Abstract
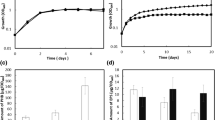
Establishment of the rhizobia-legume symbiosis is usually accompanied by hydrogen peroxide (H2O2) production by the legume host at the site of infection, a process detrimental to rhizobia. In Azorhizobium caulinodans ORS571, deletion of chp1, a gene encoding c-di-GMP phosphodiesterase, led to increased resistance against H2O2 and to elevated nodulation efficiency on its legume host Sesbania rostrata. Three domains were identified in the Chp1: a PAS domain, a degenerate GGDEF domain, and an EAL domain. An in vitro enzymatic activity assay showed that the degenerate GGDEF domain of Chp1 did not have diguanylate cyclase activity. The phosphodiesterase activity of Chp1 was attributed to its EAL domain which could hydrolyse c-di-GMP into pGpG. The PAS domain functioned as a regulatory domain by sensing oxygen. Deletion of Chp1 resulted in increased intracellular c-di-GMP level, decreased motility, increased aggregation, and increased EPS (extracellular polysaccharide) production. H2O2-sensitivity assay showed that increased EPS production could provide ORS571 with resistance against H2O2. Thus, the elevated nodulation efficiency of the ∆chp1 mutant could be correlated with a protective role of EPS in the nodulation process. These data suggest that c-di-GMP may modulate the A. caulinodans-S. rostrata nodulation process by regulating the production of EPS which could protect rhizobia against H2O2.








Similar content being viewed by others
References
Akiba N, Aono T, Toyazaki H, Sato S, Oyaizu H (2010) phrR-like gene praR of Azorhizobium caulinodans ORS571 is essential for symbiosis with Sesbania rostrata and is involved in expression of reb genes. Appl Environ Microbiol 76(11):3475–3485. https://doi.org/10.1128/AEM.00238-10
An S, Wu J, Zhang LH (2010) Modulation of Pseudomonas aeruginosa biofilm dispersal by a cyclic-di-GMP phosphodiesterase with a putative hypoxia-sensing domain. Appl Environ Microbiol 76(24):8160–8173. https://doi.org/10.1128/aem.01233-10
Boehm A, Kaiser M, Li H, Spangler C, Kasper CA, Ackermann M, Kaever V, Sourjik V, Roth V, Jenal U (2010) Second messenger-mediated adjustment of bacterial swimming velocity. Cell 141(1):107–116. https://doi.org/10.1016/j.cell.2010.01.018
Bourret RB (2010) Receiver domain structure and function in response regulator proteins. Curr Opin Microbiol 13(2):142–149
Caldara M, Friedlander RS, Kavanaugh NL, Aizenberg J, Foster KR, Ribbeck K (2012) Mucin biopolymers prevent bacterial aggregation by retaining cells in the free-swimming state. Curr Biol 22(24):2325–2330. https://doi.org/10.1016/j.cub.2012.10.028
Capoen W, Oldroyd G, Goormachtig S, Holsters M (2010) Sesbania rostrata: a case study of natural variation in legume nodulation. New Phytol 186(2):340–345. https://doi.org/10.1111/j.1469-8137.2009.03124.x
Cardenas L, Martinez A, Sanchez F, Quinto C (2008) Fast, transient and specific intracellular ROS changes in living root hair cells responding to Nod factors (NFs). Plant J 56(5):802–813. https://doi.org/10.1111/j.1365-313X.2008.03644.x
Chan C, Paul R, Samoray D, Amiot NC, Giese B, Jenal U, Schirmer T (2004) Structural basis of activity and allosteric control of diguanylate cyclase. P Natl Acad Sci USA 101(49):17084–17089. https://doi.org/10.1073/pnas.0406134101
Chang AL, Tuckerman JR, Gonzalez G, Mayer R, Weinhouse H, Volman G, Amikam D, Benziman M, Gonzalez MA (2001) Phosphodiesterase A1, a regulator of cellulose synthesis in Acetobacter xylinum, is a heme-based sensor. Biochemistry 40:3420–3426
