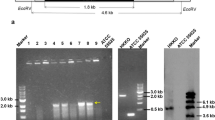
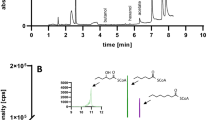
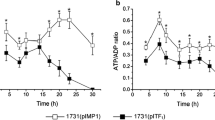
Abstract
The industrial Gram-positive anaerobe Clostridium acetobutylicum is a valued acetone, butanol, and ethanol (ABE) solvent producer that is able to utilize a vast array of carbon sources in fermentation. When glucose is present in the growth medium, however, C. acetobutylicum, like many Gram-positive organisms, exhibits biphasic growth characteristics in which glucose is used preferentially over secondary carbon sources, a phenomenon known as carbon catabolite repression (CCR). The secondary carbon source is only utilized when the supply of glucose is exhausted, resulting in inefficient use of complex carbon sources. As biofuel production is sought from cheap feedstock, attention has turned to lignocellulosic biomass. Growth of C. acetobutylicum on lignocellulose, however, can be limited by CCR. Here, we present a method to relieve the inhibitory effect of CCR and allow simultaneous utilization of the lignocellulosic sugars of glucose and xylose by C. acetobutylicum. First, we utilized an in vivo gene reporter assay to demonstrate that an identified 14-nucleotide catabolite responsive element (CRE) sequence was sufficient to introduce CCR-mediated transcriptional inhibition, while subsequent mutation of the CRE sequence relieved the inhibitory effect. Next, we demonstrated that C. acetobutylicum harboring a CRE-less plasmid-borne xylose and pentose phosphate pathway operon afforded a 7.5-fold increase in xylose utilization in the presence of glucose as compared to a wild-type CRE plasmid-borne operon, effectively overcoming native CCR effects. The methodology presented here should translate to other members of Clostridium that exhibit CCR to enable simultaneous utilization of a vast array of carbon sources.



Similar content being viewed by others
References
Abdou L, Boileau C, de Philip P, Pagès S, Fiérobe H-P, Tardif C (2008) Transcriptional regulation of the Clostridium cellulolyticum cip-cel operon: a complex mechanism involving a catabolite-responsive element. J Bacteriol 190:1499–1506. doi:10.1128/jb.01160-07
Al-Hinai MA, Fast AG, Papoutsakis ET (2012) Novel system for efficient isolation of Clostridium double-crossover allelic exchange mutants enabling markerless chromosomal gene deletions and DNA integration. Appl Environ Microbiol 78:8112–8121. doi:10.1128/AEM.02214-12
Baer SH, Blaschek HP, Smith TL (1987) Effect of butanol challenge and temperature on lipid composition and membrane fluidity of butanol-tolerant Clostridium acetobutylicum. Appl Environ Microbiol 53:2854–2861
Bowles LK, Ellefson WL (1985) Effects of butanol on Clostridium acetobutylicum. Appl Environ Microbiol 50:1165–1170
Choi KY, Zalkin H (1992) Structural characterization and corepressor binding of the Escherichia coli purine repressor. J Bacteriol 174:6207–6214
Egeter O, Brückner R (1996) Catabolite repression mediated by the catabolite control protein CcpA in Staphylococcus xylosus. Mol Microbiol 21:739–749. doi:10.1046/j.1365-2958.1996.301398.x
Eiteman MA, Lee SA, Altman E (2008) A co-fermentation strategy to consume sugar mixtures effectively. J Biol Eng 2:3–3. doi:10.1186/1754-1611-2-3
Garcia Hernan G, Lee Heun J, Boedicker James Q, Phillips R (2011) Comparison and calibration of different reporters for quantitative analysis of gene expression. Biophys J 101:535–544. doi:10.1016/j.bpj.2011.06.026
Gorke B, Stulke J (2008) Carbon catabolite repression in bacteria: many ways to make the most out of nutrients. Nat Rev Microbiol 6:613–624. doi:10.1038/nrmicro1932
Grimmler C, Held C, Liebl W, Ehrenreich A (2010) Transcriptional analysis of catabolite repression in Clostridium acetobutylicum growing on mixtures of d-glucose and d-xylose. J Biotechnol 150:315–323. doi:10.1016/j.jbiotec.2010.09.938
Grundy FJ, Waters DA, Allen SH, Henkin TM (1993) Regulation of the Bacillus subtilis acetate kinase gene by CcpA. J Bacteriol 175:7348–7355
