of their derivatives. J Essent Oil Res 19(3):288–291. https://doi.org/10.1080/10412905.2007.9699283
Kesavan C, Raghunathan M, Ganesan N (2005) Morphological and growth altering effects of Cisplatin in C. albicans using fluorescence microscopy. Ann Clin Microbiol Antimicrob 4(1):7. https://doi.org/10.1186/1476-0711-4-7
Lan CY, Rodarte G, Murillo LA, Jones T, Agabian N (2004) Regulatory networks affected by iron availability in Candida albicans: iron regulation in C. albicans. Mol Microbiol 53(5):1451–1469. https://doi.org/10.1111/j.1365-2958.2004.04214.x
Maesaki S, Hossain MA, Miyazaki Y, Tomono K, Tashiro T, Kohno S (2000) Efficacy of FK463, a (1,3)-beta-D-glucan synthase inhibitor, in disseminated azole-resistant Candida albicans infection in mice. Antimicrob Agents Chemother 44(6):1728–1730. https://doi.org/10.1128/aac.44.6.1728-1730.2000
Manavathu EK, Dimmock JR, Vashishtha SC, Chandrasekar PH (1999) Proton-pumping-ATPase-targeted antifungal activity of a novel conjugated styryl ketone. Antimicrob Agents Chemother 43(12):2950–2959. https://doi.org/10.1128/aac.43.12.2950
Martin R, Albrecht-Eckardt D, Brunke S, Hube B, Hunniger K, Kurzai O (2013) A core filamentation response network in Candida albicans is restricted to eight genes. PLoS One 8(3):e58613. https://doi.org/10.1371/journal.pone.0058613
Matuschak GM, Klein CA, Tredway TL, Schilly DR, Lechner AJ (1993) Tnf-alpha and cyclooxygenase metabolites do not modulate Candida albicans septic shock with disseminated candidiasis. J Appl Physiol 74(5):2432–2442. https://doi.org/10.1152/jappl.1993.74.5.2432
Mayer FL, Wilson D, Hube B (2013) Candida albicans pathogenicity mechanisms. Virulence 4(2):119–128. https://doi.org/10.4161/viru.22913
Munoz JE, Rossi DCP, Ishida K, Spadari CC, Melhem MSC, Garcia DM, Caires ACF, Taborda CP, Rodrigues EG (2017) Antifungal activity of the biphosphinic cyclopalladate C7a against Candida albicans yeast forms in vitro and in vivo. Front Microbiol 8:771. https://doi.org/10.3389/fmicb.2017.00771
Nobile CJ, Johnson AD (2015) Candida albicans biofilms and human disease. Annu Rev Microbiol 69(1):71–92. https://doi.org/10.1146/annurev-micro-091014-104330
Peters BM, Yano J, Noverr MC, Fidel PL Jr (2014) Candida vaginitis: when opportunism knocks, the host responds. PLoS Pathog 10(4):e1003965. https://doi.org/10.1371/journal.ppat.1003965
Qu S, Chen L, Tian H, Wang Z, Wang F, Wang L, Li J, Ji H, Xi L, Feng Z, Tian J, Feng Z (2019) Effect of perillaldehyde on prophylaxis and treatment of vaginal candidiasis in a murine model. Front Microbiol 10:1466. https://doi.org/10.3389/fmicb.2019.01466
Reus O, Vik A, Kolter R, Morschhauser J (2004) The SAT1 flipper, an optimized tool for gene disruption in Candida albicans. Gene 341:119–127. https://doi.org/10.1016/j.gene.2004.06.021
Rodriguez RJ, Low C, Bottema CD, Parks LW (1985) Multiple functions for sterols in Saccharomyces cerevisiae. Biochim Biophys Acta 837(3):336–343. https://doi.org/10.1016/0005-2760(85)90057-8
Semlali A, Killer K, Alanazi H, Chmielewski W, Rouabhia M (2014) Cigarette smoke condensate increases C. albicans adhesion, growth, biofilm formation, and EAP1, HWP1 and SAP2 gene expression. BMC Microbiol 14(1):61. https://doi.org/10.1186/1471-2180-14-61
Shao J, Zhang M, Wang T, Li Y, Wang C (2016) The roles of CDR1, CDR2, and MDR1 in kaempferol-induced suppression with fluconazole-resistant Candida albicans. Pharm Biol 54(6):984–992. https://doi.org/10.3109/13880209.2015.1091483
Staab JF, Sundstrom P (2003) URA3 as a selectable marker for disruption and virulence assessment of Candida albicans genes. Trends Microbiol 11(2):69–73. https://doi.org/10.1016/S0966-842x(02)00029-X