Cho US, Bader MW, Amaya MF, Daley ME, Klevit RE, Miller SI, Xu W (2006) Metal bridges between the PhoQ sensor domain and the membrane regulate transmembrane signaling. J Mol Biol 356(5):1193–1206. https://doi.org/10.1016/j.jmb.2005.12.032
Christen M, Christen B, Folcher M, Schauerte A, Jenal U (2005) Identification and characterization of a cyclic di-GMP-specific phosphodiesterase and its allosteric control by GTP. J Biol Chem 280(35):30829–30837. https://doi.org/10.1074/jbc.M504429200
Chua S, Ding Y, Liu Y, Cai Z, Zhou J, Swarup S, Moses DI, Schuster SC, Kjelleberg S, Givskov M, Yang L (2016) Reactive oxygen species drive evolution of pro-biofilm variants in pathogens by modulating cyclic-di-GMP levels. Open Biol 6(11):160162. https://doi.org/10.1098/rsob.160162
Colley B, Dederer V, Carnell M, Kjelleberg S, Rice SA, Klebensberger J (2016) SiaA/D interconnects c-di-GMP and RsmA signaling to coordinate cellular aggregation of Pseudomonas aeruginosa in response to environmental conditions. Front Microbiol 7:179. https://doi.org/10.3389/fmicb.2016.00179
D'Haeze W, De Rycke R, Mathis R, Goormachtig S, Pagnotta S, Verplancke C, Capoen W, Holsters M (2003) Reactive oxygen species and ethylene play a positive role in lateral root base nodulation of a semiaquatic legume. P Natl Acad Sci USA 100(20):11789–11794. https://doi.org/10.1073/pnas.1333899100
D'Haeze W, Glushka J, De Rycke R, Holsters M, Carlson RW (2004) Structural characterization of extracellular polysaccharides of Azorhizobium caulinodans and importance for nodule initiation on Sesbania rostrata. Mol Microbiol 52(2):485–500. https://doi.org/10.1111/j.1365-2958.2004.03989.x
Dénarié J, Debellé F, Promé J (1996) Rhizobium lipo-chitooligosaccharide nodulation factors: signaling molecules mediating recognition and morphogenesis. Annu Rev Biochem 65(1):503–535
Dreyfus B, Garcia JL, Gillis M (1988) Characterization of Azorhizobium caulinodans gen. nov., sp. nov. , a stem nodulating nitrogen-fixing bacterium isolated from Sesbania rostrata. Int J Syst Bacteriol 38(1):89–98
Enomoto G, Win N, Narikawa R, Ikeuchi M (2015) Three cyanobacteriochromes work together to form a light color-sensitive input system for c-di-GMP signaling of cell aggregation. P Natl Acad Sci USA 112(26):8082–8087. https://doi.org/10.1073/pnas.1504228112
Ferreira RB, Antunes LC, Greenberg EP, McCarter LL (2008) Vibrio parahaemolyticus ScrC modulates cyclic dimeric GMP regulation of gene expression relevant to growth on surfaces. J Bacteriol 190(3):851–860. https://doi.org/10.1128/JB.01462-07
Flemming HC, Wingender J (2010) The biofilm matrix. Nat Rev Microbiol 8(9):623–633. https://doi.org/10.1038/nrmicro2415
Flint A, Stintzi A, Saraiva LM (2016) Oxidative and nitrosative stress defences of Helicobacter and Campylobacter species that counteract mammalian immunity. FEMS Microbiol Rev 40(6):938–960. https://doi.org/10.1093/femsre/fuw025
Galperin MY (2004) Bacterial signal transduction network in a genomic perspective. Environ Microbiol 6(6):552–567. https://doi.org/10.1111/j.1462-2920.2004.00633.x
Gao L, Pan X, Zhang D, Mu S, Lee DJ, Halik U (2015) Extracellular polymeric substances buffer against the biocidal effect of H2O2 on the bloom-forming cyanobacterium Microcystis aeruginosa. Water Res 69:51–58. https://doi.org/10.1016/j.watres.2014.10.060
Gao M, D'Haeze W, Rycke R, Wolucka B, Holsters M (2001) Knockout of an azorhizobial dTDP-L-rhamnose synthase affects lipopolysaccharide and extracellular polysaccharide production and disables symbiosis with Sesbania rostrata. Mol Plant Microbe In 14(7):857–866
Gao S, Romdhane SB, Beullens S, Kaever V, Lambrichts I, Fauvart M, Michiels J (2014) Genomic analysis of cyclic-di-GMP-related genes in rhizobial type strains and functional analysis in Rhizobium etli. Appl Microbiol Biotechnol 98(10):4589–4602
