Abstract
Advances in genome sequencing technology have enabled genomes of extinct species to be sequenced. However, given the fragmented nature of these genome assemblies, it is not clear whether it is possible to comprehensively annotate highly variable and repetitive genes such as those involved in immunity. As such, immune genes have only been investigated in a handful of extinct genomes, mainly in human lineages. In 2018 the genome of the thylacine (Thylacinus cynocephalus), a carnivorous marsupial from Tasmania that went extinct in 1936, was sequenced. Here we attempt to characterise the immune repertoire of the thylacine and determine similarity to its closest relative with a genome available, the Tasmanian devil (Sarcophilus harrisii), as well as other marsupials. Members from all major immune gene families were identified. However, variable regions could not be characterised, and complex families such as the major histocompatibility complex (MHC) were highly fragmented and located across multiple small scaffolds. As such, at a gene level we were unable to reconstruct full-length coding sequences for the majority of thylacine immune genes. Despite this, we identified genes encoding functionally important receptors and immune effector molecules, which suggests the functional capacity of the thylacine immune system was similar to other mammals. However, the high number of partial immune gene sequences identified limits our ability to reconstruct an accurate picture of the thylacine immune repertoire.


Similar content being viewed by others
Data availability
The thylacine genome is published (Feigin et al. 2018); genome sequence reads are available through the Sequence Read Archive under BioProject PRJNA354646, BioSample SAMN06049672. Characterised thylacine immune gene sequences are available in the supplementary material.
References
Abi-Rached L et al (2011) The shaping of modern human immune systems by multiregional admixture with archaic humans. Science 334:89–94
Abts KC, Ivy JA, DeWoody JA (2015) Immunomics of the koala (Phascolarctos cinereus). Immunogenetics 67:305–321. https://doi.org/10.1007/s00251-015-0833-6
Basden K, Cooper DW, Deane EM (1997) Development of the lymphoid tissues of the tammar wallaby Macropus eugenii Reproduction. Fertil Dev 9:243–254
Belov K et al (2006) Reconstructing an ancestral mammalian immune supercomplex from a marsupial major histocompatability complex. PLOS Biol 4:317–328
Belov K et al (2007) Characterization of the opossum immune genome provides insight into the evolution of the mammalian immune system. Genome Res 17:982–991
Bininda-Emonds ORP et al (2007) The delayed rise of present-day mammals. Nature 445:507–512
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL (2009) BLAST+: architecture and applications. BMC bioinformatics 10:421
Cheng Y, Polkinghorne A, Gillett A, Jones EA, O’Meally D, Timms P, Belov K (2018) Characterisation of MHC class I genes in the koala. Immunogenetics 70:125–133
Cheng Y, Sanderson CE, Jones M, Belov K (2012) Low MHC class II diversity in the Tasmanian devil. Immunogenetics 64:525–533
Cheng Y, Siddle HV, Beck S, Eldridge MDB, Belov K (2009) High levels of genetic variation at MHC class II DBB loci in the tammar wallaby (Macropus eugenii). Immunogenetics 61:111–118
Cheng Y et al (2012) Antigen-presenting genes and genomic copy number variations in the Tasmanian devil MHC. BMC Genomics 13:87
Commins SP, Borish L, Steinke JW (2010) Immunological messenger molecules: cytokines, interferons and chemokines. J Allergy Clin Immunol 125:S53–S72
Cui J, Cheng Y, Belov K (2015) Diversity in the Toll-like receptor genes of the Tasmanian devil (Sarcophilus harrisii). Immunogenetics 67:195–201. https://doi.org/10.1007/s00251-014-0823-0
Cui J et al (2015) SNP marker discovery in koala TLR genes. PLOS ONE 10:1–15