Tian J, Ban X, Zeng H, He J, Chen Y, Wang Y (2012a) The mechanism of antifungal action of essential oil from dill (Anethum graveolens L.) on Aspergillus flavus. PLoS One 7(1):e30147. https://doi.org/10.1371/journal.pone.0030147
Tian J, Huang B, Luo XL, Zeng H, Ban XQ, He JS, Wang YW (2012b) The control of Aspergillus flavus with Cinnamomum jensenianum Hand-Mazz essential oil and its potential use as a food preservative. Food Chem 130(3):520–527. https://doi.org/10.1016/j.foodchem.2011.07.061
Tian J, Xiaobin Z, Hong Z, Zhaozhong F, Xiangmin M, Xue P (2013) Investigations on the antifungal effect of Nerol against Aspergillus flavus causing food spoilage. Sci World J:1–8. https://doi.org/10.1155/2013/230795
Tian J, Lu Z, Wang Y, Zhang M, Wang X, Tang X, Peng X, Zeng H (2017) Nerol triggers mitochondrial dysfunction and disruption via elevation of Ca2+ and ROS in Candida albicans. Int J Biochem Cell Biol 85(Complete):114–122. https://doi.org/10.1016/j.biocel.2017.02.006
Wang Y, Zeng X, Zhou Z, Xing K, Tessema A, Zeng H, Tian JJFC (2015) Inhibitory effect of nerol against Aspergillus niger on grapes through a membrane lesion mechanism. Food Control 55:54–61. https://doi.org/10.1016/j.foodcont.2015.02.029
Wang L, Wang C, Mei H, Shen Y, Lv G, Zeng R, Zhan P, Li D, Liu W (2016) Combination of estrogen and immunosuppressive agents to establish a mouse model of candidiasis with concurrent oral and vaginal mucosal infection. Mycopathologia 181(1–2):29–39. https://doi.org/10.1007/s11046-015-9947-5
White TC, Agabian N (1995) Candida albicans secreted aspartyl proteinases: isoenzyme pattern is determined by cell type, and levels are determined by environmental factors. J Bacteriol 177(18):5215–5221
Zaoutis TE, Argon J, Chu J, Berlin JA, Walsh TJ, Feudtner C (2005) The epidemiology and attributable outcomes of candidemia in adults and children hospitalized in the United States: a propensity analysis. Clin Infect Dis 41(9):1232–1239. https://doi.org/10.1086/496922
Zhang S, Ahearn DG, Mateus C, Crow SA Jr (2006) In vitro effects of Ag+ on planktonic and adhered cells of fluconazole-resistant and susceptible strains of Candida albicans, C. glabrata and C. krusei. Biomaterials 27(13):2755–2760. https://doi.org/10.1016/j.biomaterials.2005.12.010
Funding
This study was funded by National Natural Science Foundation of China (31972171, 31671944), Six Talent Peaks Project of Jiangsu Province (SWYY-026), Qing Lan Project of Jiangsu Province, Natural Science Foundation by Xuzhou City (KC17053), and the PAPD of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Contributions
JT and XP designed the experiments. ZW and YR performed the experiments. LC and QS analyzed the data. ZW and KLY drafted the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical statement
The study was carried out in accordance with recommendations for use and laboratory animal care as prescribed by the international association and approved by the ethics committee of Jiangsu Normal University. The agreement was authorized by the Animal Experiment Committee of Jiangsu Normal University (No. 201703003; March 6, 2017) and ensured that the pain of the experimental animals was minimized.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, Z., Yang, K., Chen, L. et al. Activities of Nerol, a natural plant active ingredient, against Candida albicans in vitro and in vivo. Appl Microbiol Biotechnol 104, 5039–5052 (2020). https://doi.org/10.1007/s00253-020-10559-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10559-2