Abstract
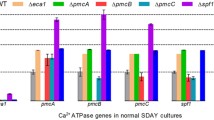
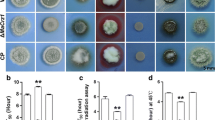
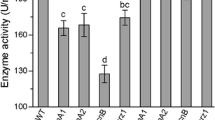
Ca2+/calmodulin-dependent protein kinases (CaMKs) are unique second-messenger molecules that impact almost all cellular processes in eukaryotes. In this study, five genes encoding different CaMKs were characterized in the nematode-trapping fungus Arthrobotrys oligospora. These CaMKs, which were retrieved from the A. oligospora genome according to their orthologs in fungi such as Aspergillus nidulans and Neurospora crassa, were expressed at a low level in vitro during mycelial growth stages. Five deletion mutants corresponding to these CaMKs led to growth defects in different media and increased sensitivity to several environmental stresses, including H2O2, menadione, SDS, and Congo red; they also reduced the ability to produce conidia and traps, thus causing a deficiency in nematicidal ability as well. In addition, the transcriptional levels of several typical sporulation-related genes, such as MedA, VelB, and VeA, were down-regulated in all ΔCaMK mutants compared with the wild-type (WT) strain. Moreover, these mutants exhibited hypersensitivity to heat shock and ultraviolet-radiation stresses compared with the WT strain. These results suggest that the five CaMKs in A. oligospora are involved in regulating multiple cellular processes, such as growth, environmental stress tolerance, conidiation, trap formation, and virulence.






Similar content being viewed by others
References
Braga FR, de Araújo JV (2014) Nematophagous fungi for biological control of gastrointestinal nematodes in domestic animals. Appl Microbiol Biotechnol 98(1):71–82. https://doi.org/10.1007/s00253-013-5366-z
Chen Y, Zhu J, Ying SH, Feng MG (2014) Three mitogen-activated protein kinases required for cell wall integrity contribute greatly to biocontrol potential of a fungal entomopathogen. PLoS One 9(2):e87948. https://doi.org/10.1371/journal.pone.0087948
Christianson TW, Sikorski RS, Dante M, Shero JH, Hieter P (1992) Multifunctional yeast high-copy-number shuttle vectors. Gene 110(1):119–122. https://doi.org/10.1016/0378-1119(92)90454-W
Dayton JS, Means AR (1996) Ca(2+)/calmodulin-dependent kinase is essential for both growth and nuclear division in Aspergillus nidulans. Mol Biol Cell 7(10):1511–1519. https://doi.org/10.1091/mbc.7.10.1511
Farnesi LC, Menna-Barreto RF, Martins AJ, Valle D, Rezende GL (2015) Physical features and chitin content of eggs from the mosquito vectors Aedes aegypti, Anopheles aquasalis and Culex quinquefasciatus: connection with distinct levels of resistance to desiccation. J Insect Physiol 83:43–52. https://doi.org/10.1016/j.jinsphys.2015.10.006
Fujisawa H (2001) Regulation of the activities of multifunctional Ca2+/calmodulin-dependent protein kinases. J Biochem 129(2):193–199. https://doi.org/10.1093/oxfordjournals.jbchem.a002843
Gasteiger E, Hoogland C, Gattiker A, Duvaud SE, Wilkins MR, Appel RD, Bairoch A (2005) Protein identification and analysis tools on the ExPASy server. In: Walker J (ed) The proteomics protocols hand-book. Humana Press, Totowa, pp 571–607. https://doi.org/10.1385/1-59259-890-0:571
Hokyoung S, Myung-Gu K, Min K, Young-Su S, Yun LJ, Ja CG, Jin-Cheol K, Suhn-Kee C, Yin-Won L (2013) AbaA regulates conidiogenesis in the ascomycete fungus Fusarium graminearum. PLoS One 8(9):e72915. https://doi.org/10.1371/journal.pone.0072915