Dal Porto JM, Gauld SB, Merrell KT, Mills D, Pugh-Bernard AE, Cambier J (2004) B cell antigen receptor signalling 101. Mol Immunol 41:599–613
Dannemann M, Andres AM, Kelso J (2016) Introgression of neandertal- and denisovan-like haplotypes contributes to adaptive variation in human toll-like receptors. Am J Med Human Genet 98:22–33
Deschamps M et al (2016) Genomic signatures of selective pressures and introgression from archaic hominins at human innate immunity genes The Americal. J Hum Genet 98:5–21
Duncan LG, Nair SV, Deane EM (2007) Molecular characterisation and expression of CD4 in two distantly related marsupials: The gray short-tailed opossum (Monodelphis domestica) and tammar wallaby (Macropus eugenii). Mol Immunol 44:3641–3652
Duncan LG, Nair SV, Deane EM (2009) The marsupial CD8 gene locus: Molecular cloning and expression analysis of the alpha and beta dequences in the gray short-tailed opossum (Monodelphis domestica) and the tammar wallaby (Macropus eugenii). Vet Immunol Immunopathol 129:14–27
Duncan LG, Webster K, Gupta V, Nair SV, Deane EM (2010) Molecular characterisation of the CD79a and CD79b subunits of the B cell receptor compled in the gray short-tailed opossum (Monodelphis domestica) and tammar wallaby (Macropus eugenii): Delayed B cell immunocompetence in marsupial neonates. Vet Immunol Immunopathol 136:235–247
Eddy SR (2009) A new generation of homology search tools based on probabilistic inference. Genome Informatics 23:205–211
Feigin CY et al (2018) Genome of the Tasmanian tiger provides insights into the evolution and demography of an extinct marsupial carnivore. Nat Ecol Evol 2:182–192. https://doi.org/10.1038/s41559-017-0417-y
Fuentes-Pardo AP, Ruzzante DE (2017) Whole-genome sequencing approaches for conservation biology: advantages, limitations and practical recommendations. Mol Ecol 26:5369–5406. https://doi.org/10.1111/mec.14264
Gaunitz C et al (2018) Ancient genomes revisit the ancestry of domestic and Przewalski’s horses. Science 360:111. https://doi.org/10.1126/science.aao3297
Genome 10K Community of Scientists (2009) Genome 10K: a proposal to obtain whole-genome sequence for 10 000 vertebrate species. J Hered 100:659–674
Govindaraj RG, Manavalan B, Lee G, Choi S (2010) Molecular modeling-based evaluation of hTLR10 and identification of potential ligans in Toll-like receptor signalling. PLoS one 5:e12713
Green EJ, Speller CE (2017) Novel substrates as sources of ancient DNA: prospects and hurdles Genes 8:7
Hajjar AM, Ernst RK, Tsai JH, Wilson CB, Miller SI (2002) Human Toll-like receptor 4 recognises host-specific LPS modifications. Nat Immunol 3:354–359
Hall T (1999) BioEdit: A user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hobbs M et al (2014) A transcriptome resource for the koala (Phascolarctos cinereus): insights into koala retrovirus transcription and sequence diversity. BMC Genomics 15:786
Huynen L, Millar CD, Lambert DM (2012) Resurrecting ancient animal genomes: the extinct moa and more. BioEssays 34:661–669
Janeway CA (2005) Immunobiology: the immune system in health and disease, 6th edn. Garland Science, New York
Jin MS et al (2007) Crystal structure of the TLR1-TLR2 heterodimer induced by binding of a tri-acylated lipopeptide. Cell 130:1071–1082. https://doi.org/10.1016/j.cell.2007.09.008
Johnson RN et al (2018) Adaptation and conservation insights from the koala genome. Nat Genet 50:1102–1111. https://doi.org/10.1038/s41588-018-0153-5
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Larkin M et al (2007) Clustal W and clustal X version 2.0. Bioinformatics 23:2947–2948
Larsen PA, Matocq MD (2019) Emerging genomic applications in mammalian ecology, evolution and conservation. J Mammal 100:786–801
Lewin H et al (2018) Earth BioGenome Project: Sequencing life for the future of life Proceedings of the National Academy of Sciences (PNAS) 115:4325–4333. https://doi.org/10.1073/pnas.1720115115
