Abstract
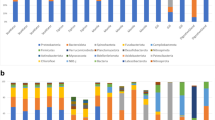
Marine invertebrate microbiota has a key function in host physiology and health. To date, knowledge about bivalve microbiota is poorly documented except public health concerns. This study used a molecular approach to characterize the microbiota associated with the bivalve Manila clam (Ruditapes philippinarum) by determining (1) the difference among organs either or not under the influence of host habitat, (2) small-scale variability of microbiota, and (3) the experimental response of the Manila clam microbiota submitted to different lateral transmissions. These questions were investigated by sampling two groups of individuals living in contrasting habitats and carrying out a transplant experiment. Manila clam microbiota (i.e., bacterial community structure) was determined at organ-scale (gills, gut, and a pool of remaining tissues) by capillary electrophoresis DNA fingerprinting (CE fingerprinting). The Manila clam microbiota structure differed among organs indicating a selection of Manila clam microbiota at organ scale. Habitat strongly influenced gill and gut microbiota. In contrast, microbiota associated with remaining tissues was similar between group individuals suggesting that these communities are mostly autochthonous, i.e., Manila clam specific. Transplant experiment showed that improving living condition did not induce any change in microbiota associated with remaining tissues. In contrast, the reduction in individual habitat quality led to individuals in declining health as strongly suggested by the increase in phagocytosis activity and decrease in condition index together with the change in internal organ microbiota. This study provides a first description of the Manila clam holobiont which can withstand disturbance and respond opportunistically to improved environmental conditions.






Similar content being viewed by others
References
Noda S, Iida T, Kitade O, Nakajima H, Kudo T, Ohkuma M (2005) Endosymbiotic Bacteroidales bacteria of the flagellated protist Pseudotrichonympha grassii in the gut of the termite Coptotermes formosanus. Appl Environ Microbiol 71:8811–8817
Kuo RC, Lin S (2013) Ectobiotic and endobiotic bacteria associated with Eutreptiella sp. isolated from long island sound. Protist 164:60–74
Gnanamanickam SS (2007) Plant-associated bacteria. Springer
Redford AJ, Bowers RM, Knight R, Linhart Y, Fierer N (2010) The ecology of the phyllosphere: geographic and phylogenetic variability in the distribution of bacteria on tree leaves. Environ Microbiol 12:2885–2893
McFall-Ngai MJ (2002) Unseen forces: the influence of bacteria on animal development. Dev Biol 242:1–14
Ruby E, Henderson B, McFall-Ngai M (2004) We get by with a little help from our (little) friends. Science 303:1305–1307
Zilber-Rosenberg I, Rosenberg E (2008) Role of microorganisms in the evolution of animals and plants: the hologenome theory of evolution. FEMS Microbiol Rev 32:723–35
Lafferty KD, Porter JW, Ford SE (2004) Are diseases increasing in the ocean? Annu Rev Ecol Evol Syst 35:31–54. doi:10.1146/annurev.ecolsys.35.021103.105704
Plowright RK, Sokolow SH, Gorman ME, Daszak P, Foley JE (2008) Causal inference in disease ecology: investigating ecological drivers of disease emergence. Front Ecol Environ 6(8):420–429. doi:10.1890/070086
Bright M, Bulgheresi S (2010) A complex journey: transmission of microbial symbionts. Nature Rev Microbiol 8:218–230
Taylor MW, Radax R, Steger D, Wagner M (2007) Sponge-associated microorganisms: evolution, ecology, and biotechnological potential. Microbiol Mol Biol Rev 71:295–347