Hook SS, Means AR (2001) Ca(2+)/CaM-dependent kinases: from activation to function. Annu Rev Pharmacol Toxicol 41(1):471–505. https://doi.org/10.1146/annurev.pharmtox.41.1.471
Hunter T (2000) Signaling-2000 and beyond. Cell 100(1):113–127. https://doi.org/10.1016/S0092-8674(00)81688-8
Jiang D, Zhou J, Bai G, Xing X, Tang L, Yang X, Li J, Zhang KQ, Yang JK (2017) Random mutagenesis analysis and identification of a novel C2H2-type transcription factor from the nematode-trapping fungus Arthrobotrys oligospora. Sci Rep 7(1):5640. https://doi.org/10.1038/s41598-017-06075-5
Jones P, Binns D, Chang HY, Fraser M, Li W, Mcanulla C, Mcwilliam H, Maslen J, Mitchell A, Nuka G (2014) InterProScan 5: genome-scale protein function classification. Bioinformatics 30(9):1236–1240. https://doi.org/10.1093/bioinformatics/btu031
Joseph JD, Means AR (2000) Identification and characterization of two Ca2+/CaM-dependent protein kinases required for normal nuclear division in Aspergillus nidulans. J Biol Chem 275(49):38230–38238. https://doi.org/10.1074/jbc.M006422200
Kameshita I, Yamada Y, Nishida T, Sugiyama Y, Sueyoshi N, Watanabe A, Asada Y (2007) Involvement of Ca2+/calmodulin-dependent protein kinases in mycelial growth of the basidiomycetous mushroom, Coprinus cinereus. Biochim Biophy Acta (BBA)-General Subjects 1770(9):1395–1403. https://doi.org/10.1016/j.bbagen.2007.05.008
Kaneko K, Tabuchi M, Sueyoshi N, Ishida A, Utsumi T, Kameshita I (2014) Cellular localization of CoPK12, a Ca(2+)/calmodulin-dependent protein kinase in mushroom Coprinopsis cinerea, is regulated by N-myristoylation. J Biochem 156(1):51–61. https://doi.org/10.1093/jb/mvu018
Krijgsheld P, Bleichrodt R, Veluw GJV, Wang F, Müller WH, Dijksterhuis J, Wösten HAB (2013) Development in Aspergillus. Stud Mycol 74(1):1–29. https://doi.org/10.3114/sim0006
Kumar R, Tamuli R (2014) Calcium/calmodulin-dependent kinases are involved in growth, thermotolerance, oxidative stress survival, and fertility in Neurospora crassa. Arch Microbiol 196(4):295–305. https://doi.org/10.1007/s00203-014-0966-2
Lengeler KB, Davidson RC, D'Souza C, Harashima T, Shen WC, Wang P, Pan X, Waugh M, Heitman J (2000) Signal transduction cascades regulating fungal development and virulence. Microbiol Mol Biol Rev 64(4):746–785. https://doi.org/10.1128/MMBR.64.4.746-785.2000
Li Y, Hyde KD, Jeewon R, Cai L, Vijaykrishna D, Zhang K (2005) Phylogenetics and evolution of nematode-trapping fungi (Orbiliales) estimated from nuclear and protein coding genes. Mycologia 97(5):1034–1046. https://doi.org/10.1080/15572536.2006.11832753
Li F, Wang ZL, Zhang LB, Ying SH, Feng MG (2015) The role of three calcineurin subunits and a related transcription factor (Crz1) in conidiation, multistress tolerance and virulence in Beauveria bassiana. Appl Microbiol Biotechnol 99(2):827–840. https://doi.org/10.1007/s00253-014-6124-6
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Liu J, Tong SM, Qiu L, Ying SH, Feng MG (2017a) Two histidine kinases can sense different stress cues for activation of the MAPK Hog1 in a fungal insect pathogen. Environ Microbiol 19(10):4091–4102. https://doi.org/10.1111/1462-2920.13851
Liu J, Wang ZA, Sun H, Ying S, Feng M (2017b) Characterization of the Hog1 MAPK pathway in the entomopathogenic fungus Beauveria bassiana. Environ Microbiol 19(5):1808–1821. https://doi.org/10.1111/1462-2920.13671