Gu Y, Li J, Zhang L, Chen J, Niu L, Yang Y, Yang S, Jiang W (2009) Improvement of xylose utilization in Clostridium acetobutylicum via expression of the talA gene encoding transaldolase from Escherichia coli. J Biotechnol 143:284–287. doi:10.1016/j.jbiotec.2009.08.009
Hu S, Zheng H, Gu Y, Zhao J, Zhang W, Yang Y, Wang S, Zhao G, Yang S, Jiang W (2011) Comparative genomic and transcriptomic analysis revealed genetic characteristics related to solvent formation and xylose utilization in Clostridium acetobutylicum EA 2018. BMC Genomics 12 doi:10.1186/1471-2164-12-93
Hueck CJ, Hillen W (1995) Catabolite repression in Bacillus subtilis: a global regulatory mechanism for the gram-positive bacteria? Mol Microbiol 15:395–401. doi:10.1111/j.1365-2958.1995.tb02252.x
Hueck CJ, Hillen W, Saier MH Jr (1994) Analysis of a cis-active sequence mediating catabolite repression in gram-positive bacteria. Res Microbiol 145:503–518. doi:10.1016/0923-2508(94)90028-
Jang Y-S, Malaviya A, Cho C, Lee J, Lee SY (2012) Butanol production from renewable biomass by clostridia. Bioresour Technol 123:653–663. doi:10.1016/j.biortech.2012.07.104
Jiang L, Cai J, Wang J, Liang S, Xu Z, Yang S-T (2010) Phosphoenolpyruvate-dependent phosphorylation of sucrose by Clostridium tyrobutyricum ZJU 8235: evidence for the phosphotransferase transport system. Bioresour Technol 101:304–309. doi:10.1016/j.biortech.2009.08.024
Jin L, Zhang H, Chen L, Yang C, Yang S, Jiang W, Gu Y (2014) Combined overexpression of genes involved in pentose phosphate pathway enables enhanced d-xylose utilization by Clostridium acetobutylicum. J Biotechnol 173:7–9. doi:10.1016/j.jbiotec.2014.01.002
Kim J-H, Chambliss GH (1997) Contacts between Bacillus subtilis catabolite regulatory protein CcpA and amyO target site. Nucleic Acids Res 25:3490–3496. doi:10.1093/nar/25.17.3490
Lewis M, Chang G, Horton NC, Kercher MA, Pace HC, Schumacher MA, Brennan RG, Lu P (1996) Crystal structure of the lactose operon repressor and its complexes with DNA and inducer. Science 271:1247–1254. doi:10.1126/science.271.5253.1247
Li Z, Xiao H, Jiang W, Jiang Y, Yang S (2013) Improvement of solvent production from xylose mother liquor by engineering the xylose metabolic pathway in Clostridium acetobutylicum EA 2018. Appl Biochem Biotechnol 171:555–568. doi:10.1007/s12010-013-0414-9
Loll B, Kowalczyk M, Alings C, Chieduch A, Bardowski J, Saenger W, Biesiadka J (2007) Structure of the transcription regulator CcpA from Lactococcus lactis. Acta Crystallogr Sect D: Biol Crystallogr 63:431–436. doi:10.1107/S0907444907000546
Lütke-Eversloh T, Bahl H (2011) Metabolic engineering of Clostridium acetobutylicum: recent advances to improve butanol production. Curr Opin Biotechnol 22:634–647. doi:10.1016/j.copbio.2011.01.011
Marasco R, Muscariello L, Rigano M, Sacco M (2002) Mutational analysis of the bglH catabolite-responsive element (cre) in Lactobacillus plantarum. FEMS Microbiol Lett 208:143–146. doi:10.1111/j.1574-6968.2002.tb11074.x
Marciniak B, Pabijaniak M, de Jong A, Duhring R, Seidel G, Hillen W, Kuipers O (2012) High- and low-affinity cre boxes for CcpA binding in Bacillus subtilis revealed by genome-wide analysis. BMC Genomics 13:401. doi:10.1186/1471-2164-13-401
Mermelstein L, Papoutsakis E (1993) In vivo methylation in Escherichia coli by the Bacillus subtilis phage phi 3T I methyltransferase to protect plasmids from restriction upon transformation of Clostridium acetobutylicum ATCC 824. Appl Environ Microbiol 59:1077–1081
Miwa Y, Nakata A, Ogiwara A, Yamamoto M, Fujita Y (2000) Evaluation and characterization of catabolite-responsive elements (cre) of Bacillus subtilis. Nucleic Acids Res 28:1206–1210. doi:10.1093/nar/28.5.1206
Noguchi T, Tashiro Y, Yoshida T, Zheng J, Sakai K, Sonomoto K (2013) Efficient butanol production without carbon catabolite repression from mixed sugars with Clostridium saccharoperbutylacetonicum N1-4. J Biosci Bioeng 116:716–721. doi:10.1016/j.jbiosc.2013.05.030