Garcia B, Latasa C, Solano C, Garcia-del Portillo F, Gamazo C, Lasa I (2004) Role of the GGDEF protein family in Salmonella cellulose biosynthesis and biofilm formation. Mol Microbiol 54(1):264–277. https://doi.org/10.1111/j.1365-2958.2004.04269.x
Gilles-Gonzalez MA, Gonzalez G (2005) Heme-based sensors: defining characteristics, recent developments, and regulatory hypotheses. J Inorg Biochem 99(1):1–22. https://doi.org/10.1016/j.jinorgbio.2004.11.006
Goethals K, Leyman B, Den G, Montagu M, Holsters M (1994) An Azorhizobium caulinodans ORS571 locus involved in lipopolysaccharide production and nodule formation on Sesbania rostrata stems and roots. J Bacteriol 176(1):92–99
Gong W, Hao B, Mansy SS, Gonzalez G, Gonzalez MA, Chan MK (1998) Structure of a biological oxygen sensor a new mechanism for heme-driven signal transduction. P Natl Acad Sci USA 95:15177–15182
Goormachtig S, Capoen W, Holsters M (2004a) Rhizobium infection: lessons from the versatile nodulation behaviour of water-tolerant legumes. Trends Plant Sci 9(11):518–522. https://doi.org/10.1016/j.tplants.2004.09.005
Goormachtig S, Capoen W, James EK, Holsters M (2004b) Switch from intracellular to intercellular invasion during water stress-tolerant legume nodulation. P Natl Acad Sci USA 101(16):6303–6308. https://doi.org/10.1073/pnas.0401540101
Heikaus CC, Pandit J, Klevit RE (2009) Cyclic nucleotide binding GAF domains from phosphodiesterases: structural and mechanistic insights. Structure 17(12):1551–1557
Hengge R (2009) Principles of c-di-GMP signalling in bacteria. Nat Rev Microbiol 7(4):263–273
Henry JT, Crosson S (2011) Ligand-binding PAS domains in a genomic, cellular, and structural context. Annu Rev Microbiol 65(1):261–286
Hickman JW, Harwood CS (2008) Identification of FleQ from Pseudomonas aeruginosa as a c-di-GMP-responsive transcription factor. Mol Microbiol 69(2):376–389. https://doi.org/10.1111/j.1365-2958.2008.06281.x
Jamet A, Mandon K, Puppo A, Herouart D (2007) H2O2 is required for optimal establishment of the Medicago sativa/Sinorhizobium meliloti symbiosis. J Bacteriol 189(23):8741–8745. https://doi.org/10.1128/JB.01130-07
Jamet A, Sigaud S, Sype GV, Puppo A, Hérouart D (2003) Expression of the bacterial catalase genes during Sinorhizobium meliloti-Medicago sativa symbiosis and their crucial role during the infection process. Mol Plant Microbe In 16(3):217–225
Jang IA, Kim J, Park W (2016) Endogenous hydrogen peroxide increases biofilm formation by inducing exopolysaccharide production in Acinetobacter oleivorans DR1. Sci Rep-UK 6:21121. https://doi.org/10.1038/srep21121
Jenal U, Malone J (2006) Mechanisms of cyclic-di-GMP signaling in bacteria. Annu Rev Genet 40:385–407. https://doi.org/10.1146/annurev.genet.40.110405.090423
Jiang N, Liu W, Li Y, Wu H, Zhang Z, Alexandre G, Elmerich C, Xie Z (2016) A chemotaxis receptor modulates nodulation during the Azorhizobium caulinodans-Sesbania rostrata symbiosis. Appl Environ Microbiol 82(11):3174–3184. https://doi.org/10.1128/AEM.00230-16
Kazmierczak BI, Lebron MB, Murray TS (2006) Analysis of FimX, a phosphodiesterase that governs twitching motility in Pseudomonas aeruginosa. Mol Microbiol 60(4):1026–1043. https://doi.org/10.1111/j.1365-2958.2006.05156.x
Key J, Moffat K (2005) Crystal structures of deoxy and CO-bound bjFixLH reveal details of ligand recognition and signaling. Biochemistry 44(12):4627–4635
Koo H, Falsetta ML, Klein MI (2013) The exopolysaccharide matrix: a virulence determinant of cariogenic biofilm. J Dent Res 92(12):1065–1073. https://doi.org/10.1177/0022034513504218
