Abstract
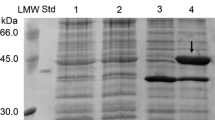
Thirteen different chitin-degrading bacteria were isolated from soil and sediment samples. Five of these strains (SGE2, SGE4, SSL3, MG1, and MG3) exhibited antifungal activity against phytopathogenic fungi. Analyses of the 16S rRNA genes and the substrate spectra revealed that the isolates belong to the genera Bacillus or Streptomyces. The closest relatives were Bacillus chitinolyticus (SGE2, SGE4, and SSL3), B. ehimensis (MG1), and Streptomyces griseus (MG3). The chitinases present in the culture supernatants of the five isolates revealed optimal activity between 45°C and 50°C and at pH values of 4 (SSL3), 5 (SGE2 and MG1), 6 (SGE4), and 5–7 (MG3). The crude chitinase preparations of all five strains possessed antifungal activity. The chitinase of MG3 (ChiIS) was studied further, since the crude enzyme conferred strong growth suppression of all fungi tested and was very active over the entire pH range tested. The chiIS gene was cloned and the gene product was purified. The deduced protein consisted of 303 amino acids with a predicted molecular mass of 31,836 Da. Sequence analysis revealed that ChiIS of MG3 is similar to chitinases of Streptomyces species, which belong to family 19 of glycosyl hydrolases. Purified ChiIS showed remarkable antifungal activity and stability.



Similar content being viewed by others
References
Alam M, Nikaidou N, Tanaka H, Watanaba T (1995) Cloning and sequencing of chiC gene of Bacillus circulans WL-12 and relationship of its product to some other chitinases and chitinase-like proteins. J Ferment Bioeng 80:454–461
Andersen MD, Jensen A, Robertus JD, Leah R, Skriver K (1997) Heterologous expression and characterization of wild-type and mutant forms of a 26 kDa endochitinase from barley (Hordeum vulgare L.). Biochem J 322:815–822
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1987) Current protocols in molecular biology. Wiley, New York
Bennett JW, Lasure LL (1991) Growth media. In: Bennett JW, Lasure LL (eds) More gene manipulations in fungi. Academic, San Diego, pp 441–458
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chang MM, Horovitz D, Culley D, Hadwiger LA (1995) Molecular cloning and characterization of a pea chitinase gene expressed in response to wounding, fungal infection and the elicitor chitosan. Plant Mol Biol 28:105–111
Chen HC, Huang MY, Moody MW, Jiang ST (1991) Distribution and hydrolytic enzyme activities of aerobic, heterotrophic bacteria isolated from grass prawn, Penaeus monodon. J Fish Soc Taiwan 18:301–310
Chernin LS, de la Fuente L, Sobolev V, Haran S, Vorgias CE, Oppenheim AB, Chet I (1997) Molecular cloning, structural analysis, and expression in Escherichia coli of a chitinase gene from Enterobacter agglomerans. Appl Environ Microbiol 63:834–839
Cody RM, Davis ND, Lin J, Shaw D (1990) Screening microorganisms for chitin hydrolysis and production of ethanol from amino sugars. Biomass 21:285–295
Davis G, Henrissat B (1995) Structures and mechanisms of glycosyl hydrolases. Structure 3:853–859
Dempsey DMA, Silva H, Klessig DF (1998) Engineering disease and pest resistance in plants. Trends Microbiol 6:54–61
Devereux J, Haeberli P, Smithies O (1984) A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res 12:387–395
Downing KJ, Thomson JA (2000) Introduction of the Serratia marcescenschiA gene into an endophytic Pseudomonas fluorescens for the biocontrol of phytopathogenic fungi. Can J Microbiol 46:363–369
Fujii T, Miyashita K (1993) Multiple domain structure in a chitinase gene (chiC) of Streptomyces lividans. J Gen Microbiol 139:677–686
Henrissat B, Bairoch A (1993). New families in the classification of glycosyl hydrolases based on amino acid sequence. Biochem J 293:781–788
Honee G (1999) Engineered resistance against fungal plant pathogens. Eur J Plant Pathol 105:319–326
Ikegami T, Okada T, Hashimoto M, Seino S, Watanabe T, Shirakawa M (2000) Solution structure of the chitin-binding domain of Bacillus circulans WL-12 chitinase A1. J Biol Chem 275:13654–13661
Imoto T, Yagashita K (1971) A simple activity measurement of lysozyme. Agric Biol Chem 35:1154–1156
Itoh Y, Takahashi K, Takizawa H, Nikaidou N, Tanaka H, Nishihashi H, Watanabe T, Nishizawa Y (2003) Family 19 chitinase of Streptomyces griseus HUT6037 increases plant resistance to the fungal disease. Biosci Biotechnol Biochem 67:847–855