Ohta K, Beall DS, Mejia JP, Shanmugam KT, Ingram LO (1991) Metabolic engineering of Klebsiella oxytoca M5A1 for ethanol production from xylose and glucose. Appl Environ Microbiol 57:2810–2815
Ounine K, Petitdemange H, Raval G, Gay R (1985) Regulation and butanol inhibition of d-xylose and d-glucose uptake in Clostridium acetobutylicum. Appl Environ Microbiol 49:874–878
Paredes CJ, Rigoutsos I, Papoutsakis ET (2004) Transcriptional organization of the Clostridium acetobutylicum genome. Nucleic Acids Res 32:1973–1981. doi:10.1093/nar/gkh509
Paulsen IT, Chauvaux S, Choi P, Saier MH (1998) Characterization of glucose-specific catabolite repression-resistant mutants of Bacillus subtilis: identification of a novel hexose:H+ symporter. J Bacteriol 180:498–504
Pyne ME, Bruder M, Moo-Young M, Chung DA, Chou CP (2014a) Technical guide for genetic advancement of underdeveloped and intractable Clostridium. Biotechnol Adv 32:623–641. doi:10.1016/j.biotechadv.2014.04.003
Pyne ME, Moo-Young M, Chung DA, Chou CP (2013) Development of an electrotransformation protocol for genetic manipulation of Clostridium pasteurianum. Biotechnol Biofuels 6:50
Pyne ME, Moo-Young M, Chung DA, Chou CP (2014b) Expansion of the genetic toolkit for metabolic engineering of Clostridium pasteurianum: chromosomal gene disruption of the endogenous CpaAI restriction enzyme. Biotechnol Biofuels 7:163
Ren C, Gu Y, Hu S, Wu Y, Wang P, Yang Y, Yang C, Yang S, Jiang W (2010) Identification and inactivation of pleiotropic regulator CcpA to eliminate glucose repression of xylose utilization in Clostridium acetobutylicum. Metab Eng 12:446–454. doi:10.1016/j.ymben.2010.05.002
Ren C, Gu Y, Wu Y, Zhang W, Yang C, Yang S, Jiang W (2012) Pleiotropic functions of catabolite control protein CcpA in butanol-producing Clostridium acetobutylicum. BMC Genomics 13:349. doi:10.1186/1471-2164-13-349
Rodionov DA, Mironov AA, Gelfand MS (2001) Transcriptional regulation of pentose utilisation systems in the Bacillus/Clostridium group of bacteria. FEMS Microbiol Lett 205:305–314. doi:10.1111/j.1574-6968.2001.tb10965.x
Sadler JR, Sasmor H, Betz JL (1983) A perfectly symmetric lac operator binds the lac repressor very tightly. Proc Natl Acad Sci U S A 80:6785–6789
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning, vol 2. Cold spring harbor laboratory press, New York
Schumacher M, Allen G, Diel M, Seidel G, Hillen W, Brennan R (2004) Structural basis for allosteric control of the transcription regulator CcpA by the phosphoprotein HPr-Ser46-P. Cell 118:731–741
Schumacher MA, Choi KY, Zalkin H, Brennan RG (1994) Crystal structure of LacI member, PurR, bound to DNA: minor groove binding by alpha helices. Science 266:763–770. doi:10.1126/science.7973627
Schumacher MA, Seidel G, Hillen W, Brennan RG (2006) Phosphoprotein Crh-Ser46-P displays altered binding to CcpA to effect carbon catabolite regulation. J Biol Chem 281:6793–6800. doi:10.1074/jbc.M509977200
Schumacher MA, Seidel G, Hillen W, Brennan RG (2007) Structural mechanism for the fine-tuning of CcpA function by the small molecule effectors glucose 6-phosphate and fructose 1,6-bisphosphate. J Mol Biol 368:1042–1050. doi:10.1016/j.jmb.2007.02.054
Schumacher MA, Sprehe M, Bartholomae M, Hillen W, Brennan RG (2011) Structures of carbon catabolite protein A-(HPr-Ser46-P) bound to diverse catabolite response element sites reveal the basis for high-affinity binding to degenerate DNA operators. Nucleic Acids Res 39:2931–2942. doi:10.1093/nar/gkq1177
Swint-Kruse L, Matthews KS (2009) Allostery in the LacI/GalR family: variations on a theme. Curr Opin Microbiol 12:129–137. doi:10.1016/j.mib.2009.01.009
Tangney M, Galinier A, Deutscher J, Mitchell WJ (2003) Analysis of the elements of catabolite repression in Clostridium acetobutylicum ATCC 824. J Mol Microbiol Biotechnol 6:6–11. doi:10.1159/000073403
Tobisch S, Zühlke D, Bernhardt J, Stülke J, Hecker M (1999) Role of CcpA in regulation of the central pathways of carbon catabolism in Bacillus subtilis. J Bacteriol 181:6996–7004