Krasteva PV, Fong J, Shikuma NJ, Beyhan S, Navarro M, Yildiz FH, Sondermann H (2010) Vibrio cholerae VpsT regulates matrix production and motility by directly sensing cyclic di-GMP. Science 327(5967):866–868. https://doi.org/10.1126/science.1181185
Krasteva PV, Giglio KM, Sondermann H (2012) Sensing the messenger: the diverse ways that bacteria signal through c-di-GMP. Protein Sci 21(7):929–948. https://doi.org/10.1002/pro.2093
Kuchma SL, Brothers KM, Merritt JH, Liberati NT, Ausubel FM, O'Toole GA (2007) BifA, a cyclic-Di-GMP phosphodiesterase, inversely regulates biofilm formation and swarming motility by Pseudomonas aeruginosa PA14. J Bacteriol 189(22):8165–8178. https://doi.org/10.1128/JB.00586-07
Lee HS, Gu F, Ching SM, Lam Y, Chua KL (2010) CdpA is a Burkholderia pseudomallei cyclic di-GMP phosphodiesterase involved in autoaggregation, flagellum synthesis, motility, biofilm formation, cell invasion, and cytotoxicity. Infect Immun 78(5):1832–1840. https://doi.org/10.1128/IAI.00446-09
Lee KB, De Backer P, Aono T, Liu CT, Suzuki S, Suzuki T, Kaneko T, Yamada M, Tabata S, Kupfer DM, Najar FZ, Wiley GB, Roe B, Binnewies TT, Ussery DW, D'Haeze W, Herder JD, Gevers D, Vereecke D, Holsters M, Oyaizu H (2008) The genome of the versatile nitrogen fixer Azorhizobium caulinodans ORS571. BMC Genomics 9(1):271. https://doi.org/10.1186/1471-2164-9-271
Lee VT, Matewish JM, Kessler JL, Hyodo M, Hayakawa Y, Lory S (2007) A cyclic-di-GMP receptor required for bacterial exopolysaccharide production. Mol Microbiol 65(6):1474–1484. https://doi.org/10.1111/j.1365-2958.2007.05879.x
Lehman AP, Long SR (2013) Exopolysaccharides from Sinorhizobium meliloti can protect against H2O2-dependent damage. J Bacteriol 195:5362–5369
Liang ZX (2015) The expanding roles of c-di-GMP in the biosynthesis of exopolysaccharides and secondary metabolites. Nat Prod Rep 32(5):663–683. https://doi.org/10.1039/c4np00086b
Lievens S, Goormachtig S, Holsters M (2001) A critical evaluation of differential display as a tool to identify genes involved in legume nodulation looking back and looking forward. Nucleic Acids Res 29(17):3459–3468
Liu W, Yang J, Sun Y, Liu X, Li Y, Zhang Z, Xie Z (2017a) Azorhizobium caulinodans transmembrane chemoreceptor TlpA1 involved in host colonization and nodulation on roots and stems. Front Microbiol 8:1327. https://doi.org/10.3389/fmicb.2017.01327
Liu X, Liu W, Sun Y, Xia C, Elmerich C, Xie Z (2017b) Identification of a cheZ-like gene in Azorhizobium caulinodans, a key gene in the control of chemotaxis and colonization of the host plant. Appl Environ Microbiol 84(3):e-01827-17. https://doi.org/10.1128/AEM.01827-17
Marx CJ, Lidstrom ME (2002) Broad-host-range cre-lox system for antibiotic marker recycling in Gram-negative bacteria. BioTechniques 33(5):1062–1067
Matthysse AG (2014) Attachment of Agrobacterium to plant surfaces. Front Plant Sci 5:252. https://doi.org/10.3389/fpls.2014.00252
Nagata M, Murakami E, Shimoda Y, Shimoda-Sasakura F, Kucho K, Suzuki A, Abe M, Higashi S, Uchiumi T (2008) Expression of a class 1 hemoglobin gene and production of nitric oxide in response to symbiotic and pathogenic bacteria in Lotus japonicus. Mol Plant Microbe In 21(9):1175–1183. https://doi.org/10.1094/mpmi
Nakajima A, Aono T, Tsukada S, Siarot L, Ogawa T, Oyaizu H (2012) Lon protease of Azorhizobium caulinodans ORS571 is required for suppression of reb gene expression. Appl Environ Microbiol 78(17):6251–6261. https://doi.org/10.1128/AEM.01039-12
Nixon VM, Gonzalez G, Gonzalez MA (2000) Dos, a heme binding PAS protein from Escherichia coli, is a direct oxygen sensor. Biochemistry 39(10):2685–2691
