Abstract
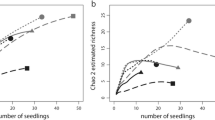
Ectomycorrhizal fungal (EMF) propagules play an important role in seedling establishment following disturbance. However, little is known about how the EMF propagule community changes with forest development. In this study, EMF propagules were examined using seedling bioassays in rhizosphere soils collected from a recently closed Pb–Zn tailing (Taolin Pb–Zn tailing (TLT)), a Cu tailing (Dexing Cu No. 2 tailing (DXT)) that had undergone 21 years of restoration, and a mature Masson pine (Pinus massoniana) forest (DXC) outside the Cu mining areas. The corresponding EMF communities colonizing Masson pine at each site were also investigated for comparison. After 8 months of running bioassays, ectomycorrhizal colonization was poor for seedlings grown in TLT (9.0 % ± 14.9 %) and DXT soils (22.4 % ± 17.7 %), while DXC seedlings were well colonized (47.5 % ± 24.9 %). Internal transcribed spacer sequencing revealed that EMF species richness increased with forest development in both the propagule bank (TLT, 6; DXT, 7; DXC, 12) and in the field (TLT, 8; DXT, 14; DXC, 26), though richness was lower in propagule banks. Several lineages, such as Cenococcum, Rhizopogon, Inocybe, Suillus, and Atheliaceae, were frequently encountered in propagule communities, but species assemblages were different among the three sites. Canonical correspondence analysis revealed that several soil parameters, i.e., N, EC, Cu, Pb, Zn, etc., were responsible for the distribution of EMF in the field and bioassay seedlings. The highest overlap in EMF species composition between the propagule bank and the field community was observed at the recently closed tailing (Morisita–Horn similarity = 0.71 for TLT), whereas the lowest overlap occurred at the mature forest (0.26 for DXC). These results indicate that EMF propagules in soil are less frequent and diverse in early primary succession and become more frequent and diverse along forest development, due mainly to the accumulation of dormant spores of Rhizopogon spp. and sclerotia of Cenococcum spp. Thus, EMF propagule communities in soil may diverge from those root-colonizing EMF communities along a gradient of forest development.






Similar content being viewed by others
References
Smith S, Read D (2008) Mycorrhizal symbiosis, 3rd edn. Academic, New York
Nara K (2006) Ectomycorrhizal networks and seedling establishment during early primary succession. New Phytol 169:169–178
Peay KG, Kennedy PG, Bruns TD (2011) Rethinking ectomycorrhizal succession: are root density and hyphal exploration types drivers of spatial and temporal zonation? Fungal Ecol 4:233–240
Nara K, Hogetsu T (2004) Ectomycorrhizal fungi on established shrubs facilitate subsequent seedling establishment of successional plant species. Ecology 85:1700–1707
Baar J, Horton TR, Kretzer AM, Bruns TD (1999) Mycorrhizal colonization of Pinus muricata from resistant propagules after a stand-replacing wildfire. New Phytol 143:409–418
Ishida TA, Nara K, Tanaka M, Kinoshita A, Hogetsu T (2008) Germination and infectivity of ectomycorrhizal fungal spores in relation to their ecological traits during primary succession. New Phytol 180:491–500
Bruns TD, Peay KG, Boynton PJ, Grubisha LC, Hynson NA et al (2009) Inoculum potential of Rhizopogon spores increases with time over the first 4 yr of a 99-yr spore burial experiment. New Phytol 181:463–470
Nguyen NH, Hynson NA, Bruns TD (2012) Stayin' alive: survival of mycorrhizal fungal propagules from 6-yr-old forest soil. Fungal Ecol 5:741–746
Izzo A, Canright M, Bruns TD (2006) The effects of heat treatments on ectomycorrhizal resistant propagules and their ability to colonize bioassay seedlings. Mycol Res 110:196–202
Peay KG, Garbelotto M, Bruns TD (2009) Spore heat resistance plays an important role in disturbance-mediated assemblage shift of ectomycorrhizal fungi colonizing Pinus muricata seedlings. J Ecol 97:537–547
Taylor DL, Bruns TD (1999) Community structure of ectomycorrhizal fungi in a Pinus muricata forest: minimal overlap between the mature forest and resistant propagule communities. Mol Ecol 8:1837–1850
Ashkannejhad S, Horton TR (2006) Ectomycorrhizal ecology under primary succession on coastal sand dunes: interactions involving Pinus contorta, suilloid fungi and deer. New Phytol 169:345–354
