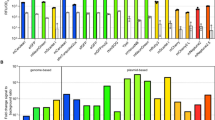
Abstract
Fluorescent reporter genes are valuable tools for real-time monitoring of gene expression in living cells. In this study we describe the construction of novel promoter-probe vectors containing a synthetic mCherry fluorescent protein gene, codon-optimized for lactic acid bacteria, divergently linked, or not, to a gene encoding the S65T and F64L variant of the green fluorescent protein. The utility of the transcriptional fusion vectors was demonstrated by the cloning of a single or two divergent promoter regions and by the quantitative evaluation of fluorescence during growth of Lactococcus lactis, Enterococcus faecalis, and Escherichia coli.


Similar content being viewed by others
References
Baird GS, Zacharias DA, Tsien RY (2000) Biochemistry, mutagenesis, and oligomerization of DsRed, a red fluorescent protein from coral. Proc Natl Acad Sci U S A 97:11984–11989
Bricker AL, Camilli A (1999) Transformation of a type 4 encapsulated strain of Streptococcus pneumoniae. FEMS Microbiol Lett 172:131–135
Chudakov DM, Lukyanov S, Lukyanov KA (2005) Fluorescent proteins as a toolkit for in vivo imaging. Trends Biotechnol 23:605–613
De la Plaza M, Peláez C, Requena T (2009) Regulation of alpha-ketoisovalerate decarboxylase expression in Lactococcus lactis IFPL730. J Mol Microbiol Biotechnol 17:96–100
Fernández de Palencia P, Nieto C, Acebo P, Espinosa M, López P (2000) Expression of green fluorescent protein in Lactococcus lactis. FEMS Microbiol Lett 183:229–234
Fernández de Palencia P, López P, Corbí AL, Peláez C, Requena T (2008) Probiotic strains: survival under simulated gastrointestinal conditions, in vitro adhesion to Caco-2 cells and effect on cytokine secretion. Eur Food Res Technol 227:1475–1484
Fiedler S, Wirth R (1991) Transformation of Enterococcus faecalis and Enterococcus faecium by electroporation. In: Dunny GM, Clearly PP, McKay LL (eds) Genetics and molecular biology of streptococci, lactococci, and enterococci. ASM, Washington, DC, p 301
Frommer WB, Davidson MW, Campbell RE (2009) Genetically encoded biosensors based on engineered fluorescent proteins. Chem Soc Rev 38:2833–2841
Fukuda H, Arai M, Kuwajima K (2000) Folding of green fluorescent protein and the cycle3 mutant. Biochemistry 39:12025–12032
Garai-Ibabe G, Dueñas MT, Irastorza A, Sierra-Filardi E, Werning ML, López P, Corbí AL, Fernández de Palencia P (2010) Naturally occurring 2-substituted (1,3)-β-d-glucan producing Lactobacillus suebicus and Pediococcus parvulus strains with potential utility in the production of functional foods. Bioresource Technol 101:9254–9263
García-Cayuela T, Mohedano ML, Pérez-Gómez de Cadiñanos L, Fernández de Palencia P, Boden D, Wells J, Peláez C, López P, Requena T (2011) Transcriptional-fusion vectors for detection of uni- and bidirectional promoter regions in lactic acid bacteria. Spanish patent P201130356
Gasson MJ (1983) Plasmid complements of Streptococcus lactis NCDO 712 and other lactic streptococci after protoplast-induced curing. J Bacteriol 154:1–9
Gerritsen J, Smidt H, Rijkers GT, de Vos WM (2011) Intestinal microbiota in human health and disease: the impact of probiotics. Genes Nutr 6:209–240
Hanahan D (1985) Techniques for transformation of E. coli. In: Glover DM (ed) DNA cloning: a practical approach, vol 1. IRL, Oxford, pp 109–135
Holo H, Nes IF (1989) High-frequency transformation, by electroporation, of Lactococcus lactis subsp. cremoris grown with glycine in osmotically stabilized media. Appl Environ Microbiol 55:3119–3123
Israelsen H, Madsen SM, Vrang A, Hansen EB, Johansen E (1995) Cloning and partial characterization of regulated promoters from Lactococcus lactis Tn917-lacZ integrants with the new promoter probe vector, pAK80. Appl Environ Microbiol 61:2540–2547
Jacob AE, Hobbs SJ (1974) Conjugal transfer of plasmid-borne multiple antibiotic resistance in Streptococcus faecalis var. zymogenes. J Bacteriol 117:360–372
Kneen M, Farinas J, Li Y, Verkman AS (1998) Green fluorescent protein as a noninvasive intracellular pH indicator. Biophys J 74:1591–1599
Lacks S (1968) Genetic regulation of maltosaccharide utilization in pneumococcus. Genetics 60:685–706