Oldroyd GE (2013) Speak, friend, and enter: signalling systems that promote beneficial symbiotic associations in plants. Nat Rev Microbiol 11(4):252–263. https://doi.org/10.1038/nrmicro2990
Paul K, Nieto V, Carlquist WC, Blair DF, Harshey RM (2010) The c-di-GMP binding protein YcgR controls flagellar motor direction and speed to affect chemotaxis by a "backstop brake" mechanism. Mol Cell 38(1):128–139. https://doi.org/10.1016/j.molcel.2010.03.001
Pei Z, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activated by hydrogen peroxide mediates abscisic acid signalling in guard cells. Nature 406(6797):731–734
Perez-Mendoza D, Bertinetti D, Lorenz R, Gallegos MT, Herberg FW, Sanjuan J (2017) A novel c-di-GMP binding domain in glycosyltransferase BgsA is responsible for the synthesis of a mixed-linkage β-glucan. Sci Rep-UK 7(1):8997. https://doi.org/10.1038/s41598-017-09290-2
Perez-Mendoza D, Sanjuan J (2016) Exploiting the commons: cyclic diguanylate regulation of bacterial exopolysaccharide production. Curr Opin Microbiol 30:36–43. https://doi.org/10.1016/j.mib.2015.12.004
Purcell EB, McKee RW, McBride SM, Waters CM, Tamayo R (2012) Cyclic diguanylate inversely regulates motility and aggregation in Clostridium difficile. J Bacteriol 194(13):3307–3316. https://doi.org/10.1128/JB.00100-12
Qi X, Nellas RB, Byrn MW, Russell MH, Bible AN, Alexandre G, Shen T (2013) Swimming motility plays a key role in the stochastic dynamics of cell clumping. Phys Biol 10(2):026005. https://doi.org/10.1088/1478-3975/10/2/026005
Rao F, Yang Y, Qi Y, Liang ZX (2008) Catalytic mechanism of cyclic di-GMP-specific phosphodiesterase: a study of the EAL domain-containing RocR from Pseudomonas aeruginosa. J Bacteriol 190(10):3622–3631. https://doi.org/10.1128/JB.00165-08
Rebbapragada A, Johnson MS, Harding GP, Zuccarelli AJ, Fletcher HM, Zhulin IB, Taylor BL (1997) The Aer protein and the serine chemoreceptor Tsr independently sense intracellular energy levels and transduce oxygen redox and energy signals for Escherichia coli behavior. P Natl Acad Sci USA 94(20):10541–10546
Russell MH, Bible AN, Fang X, Gooding JR, Campagna SR, Gomelsky M, Alexandre G (2013) Integration of the second messenger c-di-GMP into the chemotactic signaling pathway. mBio 4(2):e00001–e00013. https://doi.org/10.1128/mBio.00001-13
Ryjenkov DA, Simm R, Romling U, Gomelsky M (2006) The PilZ domain is a receptor for the second messenger c-di-GMP: the PilZ domain protein YcgR controls motility in enterobacteria. J Biol Chem 281(41):30310–30314. https://doi.org/10.1074/jbc.C600179200
Ryjenkov DA, Tarutina M, Moskvin OV, Gomelsky M (2005) Cyclic diguanylate is a ubiquitous signaling molecule in bacteria: insights into biochemistry of the GGDEF protein domain. J Bacteriol 187(5):1792–1798. https://doi.org/10.1128/JB.187.5.1792-1798.2005
Santos R, Hérouart D, Sigaud S, Touati D, Puppo A (2001) Oxidative burst in alfalfa-Sinorhizobium meliloti symbiotic interaction. Mol Plant Microbe In 14(1):86–89
Schäfer A, Tauch A, Jäger W, Kalinowski J, Thierbach G, Pühler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19 selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145:69–73
Schäper S, Krol E, Skotnicka D, Kaever V, Hilker R, Sogaard-Andersen L, Becker A (2016) Cyclic di-GMP regulates multiple cellular functions in the symbiotic alphaproteobacterium Sinorhizobium meliloti. J Bacteriol 198(3):521–535
Schmidt AJ, Ryjenkov DA, Gomelsky M (2005) The ubiquitous protein domain EAL is a cyclic diguanylate-specific phosphodiesterase: enzymatically active and inactive EAL domains. J Bacteriol 187(14):4774–4781. https://doi.org/10.1128/jb.187.14.4774-4781.2005
