Abstract
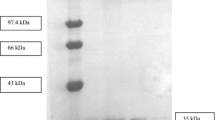
A laccase from Pycnoporus sanguineus was purified by two steps using phenyl-Sepharose columm. A typical procedure provided 54.1-fold purification, with a yield of 8.37%, using syringaldazine as substrate. The molecular weight of the purified laccase was 69 and 68 kDa as estimated by 12% (w/v) SDS-PAGE gel and by gel filtration, respectively. The K m values for the substrates ABTS, syringaldazine, and guaiacol were 58, 8.3, and 370 μM, respectively. The enzyme’s pH optimum for syringaldazine was 4.2 and optimal activity was 50°C. The enzyme showed to be thermostable because when kept at 50°C for 24 and 48 h it retained 93 and 76% activity. This laccase was inhibited by l-cysteine, β-mercaptoethanol, NaN3, NaF, and HgCl2.





Similar content being viewed by others
References
Antorini M, Herpöel-Gimbert I, Choinowski T, Sigoillot, J-C, Asther M, Winterhalter K, Piontek K (2002) Purification, crystallization and X-ray diffraction study of fully functional laccases from two ligninolytic fungi. Biochim Biophys Acta 1594:109–114
Baldrian P (2004) Purification and characterization of laccase from the white-rot fungus Daedalea quercina and decolorization of synthetic dyes by the enzyme. Appl Microbiol Biotechnol 63:560–563
Blum H, Beier H, Gross H (1987) Improved silver staining of plant proteins, RNA and DNA in polyacrylamide gels. Electrophoresis 8:93–99
Bourbonnais R, Paice MG (1990) Oxidation of non-phenolic substrates. An expanded role for laccase in lignin biodegradation. FEBS Lett 267:99–102
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of proteins utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chefetz B, Chen Y, Hadar Y (1998) Purification and characterization of laccase from Chaetomium thermophilium and its role in humification. Appl Environ Microbiol 64:3175–3179
Dong JL, Zhang YZ (2004) Purification and characterization of two laccase isoenzymes from a lignolytic fungus Trametes gallica. Prep Biochem Biotechnol 34:179–194
Edens WA, Goins TQ, Dooley D, Henson JM (1999) Purification and characterization of a secreted laccase of Gaeumannomyces graminis var. tritici. Appl Environ Microbiol 65:3071–3074
Eggert C, Temp U, Eriksson K-EL (1996) The lignolytic system of the white rot fungus Pycnoporus cinnabarinus: purification and characterization of the laccase. Appl Environ Microbiol 62:1151–1158
Esposito E, Innocentini-Mei LH, Ferraz A, Canhos VP, Duran N (1993) Phenoloxidases and hydrolases from Pycnoporus sanguineus (EUC-2050 strain): applications. J Biotechnol 29:219–228
Farnet AM, Criquet S, Tagger S, Gil G, Le Petit J (2000) Purification, partial characterization, and reactivity with aromatic compounds of two laccases from Marasmius quercophilus strain 17. Can J Microbiol 46:189–194
Galhaup C, Goller S, Peterbauer CK, Strauss J, Haltrich D (2002) Characterization of the major laccase isoenzyme from Trametes pubescens and regulation of its synthesis by metal ions. Microbiology 148:2159–2169
Garcia TA, Santiago MF, Ulhoa CJ (2006) Properties of laccases produced by Pycnoporus sanguineus induced by 2,5-xylidine. Biotechnol Lett 28:633–636
Gianfreda S, Xu F, Bollag J-M (1999) Laccases: a useful group of oxidoreductive enzymes. Bioremediation J 3:1–25
Gonçalves MLFC, Steiner W (1996) Detection of laccase activity in polyacrylamide gels after electrophoresis under denaturing conditions. Biotechnol Tech 10:667–668
Harkin JM, Obst JR (1973) Syringaldazine, an efective reagent for detecting laccase and peroxidases in fungi. Mycologia 29:381–387
Jaouani A, Guillén F, Penninckx MJ, Martínez AT, Martínez MJ (2005) Role of Pycnoporus coccineus laccase in the degradation of aromatic compounds in olive oil mill wastewater. Enzyme Microb Technol 36:478–486
Kiiskinen L-L, Viikari L, Kruus K (2002) Purification and characterization of a novel laccase from the ascomycete Melanocarpus albomyces. Appl Microbiol Biotechnol 59:198–204
Kiiskinen L-L, Kruus K, Bailey M, Ylösmäki E, Siika-Aho M, Saloheimo M (2004) Expression of Melanocarpus albomyces laccase in Trichoderma reesei and characterization of the purified enzyme. Microbiology 150:3065–3074
Kim Y, Cho N-S, Eom T-J, Shiun W (2002) Purification and characterization of a laccase from Cerrena unicolor and its reactivity in lignin degradation. Bull Korean Chem Soc 23:985–989