Ivanova IP, Vysotski MV, Svetashev V, Gorshkova M, Taguchi T, Yoshikawa S (1999) Characterization of Bacillus strains of marine origin. Int Microbiol 2:267–271
Kawase T, Saito A, Sato T, Kanai R, Fujii T, Nikaidou N, Miyashita K, Watanabe T (2004) Distribution and phylogenetic analysis of family 19 chitinases in Actinobacteria. Appl Environ Microbiol 70:1135–1144
Kim K-J, Yang Y-J, Kim J-G (2003) Purification and characterization of chitinase from Streptomyces sp. M-20. J Biochem Mol Biol 36:185–189
Kobayashi DY, Reedy RM, Bick JA, Oudemans PV (2002) Characterization of a chitinase gene from Stenotrophomonas maltophilia strain 34S1 and its involvement in biological control. Appl Environ Microbiol 68:1047–1054
Koga D, Mitsutomi M, Kono M, Matsumiya M (1999) Biochemistry of chitinases. EXS 87:111–123
Kuroshima K-I, Sakane T, Takata R, Yokota A (1996) Bacillus ehimensis sp. nov. and Bacillus chitinolyticus sp. nov., new chitinolytic members of the genus Bacillus. Int J Syst Bacteriol 46:76–80
Matsuda Y, Ida Y, Shinogi T, Kakutani K, Nonomura T, Toyoda H (2001) In vitro suppression of mycelial growth of Fusarium oxysporum by extracellular chitosanase of Sphingobacterium multivorum and cloning of the chitosanase gene csnSM1. J Gen Plant Pathol 67:318–324
Ohno T, Armand S, Hata T, Nikaidou N, Henrissat B, Mitsutomi M, Watanabe T (1996) A modular family 19 chitinase found in the prokaryotic organism Streptomyces griseus HUT 6037. J Bacteriol 178:5065–5070
Pfennig N, Lippert KD (1966) Über das Vitamin B12-Bedürfnis phototropher Schwefelbakterien. Arch Microbiol 55:245–256
Pisano MA, Sommer MJ, Tars L (1992) Bioactivity of chitinolytic actinomycetes from marine origin. Appl Microbiol Biotechnol 36:553–555
Roberts WK, Selitrennikoff CP (1986) Isolation and partial characterization of two antifungal proteins from barley. Biochim Biophys Acta 880:161–170
Robinson SP, Jacobs AK, Dry IB (1997) A class IV chitinase is highly expressed in grape berries during ripening. Plant Physiol 114:771–778
Ruiz-Herrera J, Martinez-Espinoza AD (1999) Chitin biosynthesis and structural organization in vivo. In: Julles P, Muzzarelli RAA (eds) Chitin and chitinases. Birkhäuser, Basel, pp 39–53
Saito A, Fujii T, Yoneyama T, Redenbach M, Ohno T, Watanabe T, Miyashita K (1999) High-multiplicity of chitinase genes in Streptomyces coelicolor A3(2). Biosci Biotechnol Biochem 63:710–718
Schägger H, von Jagow G (1987) Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem 166:368–379
Singh P, Shin YC, Park CS, Chung, YR (1998) Biological control of Fusarium wilt of cucumber by chitinolytic bacteria. Phytopathology 89:92–99
Tsujibo H, Orikoshi C, Shiotani K, Hayashi M, Umeda J, Miyamoto K, Imada C, Okami Y, Inamori Y (1998) Characterization of chitinase C from a marine bacterium, Alteromonas sp. strain O-7, and its corresponding gene and domain structure. Appl Environ Microbiol 64:472–478
Wang S-L, Chang W-T (1997) Purification and characterization of two bifunctional chitinases/lysozymes extracellulary produced by Pseudomonas aeruginosa K-187 in a shrimp and crab shell powder medium. Appl Environ Microbiol 63:380–386
Watanabe T, Kanai R, Kawase T, Tanabe T, Mitsutomi M, Sakuda S, Miyashita K (1999) Family 19 chitinases of Streptomyces species: characterization and distribution. Microbiology 145:3353–3363
Williams ST, Goodfellow M, Wellington EM, Vickers JC, Alderson G, Sneath PH, Sackin MJ, Mortimer AM (1983) A probability matrix for identification of some Streptomycetes. J Gen Microbiol 129:1815–1830
Wiwat C, Siwayaprahm P, Bhumiratana A (1999) Purification and characterization of chitinase from Bacillus circulans No. 4.1. Curr Microbiol 39:134–140
Wolin EA, Wolfe RS, Wolin MJ (1964) Viologen dye inhibition of methane formation by Methanobacterium omelianskii. J Bacteriol 87:993–998
Acknowledgements
We thank Gerhard Gottschalk for generous support and helpful discussions. This work was supported by the Deutsche Bundesstiftung Umwelt.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoster, F., Schmitz, J.E. & Daniel, R. Enrichment of chitinolytic microorganisms: isolation and characterization of a chitinase exhibiting antifungal activity against phytopathogenic fungi from a novel Streptomyces strain. Appl Microbiol Biotechnol 66, 434–442 (2005). https://doi.org/10.1007/s00253-004-1664-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-004-1664-9