Zurel D, Benayahu Y, Or A, Kovacs A, Gophna U (2011) Composition and dynamics of the gill microbiota of an invasive indo-pacific oyster in the eastern Mediterranean Sea. Environ Microbiol 13:1467–1476
Dubilier N, Bergin C, Lott C (2008) Symbiotic diversity in marine animals: the art of harnessing chemosynthesis. Nature Rev Microbiol 6:725–740
Tanaka R, Ootsubob M, SawabecT EY, Tajimac K (2004) Biodiversity and in situ abundance of gut microflora of abalone (Haliotis discus hannai) determined by culture-independent techniques. Aquaculture 241:453–463
Beleneva N, Zhukova V (2009) Seasonal dynamics of cell numbers and biodiversity of marine heterotrophic bacteria inhabiting invertebrates and water ecosystems of the peter the great Bay, Sea of Japan. Microbiology 78:369–375
Dame RF (1996) Ecology of marine bivalves: an ecosystem approach, CRC Marine Science, CRC press, p. 254
Pruzzo C, Gallo G, Canesi L (2005) Persistence of vibrios in marine bivalves: the role of interactions with haemolymph components. Environ Microbiol 7:761–772
Antunes F, Hinzmann M, Lopes-Lima M, Machado J, da Costa PM (2010) Association between environmental microbiota and indigenous bacteria found in hemolymph, extrapallial fluid and mucus of Anodonta cygnea (Linnaeus, 1758). Microb Ecol 60:304–309
Rosenberg E, Koren O, Reshef L, Efrony R, Rosenberg LZ (2007) The role of microorganisms in coral health, disease and evolution. Nat Rev Microbiol 355:349–360
Webster NS, Blackall LL (2009) What do we really know about sponge-microbial symbioses? The ISME J 3:1–3
Rohwer F, Seguritan V, Azam F, Knowlton N (2002) Diversity and distribution of coral-associated bacteria. Mar Ecol Prog Ser 243:1–10
Hansson L, Agis M, Maier C, Weinbauer MG (2009) Community composition of bacteria associated with cold-water coral Madrepora oculata: within and between colony variability. Mar Ecol Prog Ser 397:89–102
Porporato EMD, Lo Giudice A, Michaud L, De Domenico E, Spanò N (2013) Diversity and antibacterial activity of the bacterial communities associated with Two Mediterranean Sea pens, Pennatula phosphorea and Pteroeides spinosum (Anthozoa: Octocorallia). Microb Ecol 66:701–714
Frias-Lopez J, Zerkle AL, Bonheyo GT, Fouke BW (2002) Partitioning of bacterial communities between seawater and healthy, black band diseased and dead coral surfaces. Appl Environ Microbiol 68:2214–2228
Cárdenas CA, Bell JJ, Davy SK, Hoggard M, Taylor MW (2014) Influence of environmental variation on symbiotic bacterial communities of two temperate sponges. FEMS Microbiol Ecol. doi:10.1111/1574-6941.12317
Neulinger SC, Gartner A, Jarnegren J, Ludvigsen M, Lochte K, Dullo WC (2009) Tissue-associated “candidatus Mycoplasma corallicola” and filamentous bacteria on the cold-water coral Lophelia pertusa (Scleractinia). Appl Environ Microbiol 75:1437–1444
Sweet MJ, Croquer A, Bythell JC (2011) Bacterial assemblages differ between compartments within the coral holobiont. Coral Reefs 30:39–52
Fan L, Liu M, Simister R, Webster NS, Thomas T (2013) Marine microbial symbiosis heats up: the phylogenetic and functional response of a sponge holobiont to thermal stress. The ISME J 7:991–1002
Olson JB, Thacker RW, Gochfeld DJ (2014) Molecular community profiling reveals impacts of time, space, and disease status on the bacterial community associated with the Caribbean sponge Aplysina cauliformis. FEMS Microbiol Ecol 87:268–79. doi:10.1111/1574-6941.12222
Klaus JS, Frias-Lopez J, Bonheyo GT, Heikoop JM, Fouke BW (2005) Bacterial communities inhabiting the healthy tissues of two Caribbean reef corals: interspecific and spatial variation. Coral Reefs 24:129–137