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Leonowicz A, Grzywnowicz K (1981) Quantitative estimation of laccase forms in some white-rot fungi using syringaldazine as a substrate. Enzyme Microb Technol 3:55–58
Lomascolo A, Cayol J-L, Roche M, Guo L, Robert J-L, Record E, Lesage-Meessen L, Ollivier B, Sigoillot J-C, Asther M (2002) Molecular clustering of Pycnoporus strains from various geographic origins and isolation of monocaryotic strains for laccase hyperproduction. Mycol Res 106:1193–1203
Mayer AM, Staples RC (2002) Review—laccase: new functions for an old enzyme. Phytochemistry 60:551–565
Nagai M, Sato T, Watanabe H, Saito K, Kawata M, Enei H (2002) Purification and characterization of an extracellular laccase from the edible mushroom Lentinula edodes, and decolorization of chemical different dyes. Appl Microbiol Biotechnol 60:327–335
Niku-Paavola M-L, Fagerström R, Kruus K, Viikari L (2004) Thermostable laccases produced by a white-rot fungus from Peniophora species. Enzyme Microb Technol 35:100–102
Oscarsson S, Kårsnäs P (1998) Salt-promoted adsorption of proteins onto amphiphilic agarose-based adsorbents. II. Effects of salt and salt concentration. J Chromatogr A 803:83–93
Palmieri G, Giardina P, Bianco C, Scalonit A, Capassol A, Sannia G (1997) A novel white laccase from Pleurotus ostreatus. J Biol Chem 272:31301–31307
Pérez J, Martínez J, DeLa Rubia T (1996) Purification and partial characterization of a laccase from the white rot fungus Phanerochaete flavido-alba. Appl Environ Microbiol 62:4263–4267
Pezet R (1998) Purification and characterization of a 32-kDa laccase-like stilbene oxidase produced by Botrytis cinerea Pers.: Fr. FEMS Microbiol Lett 167:203–208
Pointing SB, Jones EBG, Vrijmoed LLP (2000) Optimization of laccase production by Pycnoporus sanguineus in submerged liquid culture. Mycologia 92:139–144
Record E, Punt PJ, Chamkha M, Labat M, Van Den Hondel CAMJJ (2002) Expression of the Pycnoporus cinnabarinus laccase gene in Aspergillus niger and characterization of the recombinant enzyme. Eur J Biochem 269:602–609
Rehman AU, Thurston CF (1992) Purification of laccase-I from Armillaria mellea. J Gen Microbiol 138:1251–1257
Robles A, Lucas R, Martínez-Cañamero M, Omar NB, Pérez R, Gálvez A (2002) Characterization of laccase activity produced by the hyphomycete Chalara (Syn. Thielavopsis) paradoxa CH32. Enzyme Microb Technol 31:516–522
Salas C, Lobos S, Larrain J, Salas L, Cullen D, Vicuna R (1995) Properties of laccase isoenzymes produced by the basidiomycete Ceriporiopsis subvermispora. Biotechnol Appl Biochem 21:323–333
Schliephake K, Mainwaring DE, Lonergan GT, Jones K, Baker WL (2000) Transformation and degradation of the disazo dye Chicago Sky Blue by a purified laccase from Pycnoporus cinnabarinus. Enzyme Microb Technol 27:100–107
Scherer M, Fischer R (1998) Purification and characterization of laccase II of Aspergillus nidulans. Arch Microbiol 170:78–84
Souza CGM, Peralta RM (2003) Purification and characterization of the main laccase produced by the white-rot fungus Pleurotus pulmonarius on wheat bran solid state medium. J Basic Microbiol 43:278–286
Thurston CF (1994) The structure and function of fungal laccases. Microbiology 140:19–26
Vasdev K, Dhawan S, Kapoor RK, Kuhad RC (2005) Biochemical characterization and molecular evidence of a laccase from the bird’s nest fungus Cyathus bulleri. Fungal Genet Biol 42:684–693
Vikineswary S, Noorlidah A, Renuvathani M, Sekaran M, Pandey A, Jones EBG (2006) Productivity of laccase in solid substrate fermentation of selected agro-residues by Pycnoporus sanguineus. Bioresour Technol 97:171–177
Xu F (1996) Oxidation of phenols, anilines, and benzenethiols by fungal laccases: correlation between activity and redox potentials as well as halide inhibition. Biochemistry 35:7608–7614
Xu F (1997) Effects of redox potential and hydroxide inhibition on the pH activity profile of fungal laccases. J Biol Chem 272:924–928
Acknowledgments
This work was supported by Grants from CNPq, FUNAPE/UFG, and International Foundation for Science, Stockholm, Sweden, through a grant to Dr MFS (IFS-W/3433-1). TAG was supported by CAPES/Brazil.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garcia, T.A., Santiago, M.F. & Ulhoa, C.J. Studies on the Pycnoporus sanguineus CCT-4518 laccase purified by hydrophobic interaction chromatography. Appl Microbiol Biotechnol 75, 311–318 (2007). https://doi.org/10.1007/s00253-006-0817-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-006-0817-4