Abstract
Objective
To evaluate the effect of the CYP2D6 genotype on the pharmacokinetics of tropisetron in healthy Korean subjects.
Methods
A single 5-mg capsule of tropisetron was administered orally to 13 healthy subjects. Plasma concentrations were determined by validated HPLC procedures and data were analyzed by using noncompartmental linear PK methods. Four alleles, CYP2D6*1, CYP2D6*2 ×2, CYP2D6*5, and CYP2D6*10, were identified by PCR.
Results
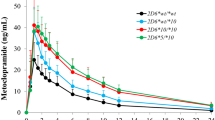
Thirteen subjects, consisting of two homozygous carriers of the wild type allele (*1/*1), four heterozygous carriers of poor metabolizer (PM)-associated allele (*1/*10), six homozygous carriers of PM-associated alleles (four with *10/*10 and two with *5/*10), and one carrier of a duplicated allele *1/*2 ×2. All tested pharmacokinetic parameters (AUCinf, AUCinf NL70, Cmax, CmaxNL70, T1/2, and Tec) were significantly different among four different genotypic groups. The mean AUCs of carriers with the heterozygous PM-associated allele and the homozygous PM-associated allele were 1.9- and 6.8-higher than those of carriers with the wild type allele, respectively. In contrast, the mean AUC of carriers with a duplicated allele was 0.5-fold lower than that of those carriers with the wild type allele.
Conclusion
The presence of CYP2D6*5, CYP2D6*10, and CYP2D6*2 ×2 has an important impact on the pharmacokinetics of tropisetron, which may influence clinical response to tropisetron therapy.


Similar content being viewed by others
References
Alvan G, Bechtel P, Iselius L, Gundert-Remy U (1990) Hydroxylation polymorphisms of debrisoquine and mephenytoin in European populations. Eur J Clin Pharmacol 39:533–537
Bertilsson L, Dahl ML, Sjoqvist F, Aberg-Wistedt A, Humble M, Johansson I, Lundqvist E, Ingelman-Sundberg M (1993) Molecular basis for rational megaprescribing in ultrarapid hydroxylators of debrisoquine. Lancet 341:63
Dahl ML, Johansson I, Bertilsson L, Ingelman-Sundberg M, Sjoqvist F (1995) Ultrarapid hydroxylation of debrisoquine in a Swedish population. Analysis of the molecular genetic basis. J Pharmacol Exp Ther 274:516–520
de Bruijn KM (1992) Tropisetron. A review of the clinical experience. Drugs 43[Suppl 3]:11–22
Eichelbaum M, Gross AS (1992) The genetic polymorphism of debrisoquine/sparteine metabolism—clinical aspects. In: Kalow W (ed) Pharmacokinetics of drug metabolism. Pergamon Press Inc., New York, pp 625–648
Eichelbaum M, Spannbrucker N, Dengler HJ (1979) Influence of the defective metabolism of sparteine on its pharmacokinetics. Eur J Clin Pharmacol 16:189–194
Fischer V, Vickers AE, Heitz F, Mahadevan S, Baldeck JP, Minery P, Tynes R (1994) The polymorphic cytochrome P-4502D6 is involved in the metabolism of both 5-hydroxytryptamine antagonists, tropisetron and ondansetron. Drug Metab Dispos 22:269–274
Fukuda T, Yamamoto I, Nishida Y, Zhou Q, Ohno M, Takada K, Azuma J (1999) Effect of the CYP2D6*10 genotype on venlafaxine pharmacokinetics in healthy adult volunteers. Br J Clin Pharmacol 47:450–453
Gaedigk A, Gotschall RR, Forbes NS, Simon SD Kearns GL, Leeder JS (1999) Optimization of cytochrome P4502D6 (CYP2D6) phenotype assignment using a genotyping algorithm based on allele frequency data. Pharmacogenetics 9:669–682
Horai Y, Nakano M, Ishizaki T, Ishikawa K, Zhou HH, Zhou BI, Liao CL, Zhang LM (1989) Metoprolol and mephenytoin oxidation polymorphisms in Far Eastern Oriental subjects: Japanese versus mainland Chinese. Clin Pharmacol Ther 46:198–207
Johansson I, Lundqvist E, Bertilsson L, Dahl ML, Sjoqvist F, Ingelman-Sundberg M (1993) Inherited amplification of an active gene in the cytochrome P450 CYP2D locus as a cause of ultrarapid metabolism of debrisoquine. Proc Natl Acad Sci U S A 90:11825–11829