Tojo S, Satomura T, Morisaki K, Deutscher J, Hirooka K, Fujita Y (2005) Elaborate transcription regulation of the Bacillus subtilis ilv-leu operon involved in the biosynthesis of branched-chain amino acids through global regulators of CcpA, CodY and TnrA. Mol Microbiol 56:1560–1573
Tracy BP, Jones SW, Fast AG, Indurthi DC, Papoutsakis ET (2012) Clostridia: the importance of their exceptional substrate and metabolite diversity for biofuel and biorefinery applications. Curr Opin Biotechnol 23:364–381. doi:10.1016/j.copbio.2011.10.008
Tummala SB, Welker NE, Papoutsakis ET (1999) Development and characterization of a gene expression reporter system for Clostridium acetobutylicum ATCC 824. Appl Environ Microbiol 65:3793–3799
Varga J, Stirewalt VL, Melville SB (2004) The CcpA protein is necessary for efficient sporulation and enterotoxin gene (cpe) regulation in Clostridium perfringens. J Bacteriol 186:5221–5229. doi:10.1128/jb.186.16.5221-5229.2004
Warner JB, Lolkema JS (2003) CcpA-dependent carbon catabolite repression in bacteria. Microbiol Mol Biol Rev 67:475–490. doi:10.1128/mmbr.67.4.475-490.2003
Weickert MJ, Chambliss GH (1990) Site-directed mutagenesis of a catabolite repression operator sequence in Bacillus subtilis. Proc Natl Acad Sci U S A 87:6238–6242. doi:10.1073/pnas.87.16.6238
Wu Y, Yang Y, Ren C, Yang C, Yang S, Gu Y, Jiang W (2015) Molecular modulation of pleiotropic regulator CcpA for glucose and xylose coutilization by solvent-producing Clostridium acetobutylicum. Metab Eng 28:169–179. doi:10.1016/j.ymben.2015.01.006
Xiao H, Gu Y, Ning Y, Yang Y, Mitchell WJ, Jiang W, Yang S (2011) Confirmation and elimination of xylose metabolism bottlenecks in glucose phosphoenolpyruvate-dependent phosphotransferase system-deficient Clostridium acetobutylicum for simultaneous utilization of glucose, xylose, and arabinose. Appl Environ Microbiol 77:7886–7895. doi:10.1128/aem.00644-11
Xiao H, Li Z, Jiang Y, Yang Y, Jiang W, Gu Y, Yang S (2012) Metabolic engineering of d-xylose pathway in Clostridium beijerinckii to optimize solvent production from xylose mother liquid. Metab Eng 14:569–578. doi:10.1016/j.ymben.2012.05.003
Yu EK, Saddler JN (1983) Fed-batch approach to production of 2,3-butanediol by Klebsiella pneumoniae grown on high substrate concentrations. Appl Environ Microbiol 46:630–635
Yu Y, Tangney M, Aass HC, Mitchell WJ (2007) Analysis of the mechanism and regulation of lactose transport and metabolism in Clostridium acetobutylicum ATCC 824. Appl Environ Microbiol 73:1842–1850. doi:10.1128/aem.02082-06
Zhang Y-HP, Lynd LR (2005) Regulation of cellulase synthesis in batch and continuous cultures of Clostridium thermocellum. J Bacteriol 187:99–106. doi:10.1128/jb.187.1.99-106.2005
Acknowledgments
This work was supported in part by the Natural Sciences and Engineering Research Council of Canada (NSERC; grant number STPGP 430106–12) and the Canada Research Chairs (CRC; grant number 950–211471) programs.
Conflict of interest
DAC is a founder and employee of Neemo Inc., at which MB has also been employed. Neemo Inc. has a financial interest in the production of biofuels using clostridial microorganisms. The remaining authors declare no competing interests.
Authors’ contributions
MB helped conceive the study, participated in its design and coordination, carried out the experiments, and drafted the manuscript. MMY participated in the study design and coordination. DAC and CPC helped conceive the study, participated in its design and coordination, and helped to draft the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Bruder, M., Moo-Young, M., Chung, D.A. et al. Elimination of carbon catabolite repression in Clostridium acetobutylicum—a journey toward simultaneous use of xylose and glucose. Appl Microbiol Biotechnol 99, 7579–7588 (2015). https://doi.org/10.1007/s00253-015-6611-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6611-4