Buscardo E, Rodriguez-Echeverria S, Martin MP, De Angelis P, Pereira JS et al (2010) Impact of wildfire return interval on the ectomycorrhizal resistant propagules communities of a Mediterranean open forest. Fungal Biol 114:628–636
Mendez M, Maier R (2008) Phytoremediation of mine tailings in temperate and arid environments. Rev Environ Sci Biotechnol 7:47–59
Huang J, Nara K, Lian C, Zong K, Peng K et al (2012) Ectomycorrhizal fungal communities associated with Masson pine (Pinus massoniana Lamb.) in Pb–Zn mine sites of central south China. Mycorrhiza 22:589–602
Guo G, Yuan T, Wang W, Li D, Cheng J et al (2011) Bioavailability, mobility, and toxicity of Cu in soils around the Dexing Cu mine in China. Environ Geochem Health 33:217–224
Lian C, Narimatsu M, Nara K, Hogetsu T (2006) Tricholoma matsutake in a natural Pinus densiflora forest: correspondence between above- and below-ground genets, association with multiple host trees and alteration of existing ectomycorrhizal communities. New Phytol 171:825–836
White TJ, Bruns TD, Lee S, Taylor J (1990) Analysis of phylogenetic relationships by amplification and direct sequencing of ribosomal RNA genes. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic, San Diego, pp 315–322
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Koljalg U, Larsson KH, Abarenkov K, Nilsson RH, Alexander IJ et al (2005) UNITE: a database providing web-based methods for the molecular identification of ectomycorrhizal fungi. New Phytol 166:1063–1068
Colwell RK (2006) EstimateS: statistical estimation of species richness and shared species from samples. Version 8.0
Dixon P (2003) VEGAN, a package of R functions for community ecology. J Veg Sci 14:927–930
R, Development, Core, Team (2006) R: a language and environment for statistical computing. http://www.Royal-project.org
Jones MD, Durall DM, Cairney JWG (2003) Ectomycorrhizal fungal communities in young forest stands regenerating after clearcut logging. New Phytol 157:399–422
Colpaert JV, Wevers JHL, Krznaric E, Adriaensen K (2011) How metal-tolerant ecotypes of ectomycorrhizal fungi protect plants from heavy metal pollution. Ann For Sci 68:17–24
Wang Q, Guo L-D (2010) Ectomycorrhizal community composition of Pinus tabulaeformis assessed by ITS-RFLP and ITS sequences. Botany 88:590–595
Hui N, Jumpponen A, Niskanen T, Liimatainen K, Jones KL et al (2011) EcM fungal community structure, but not diversity, altered in a Pb-contaminated shooting range in a boreal coniferous forest site in Southern Finland. FEMS Microbiol Ecol 76:121–132
Gao C, Shi N-N, Liu Y-X, Peay KG, Zheng Y et al (2013) Host plant genus-level diversity is the best predictor of ectomycorrhizal fungal diversity in a Chinese subtropical forest. Mol Ecol 22:3403–3414
Tedersoo L, May TW, Smith ME (2010) Ectomycorrhizal lifestyle in fungi: global diversity, distribution, and evolution of phylogenetic lineages. Mycorrhiza 20:217–263
Acknowledgments
This research was supported by grants-in-aid from the Japan Society of the Promotion of Sciences (20380087). We thank Dr. Xinhua He for his critical review on our manuscript. We also thank Dr. Haifeng Wang (Jiangxi Agricultural University), Mr. Kaishui Wu (Bureau of Forestry, Dexing City, Jiangxi Province), and Ms. Le Liu (Asian Natural Environmental Science Center, The University of Tokyo) for their field assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Fig. S1
Eight-month-old bioassay Masson pine seedlings grown in rhizosphere soils from the DXC (non-polluted forest in Dexing), TLT (Taolin Pb–Zn tailing) and DXT (Dexing tailing) sites. (JPEG 1,011 kb)
Table S1
Molecular identification of ectomycorrhizal fungal species and propagule species associated with Pinus massoniana at Taolin Pb–Zn tailing (TLT), Dexing Cu tailing (DXT) and a non-disturbed Masson pine forest (DXC). (XLSX 14 kb)
Rights and permissions
About this article
Cite this article
Huang, J., Nara, K., Zong, K. et al. Soil Propagule Banks of Ectomycorrhizal Fungi Along Forest Development Stages After Mining. Microb Ecol 69, 768–777 (2015). https://doi.org/10.1007/s00248-014-0484-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-014-0484-4