Moosavi MR, Zare R (2012) Fungi as biological control agents of plantparasitic nematodes. In: Mérillon JM, Ramawat KG (eds) Plant defence: biological control, progress in biological control. Springer, New York, pp 67–107. https://doi.org/10.1007/978-94-007-1933-0_4
Nordbring-Hertz B, Jansson H-B, Tunlid A (2011) Nematophagous fungi. In: Encyclopedia of life sciences. John Wiley & Sons, Chichester, pp 1–13. https://doi.org/10.1002/9780470015902.a0000374.pub3
Park G, Colot HV, Collopy PD, Krystofova S, Crew C, Ringelberg C, Litvinkova L, Altamirano L, Li L, Curilla S (2011) High-throughput production of gene replacement mutants in Neurospora crassa. Methods Mol Biol 722:179–189. https://doi.org/10.1007/978-1-61779-040-9_13
Rodriguez-Caban J, Gonzalez-Velazquez W, Perez-Sanchez L, Gonzalez-Mendez R, Rodriguez-Del VN (2011) Calcium/calmodulin kinase1 and its relation to thermotolerance and HSP90 in Sporothrix schenckii: an RNAi and yeast two-hybrid study. BMC Microbiol 11(1):162. https://doi.org/10.1186/1471-2180-11-162
Solomon PS, Rybak K, Trengove RD, Oliver RP (2006) Investigating the role of calcium/calmodulin-dependent protein kinases in Stagonospora nodorum. Mol Microbiol 62(2):367–381. https://doi.org/10.1111/j.1365-2958.2006.05380.x
Staben C, Jensen BC, Singer MJ, Pollock J, Schechtman M, Kinsey JA, Selker E (1989) Use of a bacterial Hygromycin B resistance gene as a dominant selectable marker in Neurospora crassa transformation. Fungal Genet Rep 36:79–81. https://doi.org/10.4148/1941-4765.1519
Su H, Zhao Y, Zhou J, Feng H, Jiang D, Zhang KQ, Yang JK (2017) Trapping devices of nematode-trapping fungi: formation, evolution, and genomic perspectives. Biol Rev 92(1):357–368. https://doi.org/10.1111/brv.12233
Tamuli R, Kumar R, Deka R (2011) Cellular roles of neuronal calcium sensor-1 and calcium/calmodulin-dependent kinases in fungi. J Basic Microbiol 51(2):120–128. https://doi.org/10.1002/jobm.201000184
Tamuli R, Kumar R, Srivastava DA, Deka R (2013) Calcium signaling. In: Kasbekar DP, McCluskey K (eds) Neurospora: genomics and molecular biology. Caister Academic Press, Norfolk, UK, pp 35–57
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739. https://doi.org/10.1093/molbev/msr121
Tao L, Yu JH (2011) AbaA and WetA govern distinct stages of Aspergillus fumigatus development. Microbiology 157(2):313–326. https://doi.org/10.1099/mic.0.044271-0
Tsai PJ, Tu J, Chen TH (2002) Cloning of a Ca(2+)/calmodulin-dependent protein kinase gene from the filamentous fungus Arthrobotrys dactyloides. FEMS Microbiol Lett 212(1):7–13. https://doi.org/10.1016/S0378-1097(02)00648-1
Tunlid A, Ahman J, Oliver RP (1999) Transformation of the nematode-trapping fungus Arthrobotrys oligospora. FEMS Microbiol Lett 173(1):111–116. https://doi.org/10.1111/j.1574-6968.1999.tb13491.x
Vidal-Diez de Ulzurrun G, Hsueh YP (2018) Predator-prey interactions of nematode-trapping fungi and nematodes: both sides of the coin. Appl Microbiol Biotechnol 102(9):3939–3949. https://doi.org/10.1007/s00253-018-8897-5
Winston F, Dollard C, Ricupero-Hovasse SL (1995) Construction of a set of convenient Saccharomyces cerevisiae strains that are isogenic to S288C. Yeast 11(1):53–55. https://doi.org/10.1002/yea.320110107
Xie XQ, Li F, Ying SH, Feng MG (2012) Additive contributions of two manganese-cored superoxide dismutases (MnSODs) to antioxidation, UV tolerance and virulence of Beauveria bassiana. PLoS One 7(1):e30298. https://doi.org/10.1371/journal.pone.0030298
