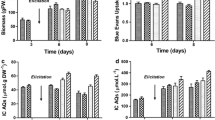
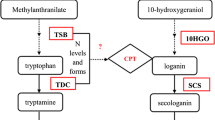
Abstract
The Camptotheca acuminata cell suspension cultures were established to produce the well-known antitumor monoterpene indole alkaloid camptothecin (CAM). Most CAM was present in the broth of the C. acuminata cell suspension cultures. The CAM production was evidenced to be attenuated when the C. acuminata cell suspension cultures were continuously subcultured and grown under identical axenic conditions. A practical cryopreservation and recovery procedure was established to maintain the C. acuminata cell suspension cultures. Biotic and abiotic elicitors were administrated to the C. acuminata cell suspension cultures to restore and enhance CAM production. Of them, sorbitol, a well-known hyperosmotic stressor, was proven to be the most effective elicitor that stimulates a ∼500-fold increase of CAM production. The committed biosynthetic precursors of CAM, tryptamine and secologanin, were feed to the C. acuminata cell suspension cultures and the CAM production is not remarkably increased. However, N 1-acetylkynuramine (NAK), an important metabolite of kynuramine pathway, was isolated and identified from the cell suspension cultures feeding with tryptamine. The present work provides an efficient method to produce CAM and NAK using the C. acuminata cell suspension cultures. The biotransformation of tryptamine to NAK sheds lights on the biosynthetic formation of the pyrroloquinoline moiety of CAM.




Similar content being viewed by others
References
Asolkar RN, Schröder D, Heckmann R, Lang S, Wagner-Döbler I, Laatsch H (2004) Helquinoline, a new tetrahydroquinoline antibiotic from Janibacter limosus Hel 1. J Antibiot 57:17–23
Bhalkar BN, Patil SM, Govindwar SP (2016) Camptothecine production by mixed fermentation of two endophytic fungi from Nothapodytes nimmoniana. Fungal Biol 120:873–883 and references therein
Cai Z, Kastell A, Knorr D (2012) Exudation: an expanding technique for continuous production and release of secondary metabolites from plant cell suspension and hairy root cultures. Plant Cell Rep 31:461–477
Deepthi S, Satheeshkumar K (2016) Enhanced camptothecin production induced by elicitors in the cell suspension cultures of Ophiorrhiza mungos Linn. Plant Cell Tissue Organ Cult 124:483–493 and references therein
Demain AL, Vaishnav P (2011) Natural products for cancer chemotherapy. Microb Biotechnol 4:687–699
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugar and related substances. Anal Chem 68:350–356
Entrena A, Camacho ME, Carrión MD, López-Cara LC, Velasco G, León J, Escames G, Acunña-Castroviejo D, Tapias V, Gallo MA (2005) Kynurenamines as neural nitric oxide synthase inhibitors. J Med Chem 48:8174–8181
Feng X, Zhao P, Hao J, Hu J, Kang D, Wang H (2011) Effects of sorbitol on expression of genes involved in regeneration of upland rice (Oryza sativa L.). Plant Cell Tissue Organ Cult 106:455–463
Fulzele DP, Satdive RK, Pol BB (2001) Growth and production of camptothecin by cell suspension cultures of Nothapodytes foetida. Planta Med 67:150–152
Ge X, Wu J (2005) Tanshinone production and isoprenoid pathways in Salvia miltiorrhiza hairy roots induced by Ag+ and yeast elicitor. Plant Sci 168:487–491
Giri CC, Zaheer M (2016) Chemical elicitors versus secondary metabolite production in vitro using plant cell, tissue and organ cultures: recent trends and a sky eye view appraisal. Plant Cell Tissue Organ Cult 126:1–18
Grout BWW (2007) Cryopreservation of plant cell suspensions. In: Day JG, Stacey GN (eds) Methods in molecular biology, Cryopreservation and Freeze-Drying Protocols, vol 368, 2nd edn. Humana Press Inc., Totowa, NJ, pp 153–161
Hardeland R, Tan DX, Reiter RJ (2009) Kynuramine, metabolites of melatonin and other indoles: the resurrection of an almost forgotten class of biogenic amines. J Pineal Res 47:109–126
Hsiang YH, Hertzberg R, Hecht S, Liu LF (1985) Camptothecin induces protein-linked DNA breaks via mammalian DNA topoisomerase I. J Biol Chem 260:14873–14878
Hutchinson CR, Heckendorf AH, Daddona PE, Hagaman E, Wenkert E (1974) Biosynthesis of camptothecin. I. Definition of the overall pathway assisted by carbon-13 nuclear magnetic resonance analysis. J Am Chem Soc 96:5609–5611
Khalil IM, Barker D, Copp BR (2016) Bioinspired syntheses of the pyridoacridine marine alkaloids demethyldeoxyamphimedine, deoxyamphimedine, and amphimedine. J Org Chem 81:282–289
Kitajima M, Fischer U, Nakamura M, Ohsawa M, Ueno M, Takayama H, Unger M, Stockigt J, Aimi N (1998) Anthraquinones from Ophiorrhiza pumila tissue and cell cultures. Phytochemistry 48:107–111
Liu F, Peng K, Peng Z, Xia S, Xiao L (2010) Establishment of the cell suspension culture system of Camptotheca acuminata. J Hunan Agr Univ 36:528–530