Lagendijk EL, Validov S, Lamers GEM, de Weert S, Bloemberg GV (2010) Genetic tools for tagging Gram-negative bacteria with mCherry for visualization in vitro and in natural habitats, biofilm and pathogenicity studies. FEMS Microbiol Lett 305:81–90
Leroy F, de Vuyst L (2004) Lactic acid bacteria as functional starter cultures for the food fermentation industry. Trends Food Sci Technol 15:67–78
Maddocks SE, Oyston PCF (2008) Structure and function of the LysR-type transcriptional regulator (LTTR) family proteins. Microbiology 154:3609–3623
Miller WG, Lindow SE (1997) An improved GFP cloning cassette designed for prokaryotic transcriptional fusions. Gene 191:149–153
Montalbán-López M, Sánchez-Hidalgo M, Valdivia E, Martínez-Bueno M, Maqueda M (2011) Are bacteriocins underexploited? Novel applications for old antimicrobials. Curr Pharm Biotechnol 12:1205–1220
Müller-Taubenberger A, Anderson KI (2007) Recent advances using green and red fluorescent protein variants. Appl Microbiol Biotechnol 77:1–12
Nieto C, Puyet A, Espinosa M (2001) MalR-mediated regulation of the Streptococcus pneumoniae malMP operon at promoter PM. Influence of a proximal divergent promoter region and competition between MalR and RNA polymerase proteins. J Biol Chem 276:14946–14954
Otto R, Ten Brink B, Veldkamp H, Konings WN (1983) The relationship between growth rate and electrochemical proton gradient of Streptococcus cremoris. FEMS Microbiol Lett 16:69–74
Pereira PM, Veiga H, Jorge AM, Pinho MG (2010) Fluorescent reporters for protein cellular localization studies in Staphylococcus aureus. Appl Environ Microbiol 76:4346–4353
Poolman B, Konings WN (1988) Relation of growth of Streptococcus lactis and Streptococcus cremoris to amino acid transport. J Bacteriol 170:700–707
Rottiers P, De Smedt T, Steidler L (2009) Modulation of gut-associated lymphoid tissue functions with genetically modified Lactococcus lactis. Int Rev Inmunol 28:465–486
Ruiz-Cruz S, Solano-Collado V, Espinosa M, Bravo A (2010) Novel plasmid-based genetic tools for the study of promoters and terminators in Streptococcus pneumoniae and Enterococcus faecalis. J Microbiol Meth 83:156–163
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor
Shaner NC, Campbell RE, Steinbach PA, Giepmans BN, Palmer AE, Tsien RY (2004) Improved monomeric red, orange and yellow fluorescent proteins derived from Discosoma sp. red fluorescent protein. Nat Biotechnol 22:1567–1572
Stanton C, Ross RP, Fitzgerald GF, Van Sindern D (2005) Fermented functional foods based on probiotics and their biogenic metabolites. Curr Opin Biotechnol 16:198–203
Tolker-Nielsen T, Brinch UC, Ragas PC, Andersen JB, Jacobsen CS, Molin S (2000) Development and dynamics of Pseudomonas sp. biofilms. J Bacteriol 182:6482–6489
Wells JM (2011) Mucosal vaccination and therapy with genetically modified lactic acid bacteria mucosal vaccination and therapy with genetically modified lactic acid bacteria. Annu Rev Food Sci Technol 2:423–445
Wells JM, Mercenier A (2008) Mucosal delivery of therapeutic and prophylactic molecules using lactic acid bacteria. Nat Rev Microbiol 6:349–362
Willems RJ, Hanage WP, Bessen DE, Feil EJ (2011) Population biology of Gram-positive pathogens: high-risk clones for dissemination of antibiotic resistance. FEMS Microbiol Rev 35:872–900
Acknowledgments
This work was supported by the Spanish Ministry of Science and Innovation (grants AGL2009-12998-C03-01, AGL2009-13361-C02-02, and CSD2007-00063 Consolider Ingenio 2010 FUN-C-FOOD), the Comunidad de Madrid (grant ALIBIRD P2009/AGR-1469), and the European Union VII framework program (Initial Training Network, grant no. 238490).
We thank Dr. Stephen Elson for the critical reading of the manuscript. We are grateful to M. Angeles Corrales for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tomás García-Cayuela, Luz P. Gómez de Cadiñanos, and M. Luz Mohedano contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 68 kb)
Rights and permissions
About this article
Cite this article
García-Cayuela, T., de Cadiñanos, L.P.G., Mohedano, M.L. et al. Fluorescent protein vectors for promoter analysis in lactic acid bacteria and Escherichia coli . Appl Microbiol Biotechnol 96, 171–181 (2012). https://doi.org/10.1007/s00253-012-4087-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4087-z