Li Y, Yin Y, Mariuzza RA (2013) Structural and biophysical insights into the role of CD4 and CD8 in T cell activation. Front Immunol 4:206
McDonough MM, Parker LD, McInerney NR, Campana MG, Maldonado JE (2018) Performance of commonly requested destructive museum samples for mammalian genomic studies. J Mammal 99:789–802
Mikkelsen TS et al (2007) Genome of the marsupial Monodelphis domestica reveals innovation in non-coding sequences. Nature 447:168–178
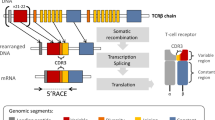
Miller RD (2010) Those other mammals: the immunoglobulins and T cell receptors of marsupials and monotremes. Semin Immunol 22:3–9
Miller W et al (2012) Polar and brown bear genomes reveal ancient admixture and demographic footprints of past climate change Proceedings of the National Academy of Sciences (PNAS) 109:E2382–E2390. https://doi.org/10.1073/pnas.1210506109
Mizel SB, West AP, Hantgan RR (2003) Identification of a sequence in human Toll-like receptor 5 required for the binding of gram-negative flagellin. J Biol Chem 278:23624–23629
Morris B, Cheng Y, Warren W, Papenfuss AT, Belov K (2015a) Identification and analysis of divergent immune gene families within the Tasmanian devil genome. BMC Genomics 16
Morris K et al (2014) The koala immunological toolkit: sequence identification and comparison of key markers of the koala (Phascolarctos cinereus) immune response Australian. J Zool 62:195–199
Morris KM, Matthew M, Waugh C, Ujvari B, Timms P, Polkinghorne A, Belov K (2015) Identification, characterisation and expression analysis of natural killer receptor genes in Chlamydia pecorum infected koalas (Phascolarctos cinereus). BMC Genomics 16:796
Morris KM, Wright B, Grueber CE, Hogg C, Belov K (2015) Lack of genetic diversity across diverse immune genes in an endangered mammal, the Tasmanian devil (Sarcophilus harrisii). Mol Ecol 24:3860–3872
Murchison EP et al (2012) Genome sequencing and analysis of the Tasmanian devil and its transmissible cancer. Cell 148:780–791
Murray GGR et al (2017) Natural selection shaped the rise and fall of passenger pigeon genomic diversity. Science 358:951–954
Nieswandt B, Watson SP (2003) Platelet-collagen interaction: is GPVI the central receptor? Blood 102:449–461
Old JM (2015) Immunological insights into the life and times of the extinct Tasmanian tiger (Thylacinus cynocephalus). PLoS one 10:e0144091
Paddle R (2000) The last Tasmanian tiger: the history and extinction of the thylacine. Cambridge University Press, Cambridge
Palkopoulou E et al (2015) Complete genomes reveal signatures of demographic and genetic declines in the woolly mammoth. Curr Biol 25:1395–1400. https://doi.org/10.1016/j.cub.2015.04.007
Papenfuss AT, Feng Z, Krasnec K, Deakin JE, Baker ML, Miller RD (2015) Marsupials and monotremes possess a novel family of MHC class I genes that is lost from the eutherian lineage. BMC Genomics 16:535
Park SDE et al (2015) Genome sequencing of the extinct Eurasian wild aurochs, Bos primigenius, illuminates the phylogeography and evolution of cattle. Genome Biol 16:234–234. https://doi.org/10.1186/s13059-015-0790-2
Parra ZE, Baker ML, Hathaway J, Lopez AM, Trujillo J, Sharp A, Miller RD (2008) Comparative genomic analysis and evolution of the T cell receptor loci in the opossum Monodelphis domestica. BMC Genomics 9:1–19
Parra ZE, Baker ML, Schwarz RS, Deakin JE, Lindblad-Toh K, Miller RD (2007) A unique T cell receptor discovered in marsupials. PNAS 104:9776–9781
Peel E and Belov K (2018) Marsupial immunology. In: Reference module in life sciences. Elsevier. ISBN: 978-0-12-809633-8, https://doi.org/10.1016/B978-0-12-809633-8.20719-X
Quach H, Quintana-Murci L (2017) Living in an adaptive world: genomic dissection of the genus Homo and its immune response. J Exp Med 214:877. https://doi.org/10.1084/jem.20161942
Renfree MB et al (2011) Genome sequence of an Australian kangaroo, Macropus eugenii, provides insight into the evolution of mammalian reproduction and development. Genome Biol 12:2–25
