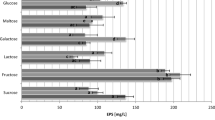
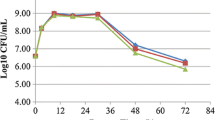
Abstract
Lactic acid bacteria are extensively used in food technology and for the production of various compounds, but they are fastidious in nutrient requirements. In order to elucidate the role of each component precisely, defined multicomponent media are required. This study focuses on determining nutrient auxotrophies and minimizing media components (amino acids, vitamins, metal ions, buffers and additional compounds) for the cultivation of Lactococcus lactis subsp. lactis IL1403, using microtitre plates and test tubes. It was shown that glutamine and asparagine were the most important media components for achieving higher biomass yields while the branched-chain amino acids were necessary to increase specific growth rate. The amino acid and glucose ratio was reduced to achieve minimal residual concentration of amino acids in the medium after the growth of cells, whereas the specific growth rate and biomass yield of cells were not considerably affected. As the percentage of each consumed amino acid compared to initial amount is larger than measurement error, these optimized media are important for achieving more precise data about amino acid utilization and metabolism.





Similar content being viewed by others
References
Bolotin A, Wincker P, Mauger S, Jaillon O, Malarme K, Weissenbach J, Ehrlich SD, Sorokin A (2001) The complete genome sequence of the lactic acid bacterium Lactococcus lactis ssp. lactis IL1403. Genome Res 11:731–753
Chambellon E, Rijnen L, Lorquet F, Gitton C, van Hylckama Vlieg JE, Wouters JA, Yvon M (2009) The D-2-hydroxyacid dehydrogenase incorrectly annotated PanE is the sole reduction system for branched-chain 2-keto acids in Lactococcus lactis. J Bacteriol 191(3):873–881. doi:10.1128/JB.01114-08
Cocaign-Bousquet M, Garrigues C, Novak L, Lindley ND, Loubiere P (1995) Rational development of a simple synthetic medium for the sustained growth of Lactococcus lactis. J Appl Bacteriol 79:108–116. doi:10.1111/j.1365-2672.1995.tb03131.x
De Man JC, Rogosa M, Sharpe ME (1960) A medium for the cultivation of lactobacilli. J Appl Bacteriol 23(1):130–135. doi:10.1111/j.1365-2672.1960.tb00188.x
Den Hengst CD, Groeneveld M, Kuipers OP, Kok J (2006) Identification and functional characterization of the Lactococcus lactis CodY-regulated branched-chain amino acid permease BcaP (CtrA). J Bacteriol 188(9):3280–3289. doi:10.1128/JB.188.9.3280-3289.2006
Duetz WA (2007) Microtiter plates as mini-bioreactors: miniaturization of fermentation methods. Trends Microbiol 15(10):469–475. doi:10.1016/j.tim.2007.09.004
Fernández M, Zúñiga M (2006) Amino acid catabolic pathways of lactic acid bacteria. Crit Rev Microbiol 32(3):155–183
Goel A, Santos F, de Vos WM, Teusink B, Molenaar D (2012) Standardized assay medium to measure Lactococcus lactis enzyme activities while mimicking intracellular conditions. Appl Environ Microbiol 78(1):134–143. doi:10.1128/AEM.05276-11
Hébert EM, Raya RR, de Giori GS (2004) Nutritional requirements of Lactobacillus delbrueckii subsp. lactis in a chemically defined medium. Curr Microbiol 49(5):341–345. doi:10.1007/s00284-004-4357-9
Jensen PR, Hammer K (1993) Minimal requirements for exponential growth of Lactococcus lactis. Appl Environ Microbiol 59(12):4363–4366
Jensen NBS, Melchiorsen CR, Jokumsen KV, Villadsen J (2001) Metabolic behavior of Lactococcus lactis MG1363 in microaerobic continuous cultivation at a low dilution rate. Appl Environ Microbiol 67(6):2677–2682. doi:10.1128/AEM.67.6.2677-2682.2001
Kensy F, Engelbrecht C, Büchs J (2009) Scale-up from microtiter plate to laboratory fermenter: evaluation by online monitoring techniques of growth and protein expression in Escherichia coli and Hansenula polymorpha fermentations. Microb Cell Factories 8:68. doi:10.1186/1475-2859-8-68
Kim YJ, Eom HJ, Seo EY, Lee DY, Kim JH, Han NS (2012) Development of a chemically defined minimal medium for the exponential growth of Leuconostoc mesenteroides ATCC8293. J Microbiol Biotechnol 22(11):1518–1522
Lahtvee PJ, Adamberg K, Arike L, Nahku R, Aller K, Vilu R (2011) Multi-omics approach to study the growth efficiency and amino acid metabolism in Lactococcus lactis at various specific growth rates. Microb Cell Factories 10:12. doi:10.1186/1475-2859-10-12
Lapujade P, Cocaign-Bousquet M, Loubiere P (1998) Glutamate biosynthesis in Lactococcus lactis subsp. lactis NCDO 2118. Appl Environ Microbiol 64(7):2485–2489