Littman RA, Willis BL, Pfeffer C, Bourne DG (2009) Diversities of coral-associated bacteria differ with location, but not species, for three acroporid corals on the Great Barrier Reef. FEMS Microbiol Ecol 68:152–163
Rodriguez-Lanetty M, Granados-Cifuentes C, Barberan A, Bellantuono A, Bastidas C (2013) Ecological inferences from a deep screening of the complex bacterial consortia associated with the coral, porites astreoides. Mol Ecol 22:4349–62. doi:10.1111/mec.12392
Karlińska-Batres K, Wörheide G (2013) Phylogenetic diversity and community structure of the symbionts associated with the coralline sponge Astrosclera willeyana of the great barrier reef. Microb Ecol 65:740–752
Pita L, López-Legentil S, Erwin PM (2013) Biogeography and host fidelity of bacterial communities in Ircinia spp. From the Bahamas. Microb Ecol 66:437–447
Schöttner S, Hoffmann F, Wild C, Rapp HT, Boetius A, Ramette A (2009) Inter- and intra-habitat bacterial diversity associated with cold-water corals. ISME J 3:756–759
Cleary DFR, Becking LE, de Voogd NJ, Pires ACC, Polonia ARM, Egas C, Gomes NMC (2013) Habitat- and host-related variation in sponge bacterial symbiont communities in Indonesian waters. FEMS Microbiol Ecol 85:465–482
Charlotte E, Kvennefors E, Sampayo E, Ridgway T, Barnes AC, Hoegh-Guldberg O (2010) Bacterial communities of two ubiquitous great barrier reef corals reveals both site- and species-specificity of common bacterial associates. PLoS ONE 5(4), e10401
FAO (2007) The state of world fisheries and aquaculture, Fisheries and Aquaculture Department, Food and Agriculture. Organization of the United Nations, Rome
Caill-Milly, N, Bobinet, J, Lissardy, M, Morandeau, G, Sanchez, F (2008) Campagne d'évaluation du stock de palourdes du bassin d'Arcachon. Année 2008. In: IFREMER. pp. 1–67
Caill-Milly N, de Casamajor, MN, Lissardy M, Sanchez F, Morandeau G (2003) Evaluation du stock de palourdes du bassin d'Arcachon—Campagne 2003. In: IFREMER. pp. 1–44
Caill-Milly N, Duclercq B, Morandeau G (2006) Campagne d'évaluation du stock de palourdes du bassin d'Arcachon. In: IFREMER. pp. 1–51
Lassalle G, de Montaudouin X, Soudant P, Paillard C (2007) Parasite co-infection of two sympatric bivalves, the Manila clam (Ruditapes philippinarum) and the cockle (Cerastoderma edule) along a latitudinal gradient. Aquat Living Resour 20:33–42
Dang C, de Montaudouin X, Gonzalez P, Mesmer-Dudons N, Caill-Milly N (2008) Brown Muscle Disease (BMD), an emergent pathology affecting Manila clam Ruditapes philippinarum in Arcachon Bay (SW France). Dis Aquat Org 80:219–228
Dang C, de Montaudouin X, Gam M, Paroissin C, Caill-Milly N (2010) The Manila clam population in Arcachon Bay (SW France): can it be kept sustainable? J Sea Res 63:108–118
Meisterhans G, Raymond N, Lebreton S, Salin F, Bourasseau L, de Montaudouin X, Garabetian F, Jude-Lemeilleur F (2011) Dynamics of bacterial bommunities in bockles (Cerastoderma edule) with respect to trematode parasite (Bucephalus minimus) infestation. Microb Ecol 62:620–631
SOMLIT, The French coastal monitoring network (Service d’Observation en Milieu LITtoral; INSU/CNRS; http://somlit.epoc.u-bordeaux1.fr
Dang C, Sauriau PG, Savoye N, Caill-Milly N, Martinez P, Millaret C, Haure J, de Montaudouin X (2009) Determination of diet in Manila clams by spatial analysis of stable isotopes. Mar Ecol Prog Ser 387:167–177
Walne PR, Mann R (1975) Growth and biochemical composition Ostrea edulis and Crassostrea gigas. In: H Barnes (ed) Proc. 9th European Marine Biology Symposium. Oban. Aberdeen University Press, Aberdeen, pp. 587–607
Delaporte M, Soudant P, Moal J, Lambert C, Quéré C, Miner P, Choquet G, Paillard C, Samain JF (2003) Effect of a mono-specific algal diet on immune functions in two bivalve species—Crassostrea gigas and Ruditapes philippinarum. J Exp Biol 206:3053–3064