Seaver LC, Imlay JA (2001) Hydrogen peroxide fluxes and compartmentalization inside growing Escherichia coli. J Bacteriol 183(24):7182–7189. https://doi.org/10.1128/JB.183.24.7182-7189.2001
Sekine Y, Tanzawa T, Tanaka Y, Ishimori K, Uchida T (2016) Cytoplasmic heme-binding protein (HutX) from Vibrio cholerae is an intracellular heme transport protein for the heme-degrading enzyme, HutZ. Biochemistry 55(6):884–893. https://doi.org/10.1021/acs.biochem.5b01273
Simm R, Morr M, Kader A, Nimtz M, Römling U (2004) GGDEF and EAL domains inversely regulate cyclic di-GMP levels and transition from sessility to motility. Mol Microbiol 53(4):1123–1134. https://doi.org/10.1111/j.1365-2958.2004.04206.x
Tal R, Wong H, Calhoon R, Gelfand D, Fear A, Volman G, Mayer R, Ross P (1998) Three cdg operons control cellular turnover of cyclic di-GMP in Acetobacter xylinum genetic organization and occurrence of conserved domains in isoenzymes. J Bacteriol 180(17):4416–4425
Tarutina M, Ryjenkov DA, Gomelsky M (2006) An unorthodox bacteriophytochrome from Rhodobacter sphaeroides involved in turnover of the second messenger c-di-GMP. J Biol Chem 281(46):34751–34758. https://doi.org/10.1074/jbc.M604819200
Taylor BL, Zhulin IB (1999) PAS domains internal sensors of oxygen redox potential and light. Microbiol Mol Biol R 63(2):479–506
Tsukada S, Aono T, Akiba N, Lee KB, Liu CT, Toyazaki H, Oyaizu H (2009) Comparative genome-wide transcriptional profiling of Azorhizobium caulinodans ORS571 grown under free-living and symbiotic conditions. Appl Environ Microbiol 75(15):5037–5046. https://doi.org/10.1128/aem.00398-09
Ueda A, Wood TK (2009) Connecting quorum sensing, c-di-GMP, pel polysaccharide, and biofilm formation in Pseudomonas aeruginosa through tyrosine phosphatase TpbA (PA3885). PLoS Pathog 5(6):e1000483. https://doi.org/10.1371/journal.ppat.1000483
Vicario JC, Dardanelli MS, Giordano W (2015) Swimming and swarming motility properties of peanut-nodulating rhizobia. FEMS Microbiol Lett 362(2):1–6. https://doi.org/10.1093/femsle/fnu038
Weber H, Pesavento C, Possling A, Tischendorf G, Hengge R (2006) Cyclic-di-GMP-mediated signalling within the sigma network of Escherichia coli. Mol Microbiol 62(4):1014–1034. https://doi.org/10.1111/j.1365-2958.2006.05440.x
Xu L, Xin L, Zeng Y, Yam J, Ding Y, Venkataramani P, Cheang QW, Yang X, Tang X, Zhang LH, Chiam KH, Yang L, Liang ZX (2016) A cyclic di-GMP-binding adaptor protein interacts with a chemotaxis methyltransferase to control flagellar motor switching. Sci Signal 9(450):ra 102
Zhou YF, Nan B, Nan J, Ma Q, Panjikar S, Liang YH, Wang Y, Su XD (2008) C4-dicarboxylates sensing mechanism revealed by the crystal structures of DctB sensor domain. J Mol Biol 383(1):49–61. https://doi.org/10.1016/j.jmb.2008.08.010
Acknowledgements
We thank Professors Haiping Cheng and Claudine Elmerich for the helpful and insightful comments on an earlier version of the manuscript. This work was financed by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA23050102),the NSFC-Shandong Joint Fund Key Projects (U1806206), National Natural Science Foundation of China (31870020 and 31570063).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Compliance with ethical standards
This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 769 kb)
Rights and permissions
About this article
Cite this article
Sun, Y., Liu, Y., Liu, X. et al. Azorhizobium caulinodans c-di-GMP phosphodiesterase Chp1 involved in motility, EPS production, and nodulation of the host plant. Appl Microbiol Biotechnol 104, 2715–2729 (2020). https://doi.org/10.1007/s00253-020-10404-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10404-6