Larsen R, Kloosterman TG, Kok J, Kuipers OP (2006) GlnR-mediated regulation of nitrogen metabolism in Lactococcus lactis. J Bacteriol 188(13):4978–4982. doi:10.1128/JB.00025-06
LeBlanc JG, Laiño JE, del Valle MJ, Vannini V, van Sinderen D, Taranto MP, de Valdez GF, de Giori GS, Sesma F (2011) B-Group vitamin production by lactic acid bacteria—current knowledge and potential applications. J Appl Microbiol 111(6):1297–1309. doi:10.1111/j.1365-2672.2011.05157.x
Loubiere P, Cocaign-Bousquet M, Matos J, Goma G, Lindley ND (1997) Influence of end-products inhibition and nutrient limitations on the growth of Lactococcus lactis subsp. lactis. J Appl Microbiol 82:95–100. doi:10.1111/j.1365-2672.1997.tb03302.x
Magnani D, Barré O, Gerber SD, Solioz M (2008) Characterization of the CopR regulon of Lactococcus lactis IL1403. J Bacteriol 190(2):536–545. doi:10.1128/JB.01481-07
Nierop Groot MN, de Bont JAM (1999) Involvement of manganese in conversion of phenylalanine to benzaldehyde by lactic acid bacteria. Appl Environ Microbiol 65(12):5590–5593
Nomura M, Nakajima I, Fujita Y, Kobayashi M, Kimoto H, Suzuki I, Aso H (1999) Lactococcus lactis contains only one glutamate decarboxylase gene. Microbiology 145:1375–1380. doi:10.1099/13500872-145-6-1375
Nordkvist M, Jensen NBS, Villadsen J (2003) Glucose metabolism in Lactococcus lactis MG1363 under different aeration conditions: requirement of acetate to sustain growth under microaerobic conditions. Appl Environ Microbiol 69(6):3462–3468. doi:10.1128/AEM.69.6.3462-3468.2003
Novak L, Loubiere P (2000) The metabolic network of Lactococcus lactis: distribution of 14C-labeled substrates between catabolic and anabolic pathways. J Bacteriol 182(4):1136–1143. doi:10.1128/JB.182.4.1136-1143.2000
Ogawa H, Gomi T, Fujioka M (2000) Serine hydroxymethyltransferase and threonine aldolase: are they identical? Int J Biochem Cell Biol 32(3):289–302. doi:10.1016/S1357-2725(99)00113-2
Price CE, Zeyniyev A, Kuipers OP, Kok J (2012) From meadows to milk to mucosa—adaptation of Streptococcus and Lactococcus species to their nutritional environments. FEMS Microbiol Rev 36(5):949–971. doi:10.1111/j.1574-6976.2011.00323.x
Santos F, Teusink B, Molenaar D, van Heck M, Wels M, Sieuwerts S, de Vos WM, Hugenholtz J (2009) Effect of amino acid availability on vitamin B12 production in Lactobacillus reuteri. Appl Environ Microbiol 75(12):3930–3936. doi:10.1128/AEM.02487-08
Sleator RD, Gahan CGM, Hill C (2001) Mutations in the Listerial proB gene leading to proline overproduction: effects on salt tolerance and murine infection. Appl Environ Microbiol 67(10):4560–4565. doi:10.1128/AEM.67.10.4560-4565.2001
Sperandio B, Polard P, Ehrlich DS, Renault P, Guédon E (2005) Sulfur amino acid metabolism and its control in Lactococcus lactis IL1403. J Bacteriol 187(11):3762–3778. doi:10.1128/JB.187.11.3762-3778.2005
Sybesma W, Starrenburg M, Tijsseling L, Hoefnagel MH, Hugenholtz J (2003) Effects of cultivation conditions on folate production by lactic acid bacteria. Appl Environ Microbiol 69(8):4542–4548. doi:10.1128/AEM.69.8.4542-4548.2003
Van Niel EWJ, Hahn-Hägerdal B (1999) Nutrient requirements of lactococci in defined growth media. Appl Microbiol Biotechnol 52:617–627. doi:10.1007/s002530051569
Wegkamp A, van Oorschot W, de Vos WM, Smid EJ (2007) Characterization of the role of para-aminobenzoic acid biosynthesis in folate production by Lactococcus lactis. Appl Environ Microbiol 73(8):2673–2681. doi:10.1128/AEM.02174-06
Wegkamp A, Teusink B, de Vos WM, Smid EJ (2009) Development of a minimal growth medium for Lactobacillus plantarum. Lett Appl Microbiol 50(1):57–64. doi:10.1111/j.1472-765X.2009.02752.x
Zhang G, Mills DA, Block DE (2009) Development of chemically defined media supporting high cell density growth of lactococci, enterococci, and streptococci. Appl Environ Microbiol 75(4):1080–1087. doi:10.1128/AEM.01416-08
Acknowledgments
The financial support for this research was provided by the Enterprise Estonia project EU29994, by Estonian Science Foundation through the grant G9192, and Ministry of Education, Estonia, through the grant SF0140090s08.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(XLSX 141 kb)
Rights and permissions
About this article
Cite this article
Aller, K., Adamberg, K., Timarova, V. et al. Nutritional requirements and media development for Lactococcus lactis IL1403. Appl Microbiol Biotechnol 98, 5871–5881 (2014). https://doi.org/10.1007/s00253-014-5641-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-5641-7