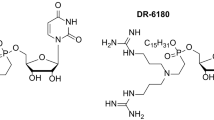
Abstract
Lantibiotics, a group of lanthionine-containing peptides, display their antibiotic activity by combining different killing mechanisms within one molecule. The prototype lantibiotic nisin was shown to possess both inhibition of peptidoglycan synthesis and pore formation in bacterial membranes by interacting with lipid II. Gallidermin, which shares the lipid II binding motif with nisin but has a shorter molecular length, differed from nisin in pore formation in several strains of bacteria. To simulate the mode of action, we applied cyclic voltammetry and quartz crystal microbalance to correlate pore formation with lipid II binding kinetics of gallidermin in model membranes. The inability of gallidermin to form pores in DOPC (1,2-dioleoyl-sn-glycero-3-phosphocholine) (C18/1) and DPoPC (1,2-dipalmitoleoyl-sn-glycero-3-phosphocholine) (C16/1) membranes was related to the membrane thickness. For a better simulation of bacterial membrane characteristics, two different phospholipids with branched fatty acids were incorporated into the DPoPC matrix. Phospholipids with methyl branches in the middle of the fatty acid chains favored a lipid II–independent DPoPC permeabilization by gallidermin, while long-branched phospholipids in which the branch is placed near the hydrophilic region induced an identical lipid II–dependent pore formation of gallidermin and nisin. Obviously, the branched lipids altered lipid packing and reduced the membrane thickness. Therefore, the duality of gallidermin activity (pore formation and inhibition of the cell wall synthesis) seems to be balanced by the bacterial membrane composition.




Similar content being viewed by others
References
Bonelli RR, Schneider T, Sahl HG, Wiedemann I (2006) Insights into in vivo activities of lantibiotics from gallidermin and epidermin mode-of-action studies. Antimicrob Agents Chemother 50:1449–1457
Breukink E, Ganz P, de Kruijff B, Seelig J (2000) Binding of nisin Z to bilayer vesicles as determined with isothermal titration calorimetry. Biochemistry 39:10247–10254
Breukink E, van Heusden HE, Vollmerhaus PJ, Swiezewska E, Brunner L, Walker S, Heck AJR, de Kruijff B (2003) Lipid II is an intrinsinc component of the pore induced by nisin in bacterial membranes. J Biol Chem 278:19898–19903
Breusch FL, Ulusoy E (1953) Synthesis of the d,l-di-n-alkyl-acetic acids with 19–23 C-atoms. Chem Ber 86:688–692
Brötz H, Bierbaum G, Reynolds PE, Sahl HG (1997) The lantibiotic mersacidin inhibits peptidoglycan biosynthesis at the level of transglycosilation. Eur J Biochem 246:193–199
Brötz H, Josten M, Wiedemann I, Schneider U, Götz F, Bierbaum G, Sahl HG (1998) Role of lipid-bound peptidoglycan precursors in the formation of pores by nisin, epidermin and other lantibiotics. Mol Microbiol 30:317–327
Chan WC, Leyland M, Clark J, Dodd HM, Lian LY, Gasson MJ, Bycroft BW, Roberts GCK (1996) Structure–activity relationships in the peptide antibiotic nisin: antibacterial activity of fragments of nisin. FEBS Lett 390:129–132
Christ K, Rüttinger HH, Höpfner M, Rothe U, Bendas G (2005) The detection of UV-induced membrane damages by a combination of two biosensor techniques. Photochem Photobiol 81:1417–1423
Christ K, Wiedemann I, Bakowsky U, Sahl HG, Bendas G (2007) The role of lipid II in membrane binding of and pore formation by nisin analyzed by two combined biosensor techniques. Biochim Biophys Acta 1768:694–704