Roach JC et al (2005) The evolution of vertebrate Toll-like receptors PNAS 102:9577–9582
Romano TA, Ridgway SH, Felton DL, Quaranta V (1999) Molecular cloning and characterization of CD4 in an aquatic mammal, the while whale Delphinapterus leucas. Immunogenetics 49:376–383
Schraven AL, Stannard HJ, Ong OTW, Old JM (2020) Immunogenetics of marsupial B-cells Molecular Immunology 117:1–11. https://doi.org/10.1016/j.molimm.2019.10.024
Shapiro B (2017) Pathways to de-extinction: how close can we get to resurrection of an extinct species. Funct Ecol 31:996–1002
Siddle HV et al (2011) The tammar wallaby major histocompatability complex shows evidence of past genomic instability. BMC Genomics 12:421
Siddle HV, Marzec J, Chen Y, Jones M, Belov K (2010) MHC gene copy number variation in Tasmanian devils: implications for the spread of a contagious cancer Proc Royal Soc B 277:2001–2006
Sleightholme SR, Robovsky J, Vohralik V (2012) Description of four newly discovered thylacine pouch young and a comparison with Boardman (1945). Aust Zool 36:232–238
Suthers AN, Young LJ (2014) Isoforms of the CD79 signal transduction component of the macropod B-cell receptor. Dev Comp Immunol 47:185–190
Tyndale-Biscoe CH (2005) Life of marsupials. CSIRO Publishing, Collingwood
van der Kraan LE, Wong ESW, Lo N, Ujvari B, Belov K (2013) Identification of natural killer cell receptor genes in the genome of the marsupial Tasmanian devil (Sarcophilus harrisii). Immunogenetics 65:25–35
Veillette A, Bookman MA, Horak EM, Samelson LE, Bolen JB (1989) Signal transduction through the CD4 receptor involved the activation of the internal membrane tyrosine-protein kinase p56lck. Nature 338:257–259
Wang X, Olp JJ, Miller RD (2009) On the genomics of immunoglobulins in the gray, short-tailed opossum Monodelphis domestica. Immunogenetics 61:581–596
Wei T, Gong J, Jamitzky F, Heckl WM, Stark RW, Rossle SC (2009) Homology modeling of human Toll-like receptors TLR7, 8 and 9 ligand-binding domains. Protein Sci 18:1664–1691
Willerslev E, Cooper A (2005) Ancient DNA Proceedings of the Royal Society B 272:3–16
Williams A, Peh CA, Elliot T (2002) The cell biology of MHC class I antigen presentation Tissue Anatomy 59:3–17
Wong ESW, Papenfuss AT, Belov K (2011) Genomic Identification of Chemokines and Cytokines in Opossum. J Interferon Cytokine Res 31:317–330
Wong ESW, Young LJ, Papenfuss AT, Belov K (2006) In silico identification of opossum cytokine genes suggest the complexity of the marsupial immune system rivals that of eutherian mammals. Immunome Research 2:4
Yin L et al (2011) A single T cell receptor bound to major histocompatibility complex class I and class II glycoproteins reveals switchable TCR conformers. Immunity 35:23–33. https://doi.org/10.1016/j.immuni.2011.04.017
Zelensky AN, Gready JE (2005) The C-type lectin-like domain superfamily FEBS Journal 272:6179–6217
Funding
KB and EP are supported by an Australian Research Council Discovery grant to KB DP 180102465. AP was supported by an Australian Research Council Future Fellowship FT140100964.
Author information
Authors and Affiliations
Contributions
KB and EP designed the study. AP and SF provided access to the thylacine genome. SF performed preliminary BLAST searches. EP conducted BLAST and HMMER searchers, characterised all immune genes, and constructed all phylogenetic trees. KB and CJH provided feedback on drafts and revised the paper. All authors read and commented on the paper.
Corresponding author
Ethics declarations
Consent for publication
All authors read and approved the manuscript.
Conflict of interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Peel, E., Frankenberg, S., Hogg, C.J. et al. Annotation of immune genes in the extinct thylacine (Thylacinus cynocephalus). Immunogenetics 73, 263–275 (2021). https://doi.org/10.1007/s00251-020-01197-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-020-01197-z