Kennedy P, Kennedy H, Papadimitriou S (2005) The effect of acidification on the determination of organic carbon, total nitrogen and their stable isotopic composition in algae and marine sediment. Rap Com Mass Spectrom 19:1063–1068
Zhou J, Bruns MA, Tiedje JM (1996) DNA recovery from soils of diverse composition. Appl Environ Microbiol 62:316–322
Normand P, Ponsonnet C, Nesme X, Neyra M, Simonet P (1996) ITS analysis of prokaryotes. In: Akkermans DL, van Elsas JD, de Bruijn FJ (eds) Molecular microbial ecology manual. Kluwer Academic Publishers, Netherlands, pp 1–12
Osborne CA, Rees GN, Bernstein Y, Janssen PH (2006) New threshold and confidence estimates for terminal restriction fragment length polymorphism analysis of complex bacterial communities. Appl Environ Microbiol 72:1270–8
Ramette A (2009) Quantitative community fingerprinting methods for estimating the abundance of operational taxonomic units in natural microbial communities. Appl Environ Microbiol 75:2495–2505
Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation, 2nd edition. PRIMER-E: Plymouth
Kropf S, Heuer H, Grüning M, Smalla K (2004) Significance test for comparing complex microbial community fingerprints using pairwise similarity measures. J Microbiol Methods 57:187–195
Ramette A (2007) Multivariate analyses in microbial ecology. FEMS Microbiol Ecol 62:142–160
Siegel S, Castellan NJ (1988) Non parametric statistics for the behavioral sciences, 2nd edn. Mc Graw-Hill, New York
Humphreys J, Caldow RWG, McGrorty S, West A.D, Jensen AC (2007). Population dynamics of naturalised Manila clams Ruditapes philippinarum in British coastal waters. Mar. Biol. 151: 2255–2270. In: Marine Biology. Springer: Berlin. ISSN 0025–3162
Ren Y, Xu B, Guo Y, Yang M, Yang J (2008) Growth, mortality and reproduction of the transplanted Manila clam (Ruditapes philippinarum, Adams & Reeve 1850) in Jiaozhou Bay. Aqua Res 39:1759–1768
Marie B, Trinkler N, Zanella-Cléon I, Guichard N, Becchi M, Paillard C, Marin F (2011) Proteomic identification of novel proteins from the calcifying shell matrix of the Manila clam Venerupis philippinarum. Mar Biotechnol 13:955–962
Dang C, de Montaudouin X (2009) Brown muscle disease and Manila clam Ruditapes philippinarum dynamics in Arcachon Bay. France J Shellfish Res 28:355–362
Dang C, de Montaudouin X, Gonzalez P, Mesmer-Dudons N, Caill-Milly N (2008) Description of a new pathology affecting the adductor muscle of manila clam (Ruditapes philippinarum) in Arcachon Bay (SW France). J Shellfish Res 27:1000–1001
Ray SM (1966) A review of the culture method of detecting Dermocystidium marinum with suggested modifications and precautions. Proc Nat Shell Assoc 54:55–69
Dang C, de Montaudouin X, Caill-Milly N, Trumbiç E (2010) Spatio-temporal patterns of perkinsosis in the Manila clam Ruditapes philippinarum from Arcachon Bay (SW France). Dis Aqua Org 91:151–159
Prieur D, Mevel G, Nicolas J, Plusquellec A, Vigneulle M (1990) Interactions between bivalve molluscs and bacteria in the marine environment. Oceanogra Mar Biol 28:277–352
Meziti A, Ramette A, Mente E, Kormas KA (2010) Temporal shifts of the Norway lobster (Nephrops norvegicus) gut bacterial communities. FEMS Microbiol Ecol 74:472–484
Canesi L, Gallo G, Gavioli M, Pruzzo C (2002) Bacteria-hemocyte interactions and phagocytosis in marine bivalves. Microsc Res Tech 57:469–476
Ellis RP, Parry H, Spicer JI, Hutchinson TH, Pipe RK, Widdicombe S (2011) Immunological function in marine invertebrates: responses to environmental perturbation. Fish Shellfish Immunol 30:1209–1222