Cotter PD, Hill C, Ross RP (2005) Bacterial lantibiotics: strategies to improve therapeutic potential. Curr Prot Pept Sci 81:61–75
Dobner B, Elsner B, Nuhn P (1989) Synthesis of methyl branched fatty acids. Pharmazie 44:758–760
Driessen AJ, van den Hooven HW, Kuiper W, van de Kamp M, Sahl HG, Konings RNH, Konings WN (1995) Mechanistic studies of lantibiotic-induced permeabilization of phospholipid vesicles. Biochemistry 34:1606–1614
Hasper HE, de Kruijff B, Breukink E (2004) Assembly and stability of nisin–lipid II pores. Biochemistry 43:11567–11575
Hsu STD, Breukink E, Tischenko E, Lutters MAG, de Kruijff B, Katein R, Bonvin AMJJ, van Nuland NAJ (2004) The nisin–lipid II complex reveals a pyrophosphate cage that provides a blueprint for novel antibiotics. Nat Struct Mol Biol 11:963–967
Johnsson T, Nikkilä P, Toivonen L, Rosenqvist H, Laakso S (1995) Cellular fatty acids profiles of lactobacillus and lactococcus strains in relation to the oleic acid content of the cultivation medium. Appl Environment Microbiol 61:4497–4499
Jung G (1991) Lantibiotics—ribosomally synthesized biologically active polypeptides containing sulfide bridges and α,β-didehydroamino acids. Angew Chem 30:1051–1192
Kellner R, Jung G, Hörner T, Zähner H, Schnell N, Entian KD, Götz F (1988) Gallidermin: a new lanthionine-containing polypeptide antibiotic. Eur J Biochem 177:53–59
Lambert LH, Cox T, Mitchell K, Roselló-Mora RA, Del Cueto C, Dodge DE, Orkand P, Cano RJ (1998) Staphylococcus succinus sp. nov., isolated from Dominican amber. Int J Syst Bacteriol 48:511–518
MacLeod P, Brown JP (1963) Fatty acid composition of lipids from Streptococcus cremoris and Streptococcus lactis var. maltigenes. J Bacteriol 85:1056–1060
Meyer HE, Heber M, Eisermann B, Korte H, Metzger JW, Jung G (1994) Sequence analysis of lantibiotics: chemical derivatization procedures allow a fast access to complete edmann degradation. Anal Biochem 223:185–190
Moll GN, Konings WN, Driessen AJM (1999) Bacteriocins: mechanism of membrane insertion and pore formation. Antonie Van Leeuwenhoek 76:185–198
Rattay B, Brezesinski G, Dobner B, Förster G, Nuhn P (1995) Influence of α-branched fatty acid chains on the thermotropic behaviour of racemic 1-O-hexadecyl-2-acyl-glycero-3-phosphocholines. Chem Phys Lipids 75:81–91
Ruhr E, Sahl HG (1985) Mode of action of the peptide antibiotic nisin and influence on the membrane potential of whole cells and on cytoplasmic and artificial membrane vesicles. Antimicrob Agents Chemother 27:841–845
Schneider T, Senn MM, Berger-Bächi B, Tossi A, Sahl HG, Wiedemann I (2004) In vitro assembly of a complete, pentaglycine interpeptide bridge containing cell wall precursor (lipid II–Gly5) of Staphylococcus aureus. Mol Microbiol 53:675–685
van Heusden HE, de Kruijff B, Breukink E (2002) Lipid II induces a transmembrane orientation of the pore forming peptide lantibiotic nisin. Biochemistry 41:12171–12178
Acknowledgments
This work was supported by the Deutsche Forschungsgemeinschaft, GRK 677, BE 2242/2-1, and SA 292/11-1.
Author information
Authors and Affiliations
Corresponding author
Additional information
Katrin Christ, Saad Al-Kaddah, and Gerd Bendas contributed equally to this article.
Rights and permissions
About this article
Cite this article
Christ, K., Al-Kaddah, S., Wiedemann, I. et al. Membrane Lipids Determine the Antibiotic Activity of the Lantibiotic Gallidermin. J Membrane Biol 226, 9–16 (2008). https://doi.org/10.1007/s00232-008-9134-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00232-008-9134-4