Abstract
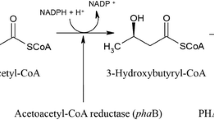
Pseudomonas putida KT2442 has been a well-studied producer of medium-chain-length (mcl) polyhydroxyalkanoate (PHA) copolymers containing C6 ~ C14 monomer units. A mutant was constructed from P. putida KT2442 by deleting its phaG gene encoding R-3-hydroxyacyl-ACP-CoA transacylase and several other β-oxidation related genes including fadB, fadA, fadB2x, and fadAx. This mutant termed P. putida KTHH03 synthesized mcl homopolymers including poly(3-hydroxyhexanoate) (PHHx) and poly(3-hydroxyheptanoate) (PHHp), together with a near homopolymer poly(3-hydroxyoctanoate-co-2 mol% 3-hydroxyhexanoate) (PHO*) in presence of hexanoate, heptanoate, and octanoate, respectively. When deleted with its mcl PHA synthase genes phaC1 and phaC2, the recombinant mutant termed P. putida KTHH08 harboring pZWJ4-31 containing PHA synthesis operon phaPCJ from Aeromonas hydrophila 4AK4 accumulated homopolymer poly(3-hydroxyvalerate) (PHV) when valerate was used as carbon source. The phaC deleted recombinant mutant termed P. putida KTHH06 harboring pBHH01 holding PHA synthase PhbC from Ralstonia eutropha produced homopolymers poly(3-hydroxybutyrate) (PHB) and poly(4-hydroxybutyrate) using γ-butyrolactone was added as precursor. All the homopolymers were physically characterized. Their weight average molecular weights ranged from 1.8 × 105 to 1.6 × 106, their thermal stability changed with side chain lengths. The derivatives of P. putida KT2442 have been developed into a platform for production of various PHA homopolymers.




Similar content being viewed by others
References
Chen GQ (2009) A microbial polyhydroxyalkanoates (PHA) based bio- and materials industry. Chem Soc Rev 38:2434–2446
Elbahloul Y, Steinbüchel A (2009) Large-scale production of poly(3-hydroxyoctanoic acid) by Pseudomonas putida GPo1 and a simplified downstream process. Appl Environ Microbiol 75:643–651
Feng LD, Watanabe T, Wang Y, Kichise T, Fukuchi T, Chen GQ, Doi Y, Inoue Y (2002) Studies on comonomer compositional distribution of bacterial poly(3-hydroxybutyrate-co-3-hydroxyhexanoate)s and thermal characteristics of their factions. Biomacromolecules 3:1071–1077
Friedrich B, Hogrefe C, Schlegel HG (1981) Naturally occurring genetic transfer of hydrogen-oxidizing ability between strains of Alcaligenes eutrophus. J Bacteriol 147:198–205
Gao HJ, Wu Q, Chen GQ (2002) Enhanced production of d-(-)-3-hydroxybutyric acid by recombinant Escherichia coli. FEMS Microbiol Lett 213:59–65
Haywood GW, Anderson AJ, Dawes EA (1989) The importance of PHA-synthase substrate specificity in polyhydroxyalkanoate synthesis by Alcaligenes eutrophus. FEMS Microbiol Lett 57:1–6
Hazer B, Steinbüchel A (2007) Increased diversification of polyhydroxyalkanoates by modification reactions for industrial and medical applications. Appl Microbiol Biotechnol 74:1–12
Hein S, Söhling B, Gottschalk G, Steinbüchel A (1997) Biosynthesis of poly(4-hydroxybutyric acid) by recombinant strains of Escherichia coli. FEMS Microbiol Lett 153:411–418
Kato M, Bao HJ, Kang CK, Fukui T, Doi Y (1996) Production of a novel copolyester of 3-hydroxybutyric acid and medium-chain-length 3-hydroxyalkanoic acids by Pseudomonas sp. 61-3 from sugars. Appl Microbiol Biotechnol 45:363–370
Kovacha ME, Elzera PH, Hillb DS, Robertsona GT, Farrisa MA, Roop RMII, Peterson KM (1995) Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene 166:175–176
Lawrence AG, Choi J, Rha C, Stubbe J, Sinskey AJ (2005) In vitro analysis of the chain termination reaction in the synthesis of poly-(R)-beta-hydroxybutyrate by the class III synthase from Allochromatium vinosum. Biomacromolecules 6:2113–2119
Liu WK, Chen GQ (2007) Production and characterization of medium chain length polyhydroxyalkanoate with high 3-hydroxytetradecanoate monomer content by fadB and fadA knockout mutant of Pseudomonas putida KT2442. Appl Microbiol Biotechnol 76:1153–1159
Lu XY, Zhang WJ, Wu Q, Chen GQ (2005) Molecular cloning and functional analysis of two polyhydroxyalkanoate synthases from two strains of Aeromonas hydrophila spp. FEMS Microbiol Lett 243:149–155
Lu JN, Tappel RC, Nomura CT (2009) Mini-review: biosynthesis of poly(hydroxyalkanoates). Polym Revt 49:226–248
Ouyang SP, Liu Q, Fang L, Chen GQ (2007a) Construction of pha-operon-defined knockout mutants of Pseudomonas putida KT2442 and their applications in poly(hydroxyalkanoate) production. Macromol Biosci 7:227–233