Yang Y, Cheng P, Zhi G, Liu Y (2001) Identification of a calcium/calmodulin-dependent protein kinase that phosphorylates the Neurospora circadian clock protein frequency. J Biol Chem 276(44):41064–41072. https://doi.org/10.1074/jbc.M106905200
Yang JK, Wang L, Ji XL, Feng Y, Li XM, Zou CG, Xu JP, Ren Y, Mi QL, Wu JL, Liu SQ, Liu Y, Huang XW, Niu XM, Li J, Liang LM, Luo YL, Ji KF, Zhou W, Yu ZF, Li GH, Liu YJ, Li L, Qiao M, Feng L, Zhang KQ (2011) Genomic and proteomic analyses of the fungus Arthrobotrys oligospora provide insights into nematode-trap formation. PLoS Pathog 7:e1002179. https://doi.org/10.1371/journal.ppat.1002179
Yang E, Xu L, Yang Y, Zhang X, Xiang M, Wang C, An Z, Liu X (2012) Origin and evolution of carnivorism in the Ascomycota (Fungi). Proc Natl Acad Sci U S A 109(27):10960–10965. https://doi.org/10.1073/pnas.1120915109
Yang JK, Yu Y, Li J, Zhu W, Geng Z, Jiang D, Wang Y, Zhang KQ (2013) Characterization and functional analyses of the chitinase-encoding genes in the nematode-trapping fungus Arthrobotrys oligospora. Arch Microbiol 195(7):453–462. https://doi.org/10.1007/s00203-013-0894-6
Yang X, Ma N, Yang L, Zheng Y, Zhen Z, Li Q, Xie M, Li J, Zhang KQ, Yang JK (2018) Two Rab GTPases play different roles in conidiation, trap formation, stress resistance, and virulence in the nematode-trapping fungus Arthrobotrys oligospora. Appl Microbiol Biotechnol 102(10):4601–4613. https://doi.org/10.1007/s00253-018-8929-1
Yu JH, Keller N (2005) Regulation of secondary metabolism in filamentous fungi. Annu Rev Phytopathol 43(1):437–458. https://doi.org/10.1146/annurev.phyto.43.040204.140214
Zhang L, Yang JK, Niu Q, Zhao X, Ye F, Liang L, Zhang KQ (2008) Investigation on the infection mechanism of the fungus Clonostachys rosea against nematodes using the green fluorescent protein. Appl Microbiol Biotechnol 78(6):983–990. https://doi.org/10.1007/s00253-008-1392-7
Zhao X, Wang Y, Zhao Y, Huang Y, Zhang KQ, Yang JK (2014) Malate synthase gene AoMls in the nematode-trapping fungus Arthrobotrys oligospora contributes to conidiation, trap formation, and pathogenicity. Appl Microbiol Biotechnol 98(6):2555–2563. https://doi.org/10.1007/s00253-013-5432-6
Zhen Z, Xing X, Xie M, Yang L, Yang X, Zheng Y, Chen Y, Ma N, Li Q, Zhang KQ, Yang JK (2018) MAP kinase Slt2 orthologs play similar roles in conidiation, trap formation, and pathogenicity in two nematode-trapping fungi. Fungal Genet Biol 116:42–50. https://doi.org/10.1016/j.fgb.2018.04.011
Acknowledgments
We thank Liwen Bianji, Edanz Group China (www.liwenbianji.cn/ac), for editing the English text of a draft of this manuscript.
Funding
The research described here is jointly supported by the National Natural Science Foundation of China (U1402265, U1502262, and 31272093) and co-supported by Yunnan University’s Research Innovation Fund for Graduate Students (YDY17016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
ESM 1
(PDF 1136 kb)
Rights and permissions
About this article
Cite this article
Zhen, Z., Zhang, G., Yang, L. et al. Characterization and functional analysis of calcium/calmodulin-dependent protein kinases (CaMKs) in the nematode-trapping fungus Arthrobotrys oligospora. Appl Microbiol Biotechnol 103, 819–832 (2019). https://doi.org/10.1007/s00253-018-9504-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9504-5