Abstract
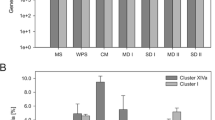
A strain of Clostridium kluyveri was isolated from the bovine rumen in a medium containing ethanol as an electron donor and acetate and succinate (common products of rumen fermentation) as electron acceptors. The isolate displayed a narrow substrate range but wide temperature and pH ranges atypical of ruminal bacteria and a maximum specific growth rate near the typical liquid dilution rate of the rumen. Quantitative real-time PCR revealed that C. kluyveri was widespread among bovine ruminal samples but was present at only very low levels (0.00002% to 0.0002% of bacterial 16S rRNA gene copy number). However, the species was present in much higher levels (0.26% of bacterial 16S rRNA gene copy number) in lucerne silage (but not maize silage) that comprised much of the cows’ diet. While C. kluyveri may account for several observations regarding ethanol utilization and volatile fatty acid production in the rumen, its population size and growth characteristics suggest that it is not a significant contributor to ruminal metabolism in typical dairy cattle, although it may be a significant contributor to silage fermentation. The ability of unadapted cultures to produce substantial levels (12.8 g L−1) of caproic (hexanoic) acid in vitro suggests that this strain may have potential for industrial production of caproic acid.



Similar content being viewed by others
References
Allison MJ, Dougherty RW, Bucklin JA, Snyder EE (1964) Ethanol accumulation in the rumen after overfeeding with readily fermentable carbohydrate. Science 144:54–56
Bader J, Günther H, Schleicher E, Simon H, Pohl S, Mannheim W (1980) Utilization of (E)-2-butenoate by Clostridium kluyveri and some other Clostridium species. Arch Microbiol 125:159–165
Balch WE, Wolfe RS (1976) New approach to the cultivation of methanogenic bacteria: 2-mercaptoethanesulfonic acid (HS-CoM)-dependent growth of Methanobacterium ruminantium in a pressurized atmosphere. Appl Environ Microbiol 32:781–791
Bornstein BT, Barker HA (1948) The nutrition of Clostridium kluyveri. J Biol Chem 55:223–230
Connor EE, Li RW, Baldwin RLVI, Li C (2010) Gene expression in the digestive tissues of ruminants and their relationship with feeding and digestive processes. Animal 4:993–1007
Esdale WJ, Broderick GA, Satter LD (1968) Measurement of ruminal volatile fatty acid production from alfalfa hay or corn silage rations using a continuous infusion isotope dilution technique. J Dairy Sci 51:1823–1830
Heijs SK, Haese RR, van der Wielen PWJJ, Forney LJ, Dirk van Elsas J (2007) Use of 16S rRNA gene based clone libraries to assess microbial communities potentially involved in anaerobic methane oxidation in a Mediterranean cold seep. Microb Ecol 53:384–398
Jeon BS, Kim BC, Um Y, Sang BI (2010) Production of hexanoic acid from D-galactitol by a newly isolated Clostridium sp. BS-1. Appl Microbiol Biotechnol 88:1161–1167
Jones DW, Kay JJ (1976) Determination of volatile fatty acids C1–C6, and lactic acid in silage juice. J Sci Food Agric 27:1005–1114
Kenealy WR, Wasalefsky DM (1985) Studies on the substrate range of Clostridium kluyveri; the use of propanol and succinate. Arch Microbiol 141:187–194
Kenealy WR, Cao Y, Weimer PJ (1995) Production of caproic acid by cocultures of ruminal cellulolytic bacteria and Clostridium kluyveri grown on cellulose and ethanol. Appl Microbiol Biotechnol 44:507–515
Levy PF, Sanderson JE, Ashare E, deRiel SR (1983) Alkane liquid fuels production from biomass. In: Wise DL (ed) Liquid fuel developments. CRC, Boca Raton, pp 159–188
Makoni NF, Broderick GA, Muck RE (1997) Effect of modified atmospheres on proteolysis and fermentation of ensiled alfalfa. J Dairy Sci 80:912–920
Moomaw CR, Hungate RE (1963) Ethanol conversion in the bovine rumen. Appl Microbiol 85:721–722
Ohyama Y, Hara S-I, Masaki S (1977) The use of caproic acid to prevent aerobic deterioration of silages after opening, with special reference to the amounts and time of application. J Sci Food Agric 28:369–374
Ørskov ER, Flatt WP, Moe PW, Oltjen RR (1967) Caproic acid and ethanol in ruminal ingesta of cattle receiving purified diets. J Dairy Sci 50:239–242
Pradhan K, Hemken RW (1970) Utilization of ethanol and its effect on fatty acid patterns in ruminants. J Dairy Sci 53:1739–1746
Raun BML, Kristensen NB (2011) Metabolic effects of feeding ethanol or propanol to postpartum transition Holstein cows. J Dairy Sci 94:2566–2580
Russell JB (1992) Another explanation for the toxicity of fermentation acids at low pH: anion accumulation versus uncoupling. J Appl Bacteriol 73:363–370
Russell JB (2002) Rumen microbiology and its role in ruminant nutrition. Russell, Ithaca
Schaefer DM, Davis CL, Bryant MP (1980) Ammonia saturation constants for predominant species of rumen bacteria. J Dairy Sci 63:1248–1263
Stevenson DM, Weimer PJ (2007) Dominance of Prevotella and low abundance of classical ruminal bacterial species in the bovine rumen revealed by relative quantification real-time PCR. Appl Microbiol Bioechnol 75:165–174
Ungerfeld EM, Kohn RA (2006) The role of thermodynamics in the control of ruminal fermentation. In: Sejrsen K, Hvelplund T, Nielsen MO (eds) Ruminant physiology. Wageningen Academic, Wageningen, pp 55–85
Acknowledgments
We thank Christine Odt for technical assistance, Nancy Betzold and the USDFRC barn crew for animal handling, and Richard Muck for helpful discussions. This research was supported by the USDA Agricultural Research Service thorough CRIS project 3655-31000-06-00D.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weimer, P.J., Stevenson, D.M. Isolation, characterization, and quantification of Clostridium kluyveri from the bovine rumen. Appl Microbiol Biotechnol 94, 461–466 (2012). https://doi.org/10.1007/s00253-011-3751-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3751-z