Romero J, García-Varela M, Laclette JP, Espejo RT (2002) Bacterial 16S rRNA gene analysis revealed that bacteria related to Arcobacter spp. constitute an abundant and common component of the oyster microbiota (Tiostrea chilensis). Microb Ecol 44:365–371
Auby I, Bost CA, Budzinski H, Dalloyau S, Desternes A, Belles A, Trut G, Plus M, Pere C, Couzi L, Feigne C, Steinmetz J (2011) Régression des herbiers de zostères dans le Bassin d’Arcachon: état des lieux et recherche des causes. http://archimer.ifremer.fr/doc/00054/16507/
Le Treut Y (1986) La palourde. Anatomie - Biologie - Elevage - Pêche - Consommation - Inspection sanitaire. Thèse de Doctorat, Ecole Nationale Vétérinaire de Nantes. pp. 161
Harris JM (1993) The presence, nature, and role of gut microflora in aquatic invertebrates: a synthesis. Microb Ecol 25:195–231
Gros O, Gaill F (2007) Extracellular bacterial association in gills of “wood mussels”. Cah Biol Mar 48:103–109
Distel DL, DeLong EF, Waterbury JB (1991) Phylogenetic characterization and in situ localization of the bacterial symbiont of shipworms (Teredinidae: Bivalvia) by using 16S rRNA sequence analysis and oligodeoxynucleotide probe hybridization. Appl Environ Microbiol 57:2376–2382
Gros O, Darrasse A, Durand P, Frenkiel L, Moueza M (1996) Environmental transmission of a sulfur-oxidizing bacterial gill endosymbiont in the tropical lucinid bivalve Codakia orbicularis. Appl Environ Microbiol 62:2324–2330
Halary S, Riou V, Gaill F, Boudier T, Duperron S (2008) 3D FISH for the quantification of methane- and sulphur-oxidizing endosymbionts in bacteriocytes of the hydrothermal vent mussel Bathymodiolus azoricus. ISME J 2:284–92. doi:10.1038/ismej.2008.3
De Montaudouin X, Paul-Pont I, Lambert C, Gonzalez P, Raymond N, Jude F, Legeay A, Baudrimont M, Dang C, Le Grand F, Le Goïc N, Bourasseau L, Paillard C (2010) Bivalve population health: multistress to identify hot spots. Mar Pol Bull 60:1307–1318
Paul-Pont I, Gonzalez P, Baudrimont M, Jude F, Raymond N, Bourrasseau L, Le Goïc N, Haynes F, Legeay A, Paillard C, de Montaudouin X (2010) Interactive effects of metal contamination and pathogenic organisms on the marine bivalve Cerastoderma edule. Mar Pol Bull 60:515–525
Reid HI, Soudant P, Lambert C, Paillard C, Birkbeck TH (2003) Salinity effects on immune parameters of Ruditapes philippinarum challenged with Vibrio tapetis. Dis Aquat Org 56:249–258
Hégaret H, da Silva PM, Wikfors GH, Lambert C, De Bettignies T, Shumway SE, Soudant P (2007) Hemocyte response of Manila clams, Ruditapes philippinarum, with varying parasite, Perkinsus olseni, severity to toxic-algal exposures. Aquat Toxicol 84:469–479
Yu JH, Song JH, Choi MC, Park SW (2009) Effects of water temperature change on immune function in surf clams, Mactra veneriformis (Bivalvia: Mactridae). J Invertebrate Pathol 102:30–35
Acknowledgments
This project is part of the REPAMEP project funding by LITEAU 3 (nL11-6778), a program of the French Ministry for Environment. The authors thank the shellfish industry CODIMER (Gujan-Mestras, 33-France) for having placed water bath used in the depuration process at our disposal. We thank the crew of R/V Planula IV (INSU) Francis Prince and Laurent Letort for help with clam sampling, and Andrea Niemi (Fisheries and Oceans Canada) for having improved the English.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meisterhans, G., Raymond, N., Girault, E. et al. Structure of Manila Clam (Ruditapes philippinarum) Microbiota at the Organ Scale in Contrasting Sets of Individuals. Microb Ecol 71, 194–206 (2016). https://doi.org/10.1007/s00248-015-0662-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-015-0662-z