López-Meyer M, Nesler CL, McKnight TD (1994) Sites of accumulation of the antitumor alkaloid camptothecin in Camptotheca acuminata. Planta Med 60:558–560
Lorence A, Nessler CL (2004) Camptothecin, over four decades of surprising findings. Phytochemistry 65:2735–2749
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Pu X, Qu X, Chen F, Bao J, Zhang G, Luo Y (2013) Camptothecin-producing endophytic fungus Trichoderma atroviride LY357: isolation, identification, and fermentation conditions optimization for camptothecin production. Appl Microbiol Biotechnol 97:9365–9375 and references therein
Pu X, Chen F, Yang Y, Qu X, Zhang G, Luo Y (2015) Isolation and characterization of Paenibacillus polymyxa LY214, a camptothecin-producing endophytic bacterium from Camptotheca acuminata. J Ind Microbiol Biotechnol 42:1197–1202 and references therein
Qin H, Chamlong P (2007) Camptotheca acuminata Decaisne. Flora of China 13:300–301
Qu X, Pu X, Chen F, Yang Y, Yang L, Zhang G, Luo Y (2015) Molecular cloning, heterologous expression, and functional characterization of an NADPH-cytochrome P450 reductase gene from Camptotheca acuminata, a camptothecin-producing plant. PLoS One 10:e0135397
Ressmeyer AR, Mayo JC, Zelosko V, Sáinz RM, Tan DX, Burkhard P, Antolín I, Zsizsik BK, Reiter RJ, Hardeland R (2003) Antioxidant properties of the melatonin metabolite N1-acetyl-5-methoxykynuramine (AMK): scavenging of free radicals and prevention of protein destruction. Redox Rep 8:205–213
Sakato K, Tanaka H, Mukai N, Misawa M (1974) Isolation and identification of camptothecin from cells of Camptotheca acuminata suspension cultures. Agr Biol Chem 38:217–218
Shaanker RU, Ramesha BT, Ravikanth G, Gunaga R, Vasudeva R, Ganeshaiah KN (2008) Chemical profiling of Nothapodytes nimmoniana for camptothecin, an important anticancer alkaloid: towards the development of a sustainable production system. In: Ramawat KG, Mérillon JM (eds) Bioactive molecules and medicinal plants. Springer-Verlag, Berlin and Heidelberg, pp 197–213
Sheriha GM, Rapoport H (1976) Biosynthesis of Camptotheca acuminata alkaloids. Phytochemistry 15:505–508
Sirikantaramas S, Asano T, Sudo H, Yamazaki M, Saito K (2007) Camptothecin: therapeutic potential and biotechnology. Curr Pharm Biotechnol 8:196–202
Skyler D, Heathcock CH (2001) A simple biomimetic synthesis of styelsamine B. Org Lett 3:4323–4324
Thomas CJ, Rahier NJ, Hecht SM (2004) Camptothecin: current perspectives. Bioorg Med Chem 12:1585–1604
Vasanthakumari MM, Jadhav SS, Sachin N, Vinod G, Shweta S, Manjunatha BL, Kumara PM, Ravikanth G, Nataraja KN, Shaanker RU (2015) Restoration of camptothecine production in attenuated endophytic fungus on re-inoculation into host plant and treatment with DNA methyltransferase inhibitor. World J Microbiol Biotechnol 31:1629–1639 and references therein
Venugopalan A, Srivastava S (2015) Endophytes as in vitro production platforms of high value plant secondary metabolites. Biotechnol Adv 33:873–887 and references therein
Venugopalan A, Potunuru UR, Dixit M, Srivastava S (2016) Effect of fermentation parameters, elicitors and precursors on camptothecin production from the endophyte Fusarium solani. Bioresource Technol 206:104–111 and references therein
Wall ME, Wani MC, Cook CE, Palmer KH, McPhail AT, Sim GA (1966) Plant antitumor agents. I. The isolation and structure of camptothecin, a novel alkaloidal leukemia and tumor inhibitor from Camptotheca acuminata. J Am Chem Soc 88:3887–3890
Wang JW, Wu JY (2013) Effective elicitors and process strategies for enhancement of secondary metabolite production in hairy root cultures. Adv Biochem Engin/Biotechnol 2013:183
Yin Z, Chen L, Zhao B, Zhu Y, Wang Q (2012) Cryopreservation of embryogenic cell suspensions by encapsulation-vitrification and encapsulation-dehydation. In: Loyola-Vargas VM, Ochoa-Alejo N (eds) Methods in molecular biology, Plant Cell Culture Protocols, vol 877. Springer Science + Business Media, LLC, pp 81–93
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported in part by the KSCX2-EW-Q-6 and ZHTS-003 projects from the Chinese Academy of Sciences, the Applied and Basic Research Program of Sichuan Province (2015JY0058), and the 21172216 project from the National Natural Science Foundation of China.
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Yun Yang and Xiang Pu authors contributed equally to this work.
Electronic supplementary material
ESM 1
(PDF 1159 kb).
Rights and permissions
About this article
Cite this article
Yang, Y., Pu, X., Qu, X. et al. Enhanced production of camptothecin and biological preparation of N 1-acetylkynuramine in Camptotheca acuminata cell suspension cultures. Appl Microbiol Biotechnol 101, 4053–4062 (2017). https://doi.org/10.1007/s00253-017-8153-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8153-4