Abstract
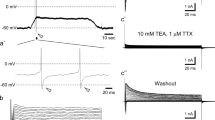
A method for microperfusion of isolated segments of the midgut epithelium of Drosophila larvae has been developed to characterize cellular transport pathways and membrane transporters. Stereological ultrastructural morphometry shows that this epithelium has unusually long tight junctions, with little or no lateral intercellular volume normally found in most epithelia. Amplification of the apical and basal aspects of the cells, by ≈ 17-fold and ≈ 7-fold, respectively, predicts an almost exclusively transcellular transport system for solutes. This correlates with the high lumen-negative transepithelial potential (Vt) of 38 to 45 mV and high resistance (Rt) of 800 to 1400 Ω • cm2 measured by terminated cable analysis, in contrast to other microperfused epithelia like the renal proximal tubule. Several blockers (amiloride 10−4 M, ouabain 10−4 M, bumetanide 10−4 M), K+-free solutions, or organic solutes such as D-glucose 10 mM or DL-alanine 0.5 mM failed to affect Vt or Rt. Bafilomycin-A1 (3 to 5 μM) decreased Vt by ≈ 40% and short-circuit current (Isc) by ≈ 50%, and decreased intracellular pH when applied from the basal side only, consistent with an inhibition of an electrogenic V-H+-ATPase located in the basal membrane. Gradients of H+ were detected by pH microelectrodes close to the basal aspect of the cells or within the basal extracellular labyrinth. The apical membrane is more conductive than the basal membrane, facilitating secretion of base (presumably HCO3−), driven by the basal V-H+-ATPase.









Similar content being viewed by others
Abbreviations
- BEL,:
-
basal extracellular labyrinth;
- DMSO,:
-
dimethyl-sulfoxide;
- HEPES,:
-
N-2-hydroxyethylpiperazine-N’-2-ethaesulfonic acid;
- Isc,:
-
short-circuit current;
- LIS,:
-
lateral intracellular space;
- Rt,:
-
transepithelial resistance;
- Va,:
-
apical cell membrane potential;
- Vb,:
-
basal cell membrane potential;
- V-H+-ATPase,:
-
vacuolar-proton-adenosinetriphosphatase;
- Vt,:
-
transepithelial potential, perfusion end;
- Vc,:
-
transepithelial potential, collection end.
References
Ashburner M. 1989. Drosophila. A Laboratory Manual. Second edition, Cold Spring Harbor Laboratory Press, New York
Berenbaum M. 1980. Adaptive significance of midgut pH in larval Lepidoptera. Am. Nat. 115:138–146
Beyenbach K.W., Pannabecker T.L. Nagel W. 2000. Central role of the apical membrane H+-ATPase in electrogenesis and epithelial transport in malpighian tubules. J. Exp. Biol. 203:1459–1468
Boudko D.Y., Moroz L.L., Harvey W.R., Linser P.J. 2001a. Alkalinization by chloride / bicarbonate pathway in larval mosquito midgut. Proc. Natl. Acad. Sci. USA 98:15354–15359
Boudko D.Y., Moroz L.L., Linser P.J., Trimarchi J.R., Smith P.J., Harvey W.R. 2001b. In situ analysis of pH gradients in mosquito larvae using non-invasive, self-referencing, pH-sensitive microelectrodes. J. Exp. Biol. 204:691–699
Brown D., Gluck S., Hartwig J. 1987. Structure of the novel membrane-coating material in proton-secreting epithelial cells and identification as an H+ATPase. J. Cell Biol. 105:1637–1648
Dadd R.H. 1975. Alkalinity within the midgut of mosquito larvae with alkaline-active digestive enzymes. J. Insect Physiol. 21:1847–1853
Dow J.A.T. 1984. Extremely high pH in biological systems; a model for carbonate transport. Am. J. Physiol. 246:R633–R635
Dow J.A.T. 1986. Insect midgut function. Adv. Insect Physiol. 19:187–328
Dow J.A.T. 1992. pH gradients in Lepidopteran midgut. J. Exp. Biol. 172:355–375