Ouyang SP, Luo RC, Chen SS, Liu Q, Chung A, Wu Q, Chen GQ (2007b) Production of polyhydroxyalkanoates with high 3-hydroxydodecanoate monomer content by fadB and fadA knockout mutant of Pseudomonas putida KT2442. Biomacromolecules 8:2504–2511
Qu XH, Wu Q, Liang J, Qu X, Wang SG, Chen GQ (2005) Enhanced vascular-related cellular affinity on surface modified copolyesters of 3-hydroxybutyrate and 3-hydroxyhexanoate (PHBHHx). Biomaterials 26:6991–7001
Qu XH, Wu Q, Liang J, Zou B, Chen GQ (2006) Effect of 3-hydroxyhexanoate content in poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) on in vitro growth and differentiation of smooth muscle cells. Biomaterials 27:2944–2950
Reddy CSK, Ghai R, Rashmi C, Kalia VC (2003) Polyhydroxyalkanoates: an overview. Bioresour Technol 87:137–146
Rehm BHA (2003) Polyester synthases: natural catalysts for plastics. Biochem J 376:15–33
Rehm BHA, Krüger N, Steinbüchel A (1998) A new metabolic link between fatty acid de novo synthesis and polyhydroxyalkanoic acid synthesis - The phaG gene from Pseudomonas putida KT2440 encodes a 3-hydroxyacyl-acyl carrier protein coenzyme A transferase. J Biol Chem 273:24044–24051
Ren Q, Grubelnik A, Hoerler M, Ruth K, Hartmann R, Felber H, Zinn M (2005) Bacterial poly(hydroxyalkanoates) as a source of chiral hydroxyalkanoic acids. Biomacromolecules 6:2290–2298
Ren Q, Ruth K, Thony-Meyer L, Zinn M (2007) Process engineering for production of chiral hydroxycarboxylic acids from bacterial polyhydroxyalkanoates. Macromol Rapid Com 28:2131–2136
Ruth K, Grubelnik A, Hartmann R, Egli T, Zinn M, Ren Q (2007) Efficient production of (R)-3-hydroxycarboxylic acids by biotechnological conversion of polyhydroxyalkanoates and their purification. Biomacromolecules 8:279–286
Schäfer A, Tauch A, Jäger W, Kalinowski J, Thierbach G, Pühler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19: selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145:69–73
Shen XW, Yang Y, Jian J, Wu Q, Chen GQ (2009) Production and characterization of homopolymer poly(3-hydroxyvalerate) (PHV) accumulated by wild type and recombinant Aeromonas hydrophila strain 4AK4. Bioresour Technol 100:4296–4299
Solaiman DKY, Ashby RD, Foglia TA (2001) Production of polyhydroxyalkanoates from intact triacylglycerols by genetically engineered Pseudomonas. Appl Microbiol Biotechnol 56:664–669
Steinbüchel A, Debzi EM, Marchessault RH, Timm A (1993) Synthesis and production of poly (3-hydroxyvaleric acid) homopolyester by Chromabacterium violaceum. Appl Microbiol Biotechnol 39:443–449
Steinbüchel A, Valentin HE, Schönebaum A (1994) Application of recombinant gene technology for production of polyhydroxyalkanoic acids: biosynthesis of poly(4-hydroxybutyric acid) homopolyester. J Environ Polym Degrad 2:67–74
Sudesh K, Abe H, Doi Y (2000) Synthesis, structure and properties of polyhydroxyalkanoates: biological polyesters. Prog Polym Sci 25:1503–1555
Sun ZY, Ramsay JA, Guay M, Ramsay BA (2007) Fermentation process development for the production of medium-chain-length poly-3-hyroxyalkanoates. Appl Microbiol Biotechnol 75:475–485
Timm A, Steinbüchel A (1992) Cloning and molecular analysis of the poly(3-hydroxyalkanoic acid) gene locus of Pseudomonas aeruginosa PAO1. Eur J Biochem 209:15–30
Wang HH, Li XT, Chen GQ (2009) Production and characterization of homopolymer polyhydroxyheptanoate (P3HHp) by a fadBA knockout mutant Pseudomonas putida KTOY06 derived from P. putida KT2442. Process Biochem 44:106–111
Acknowledgements
This research was supported by Li Ka Shing Foundation and National High Tech 863 Grant (project no. 2006AA02Z242), as well as the State Basic Science Foundation 973 (2007CB707804). We thank Dr. Ouyang SP of Tsinghua University for kindly donating P. putida KTOY06, KTOY08, and plasmid pSPK02.We also thank Dr. Haimu Ye, Mr. Linping Wu, Liang Wang, and Hao Ni for many other forms of help they gave. In addition, Mr. Shenyu Wang, Guangyao Li, Wenfeng Ou, and Jueyu Pan provided assistances in the form of the use of some facilities for cell growth and physical characterization.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hong-hui Wang and Xin-rong Zhou contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Wang, Hh., Zhou, Xr., Liu, Q. et al. Biosynthesis of polyhydroxyalkanoate homopolymers by Pseudomonas putida . Appl Microbiol Biotechnol 89, 1497–1507 (2011). https://doi.org/10.1007/s00253-010-2964-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-010-2964-x