Johansson I, Oscarson M, Yue Q-Y, Bertilsson L, Sjoqvist F, Ingelman-Sundberg M (1994) Genetic analysis of the Chinese cytochrome P4502D locus: characterization of variant CYP2D6 gene present in subjects with diminished capacity for debrisoquine hydroxylation. Mol Pharmacol 46:452–459
Kaiser R, Sezer O, Papies A, Bauer S, Schelenz C, Tremblay PB, Possinger K, Roots I, Brockmoller J (2002) Patient-tailored antiemetic treatment with 5-hydroxytryptamine type 3 receptor antagonists according to cytochrome P-450 2D6 genotypes. J Clin Oncol 20:2805–2811
Kees F, Farber L, Bucher M, Mair G, Morike K, Grobecker H (2001) Pharmacokinetics of therapeutic doses of tropisetron in healthy volunteers. Br J Clin Pharmacol 52:705–707
Kubota T, Yamaura Y, Ohkawa N, Hara H, Chiba K (2000) Frequencies of CYP2D6 mutant alleles in a normal Japanese population and metabolic activity of dextromethorphan O-demethylation in different CYP2D6 genotypes. Br J Clin Pharmacol 50:31–34
Lee CR, Plosker GL, McTavish D (1993) Tropisetron. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential as an antiemetic. Drugs 46:925–943
Lundqvist E, Johansson I, Ingelman-Sundberg M (1999) Genetic mechanisms for duplication and multiplication of the human CYP2D6 gene and methods for detection of duplicated CYP2D6 genes. Gene 226:327–338
Mahgoub A, Idle JR, Dring LG, Lancaster R, Smith RL (1977) Polymorphic hydroxylation of debrisoquine in man. Lancet 2:584–586
Nakamura K, Goto F, Ray WA, McAllister CB, Jacqz E, Wilkinson GR, Branch RA (1985) Interethnic differences in genetic polymorphism of debrisoquin and mephenytoin hydroxylation between Japanese and Caucasian populations. Clin Pharmacol Ther 38:402–408
Nishida Y, Fukuda T, Yamamoto I, Azuma J (2000) CYP2D6 genotypes in a Japanese population: low frequencies of CYP2D6 gene duplication but high frequency of CYP2D6*10. Pharmacogenetics 10:567–570
Roh HK, Dahl ML, Johansson I, Ingelman-Sundberg M, Cha YN, Bertilsson L (1996) Debrisoquine and S-mephenytoin hydroxylation phenotypes and genotypes in a Korean population. Pharmacogenetics 6:441–447
Roh HK, Kim CE, Chung WG, Park CS, Svensson JO, Bertilsson L (2001) Risperidone metabolism in relation to CYP2D6*10 allele in Korean schizophrenic patients. Eur J Clin Pharmacol 57:671–675
Sachse C, Brockmoller J, Hildebrand M, Muller K, Roots I (1998) Correctness of prediction of the CYP2D6 phenotype confirmed by genotyping 47 intermediate and poor metabolizers of debrisoquine. Pharmacogenetics 8:181–185
Sohn DR, Shin SG, Park CW, Kusaka M, Chiba K, Ishizaki T (1991) Metoprolol oxidation polymorphism in a Korean population: comparison with native Japanese and Chinese populations. Br J Clin Pharmacol 32:504–507
Suarez A, Stettler ER, Rey E, Pons G, Simonetta-Chateauneuf C, de Bruijn KM, Olive G, Lemerle J (1994) Safety, tolerability, efficacy and plasma concentrations of tropisetron after administration at five dose levels to children receiving cancer chemotherapy. Eur J Cancer 30A:1436–1441
Tetsu N, Shoutaro T, Naoki M, Akinori N, Tetsushi A, Hiroshi M, Akiharu F, Takeshi M (1995) Phase I study of tropisetron capsules in healthy volunteers. Clin Rep 29:1523–1559
Yoon YR, Cha IJ, Shon JH, Kim KA, Cha YN, Jang IJ, Park CW, Shin SG, Flockhart DA, Shin JG (2000) Relationship of paroxetine disposition to metoprolol metabolic ratio and CYP2D6*10 genotype of Korean subjects. Clin Pharmacol Ther 67:567–576
Acknowledgements
We acknowledge Seul Oh for her technical assistance with the PCR assay and individuals at the Seoul National University Hospital clinical trial center for their assistance with sampling procedures and preparation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, MK., Cho, JY., Lim, HS. et al. Effect of the CYP2D6 genotype on the pharmacokinetics of tropisetron in healthy Korean subjects. Eur J Clin Pharmacol 59, 111–116 (2003). https://doi.org/10.1007/s00228-003-0595-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-003-0595-1