Dubreuil R.R., Frankel J., Wang P., Howrylak J., Kappil M., Grushko T.A. 1998. Mutations of α Spectrin and labial block cuprophilic cell differentiation and acid secretion in the middle midgut of Drosophila larvae. Dev. Biol. 194:1–11
Dubreuil R.R., Grushko T., Baumann O. 2001. Differential effects of a labial mutation on the development, structure, and function of stomach acid-secreting cells in Drosophila melanogaster larvae and adults. Cell Tissue Res. 306:167–178
Filippova M., Ross L.S., Gill S.S. 1998. Cloning of the V ATPase B subunit cDNA from Culex quinquefasciatus and expression of the B and C subunits in mosquitoes. Insect Mol. Biol. 7:223–232
Greenberg B. 1968. Micro-potentiometric pH determinations of muscoid maggot digestive tracts. Ann. Entomol. Soc. Am. 61:365–367
Harrison J.F. 2001. Insect acid-base physiology. Annu. Rev. Entomol. 46:221–259
Klein U., Koch A., Moffett D.F. 1996. Ion transport in Lepidoptera. In: M.J. Lehane, P.F. Billingsley, editors. Biology of the Insect Midgut. Chapman & Hall, London, pp. 236–264
Martin J.S., Martin M.M. 1984. Surfactants: their role in preventing the precipitation of proteins by tannins in insect guts. Oecologia 61:342–345
Maunsbach A.B., Boulpaep E.L. 1984. Quantitative ultrastructure and functional correlates in proximal tubule of Ambystoma and Necturus. Am. J. Physiol. 246:F710–F724
Schultz J.C., Lechowicz M.J. 1986. Host plant, larval age and feeding behavior influence midgut pH in the gypsy moth (Lymantria dispar). Oecologia 71:133–137
Shanbhag S.R., Singh K., Singh R.N. 1992. Ultrastucture of the femoral chordotonal organs and their novel synaptic organization in the legs of Drosophila melanogaster Meigen (Diptera: Drosophilidae). Int. J. Insect Morphol. & Embryol 21:311–322
Shanbhag S., Tripathi S. 2005. An electrogenic V-H+-ATPase drives electrolyte transport in the isolated perfused larval Drosophila midgut. J. Physiol. 565P: C3
Terra W.R., Ferriera C., Baker J.E. 1996. Compartmentalization of digestion. In: M.J. Lehane, P.F. Billingsley, editors, Biology of the Insect Midgut. Chapman and Hall, London, pp. 226–235
Tripathi S., Morgunov N., Boulpaep E.L. 1985. Submicron tip breakage and silanization control improve ion-selective microelectrodes. Am. J. Physiol. 249:C514–C521
Tripathi S., Boulpaep E.L., Maunsbach A.B. 1987. Isolated perfused Ambystoma proximal tubule: hydrodynamics modulates ultrastructure. Am. J. Physiol. 252:F1129–F1147
Tripathi S., Boulpaep E.L. 1988. Cell membrane water permeabilities and streaming currents in Ambystoma proximal tubule. Am. J. Physiol. 255:F188–F203
Wagner C.A., Finberg K.E., Breton S., Marshansky V., Brown D., Geibel J.P. 2004. Renal Vacuolar H+-ATPase. Am. J. Physiol 84:1263–1314
Zhuang Z., Linser P.J., Harvey W.R. 1999. Antibody to H+V-ATPase subunit E colocalizes with portasomes in alkaline larval midgut of a freshwater mosquito (Aedes aegypti L.). J. Exp. Biol. 202:2449–2460
Acknowledgement
We record our gratitude to the late Peter D’Souza and to J. N. Parmar for fabrication of the microperfusion apparatus and excellent machining support. We thank T. V. Abraham for electronics and data acquisition software. Supported by Interdisciplinary Programme 10P-809.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shanbhag, S., Tripathi, S. Electrogenic H+ Transport and pH Gradients Generated by a V-H+-ATPase in the Isolated Perfused Larval Drosophila Midgut. J Membrane Biol 206, 61–72 (2005). https://doi.org/10.1007/s00232-005-0774-1
Received:
Issue Date:
DOI: https://doi.org/10.1007/s00232-005-0774-1