Abstract
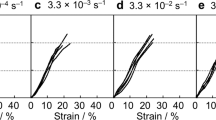
We have investigated the mechanical properties of spider dragline fibers of three Nephila species under varied relative humidity. Force maps have been collected by atomic force microscopy. The Young’s modulus E was derived from the indentation curves of each pixel by the modified Hertz model. An average decrease in E by an order of magnitude was observed upon immersion of the fiber in water. Single fiber stretching experiments were carried out for comparison, and also showed a strong dependence on relative humidity. However, the absolute values of E are significantly higher than those obtained by indentation. The results of this work thus show that the elastic properties of spider silk are highly anisotropic, and that the silk softens significantly for both tensile and compressional strain (indentation) upon water uptake. In addition, the force maps indicate a surface structure on the sub-micron scale.





Similar content being viewed by others
References
Arne Schäfer (2003) Untersuchung mechanischer Eigenschaften von Zellen mit dem Kraftmikroskop - Einfluss von Myosin II. Disseration. Georg-August-Universität Göttingen
Becker N, Oroudjev E, Mutz S, Cleveland JP, Hansma PK, Hayashi CY, Makarov DE, Hannma HG (2003) Molecular nanosprings in spider capture-silk threads. Nat Mater 2:278–283
Bell FI, McEwen IJ, Viney C (2002) Fibre science: supercontraction stress in wet spider dragline. Nature 416:37
Binnig G, Quate CF, Gerber C (1986) Atomic force microscope. Phys Rev Lett 56:930–933
Cleveland JP, Radmacher M, Hansma PK (1994) NATO advanced research workshop AIP. Schluchsee, Germany, pp 543–549
Cunniff PM, Fossey SA, Auerbach MA, Song JW (1994) Mechanical properties of major ampullate gland filk fibers extracted from Nephilia clavipes spiders. Silk polymers: materials science and biotechnology, Chap. 21
Dai L, Zhang Y, Ou-Yang Z (2003) Elastic theory of single spider silk protein molecule. Thin Solid Films 438–439:382–385
Domke J (2000) Mikromechanische Eigenschaften dünner Polymerfilme und lebender Zellen. Dissertation. Ludwig-Maximilian-Universität München
Domke J, Rachmacher M (1998) Measuring the elastic properties of thin polymer films with the atomic force microscope. Langmuir 14:3320–3325
Drake B, Prater CB, Weisenhorn AL, Gould SAC, Albrecht TR, Quate CF, Cannell DS, Hansma HG, Hansma PK (1989) Imaging crystals, polymers and biological processes in water with AFM. Science 243:1586–1589
Elices M, Pérez-Rigueiro J, Plaza GR, Guinea GV (2005) Finding inspiration in Argiope trifasciata spider silo fibers. J Mater 57:60–66
Fossey SA, Kaplan DL (1999) Silk protein. In: Polymer data handbook. Oxford University Press, Oxford
Fritz M, Radmacher M, Gaub HE (1994) Granula motion and membrane spreading during activation of human platelets imaged by AFM. Biophys J 66:1328–1334
Glišović A, Thieme J, Guttmann P, Salditt T (2007) Transmission X-ray microscopy of spider dragline silk. Int J Biol Macromol 40:87–95
Gosline JM, Pollak CC, Guerette PA, Cheng A, DeMont ME, Denny MW (1994) Elastomeric network models for the frame and viscid silks from the orb web of the spider Araneus diadematus. Silk polymers: materials science and biotechnology. In: Kaplan D, Adams WW, Farmer B, Viney C (eds). American Chemical Society, Washington, DC, pp 328–341
Gosline JM, Guerette PA, Ortlepp CS, Savage KN (1999) The mechanical design of spider silks: from Fibroin sequence to mechanical function. J Exp Biol 202:3295–3303
Grubb DT, Jelinski LW (1997) Small angle X-ray scattering of spider dragline silk. Marcomolecules 30:2860–2867
Hämmerlin G, Hoffmann K-H (1989) Numerische Mathematik. Springer, Berlin
Hertz H (1882) Über die Berührung fester elastischer Körper. Reine Angew Mathematik 92:156–171
Hinman MB, Lewis RV (1992) Isolation of a clone encoding a second dragline silk fibroin, Nephila Clavipes dragline silk is a two protein fiber. J Biol Chem 267:19320–19324
Johnson KL (1994) Contact mechanics. Cambridge University Press, Cambridge
Kesel AB, Martin A, Seidl T (2004) Getting a grip on spider attachment: an AFM approach to microstructure adhesion in arthropods. Smart Mater Struct 13:512–518
Kishore AI, Herberstein ME, Craig CL, Separovic F (2002) Solid-state NMR relaxation studies of Australian spider silks. Biopolymers 61:287–297
Landau LD, Lifschitz EM (1965) Band 7 Elastizitätstheorie. Lehrbuch der theoretischen Physik. Verlag Harri Deutsch, Frankfurt am Main
Magoshi J, Magoshi Y, Nakamura S (1985) Physical properties and structure of silk. Polym Comm 26:309–311
Pèrez-Rigueiro J, Viney C, Llorca J, Elices M (1998) Silkworm silk as an engineering material. J Appl Polym Sci 70:2439–2447
Pérez-Rigueiro J, Elices M, Llorca M, Viney C (2001) Tensile properties of Argiope trifasciata drag line silk obtained from the spider’s web. J Appl Polym Sci 82:2245–2251
Plaza GR, Guinea GV, Pérez-Rigueiro J, Elices M (2006) Thermo-hygro-mechanical behaviour of spider dragline silk: glassy and rubbery states. J Polym Sci 44:994–999
Putthanarat S, Stribeck N, Fossey SA, Eby RK, Adams WW (2000) Investigation of the nanofibrils of silk fibers. Polymer 41:7735–7747
Radmacher M (1999) Single molecus feel the force. Phys World 12(9):33–37
Radmacher M, Fritz M, Hansma HG, Hansma PK (1994a) Direct observation of enzyme activity with the atomic force microscope. Science 265:1577–1579
Radmacher M, Cleveland JP, Fitz M, Hansma HG, Hansma PK (1994b) Mapping interaction forces with the atomic force microscope. Biophys J 66:2159–2165
Radmacher M, Fritz M, Hansma PK (1995) Imaging soft samples with the atomic force microscope: gelatin in water and propanol. Biophys J 69:264–270
Radmacher M, Fritz M, Kacher MK, Cleveland JP, Hansma PK (1996) Measuring the elastic properties of human platelets with the atomic force microscope. Biophys J 70:556–567
Riekel C, Vollrath F (2001). Spider silk fibre extrusion: combined wide- and small-angle X-ray microdiffraction experiments. Int J Biol Macromol 29:203–210
Riekel C, Müller M, Vollrath F (1999) In situ X-ray diffraction during forced silking of spider silk. Macromolecules 32:4464–4466
Rotsch C, Jacobson K, Radmacher M (1999) Dimensional and mechanical dynamics of active and stable edges in motile fibroblasts investigated by atomic force microscopy. Proc Natl Acad Sci USA 96:921–926
Schäfer A, Radmacher M (2005) Influence of myosin II activity on stiffness of fibroblast cells. Acta Biomaterialia 1:273–280
Shao Z, Vollrath F (1999) The effect of solvents on the contraction and mechanical properties of spider silk. Polymer 40:1799–1806
Sneddon IN (1965) The relation between load and penetration in the axisymmetric Boussinesq problem for a punch of arbitrary profile. Int J Eng Sci 3:47–57
Tao NJ, Lindsay SM, Lees S (1992) Measuring the microelastic properties of biological material. Biophys J 63:1165–1169
Vollrath F, Edmonds DT (1989) Modulation of the mechanical properties of spider silk by coating with water. Nature 340:305–307
Vollrath F, Knight DP (2001) Liquid crystalline spinning of spider silk. Nature 410:541–548
Vollrath F, Porter D (2006) Spider silk as archetypal protein elastomer. Soft Matter 2:377–385
Weisenhorn AL, Khorsandi M, Kasas S, Gotozos V, Celio MR, Butt HJ (1993) Deformation and height anomaly of soft surfaces studied with the AFM. Nanotechnology 4:106–113
Work RW, Emerson PD (1982) An apparatus and technique for the forcible silking of spiders. J Arachnol 10:1–10
Xu M, Lewis RV (1990) Structure of a protein superfiber: spider dragline silk. Proc Natl Acad Sci 87:7120–7124
Zhou H, Zhang Y (2005) Hierarchical chain model of spider capture silk elasticity. Phys Rev Lett 94:028104-1–028104-4
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schäfer, A., Vehoff, T., Glišović, A. et al. Spider silk softening by water uptake: an AFM study. Eur Biophys J 37, 197–204 (2008). https://doi.org/10.1007/s00249-007-0216-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-007-0216-5