Abstract
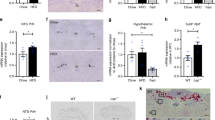
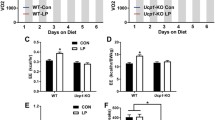
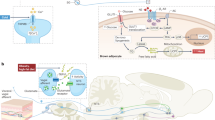
The maintenance of whole body energy homeostasis is critical to survival and mechanisms exist whereby an organism can adapt to its environment and the stresses placed upon it. Environmental temperature and thermogenesis are key components known to affect energy balance. However, little is known about how these processes are balanced against the overall energy balance. We show that even mild cold exposure has a significant effect on energy expenditure and UCP-1 levels which increase by 43% and 400%, respectively, when wild-type (WT) mice at thermoneutral (29 °C) were compared to mice at room temperature (22 °C) conditions. Interestingly, bone mass was lower in cold-stressed WT mice with significant reductions in femoral bone mineral content (− 19%) and bone volume (− 13%). Importantly, these cold-induced skeletal changes were absent in mice lacking NPY, one of the main controllers of energy homeostasis, highlighting the critical role of NPY in this process. However, energy expenditure was significantly greater in cold-exposed NPY null mice, indicating that suppression of non-thermogenic tissues, like bone, contributes to the adaptive responses to cold exposure. Altogether, this work identifies NPY as being crucial in coordinating energy and bone homeostasis where it suppresses energy expenditure, UCP-1 levels and lowers bone mass under conditions of cold exposure.





Similar content being viewed by others
Abbreviations
- BAT:
-
Brown adipose tissue
- BFR:
-
Bone formation rate
- BMD:
-
Bone mineral density
- BMC:
-
Bone mineral content
- DXA:
-
Dual x-ray absorptiometry
- MAR:
-
Mineral apposition rate
- MS:
-
Mineralising surface
- NPY:
-
Neuropeptide Y
- NPYKO:
-
Neuropeptide Y knockout
- PFA:
-
Paraformaldehyde
- RER:
-
Respiratory exchange ratio
- UCP-1:
-
Uncoupling protein-1
- WAT:
-
White adipose tissue
- WT:
-
Wild-type
References
Gordon CJ (2017) The mouse thermoregulatory system: its impact on translating biomedical data to humans. Physiol Behav 179:55–66
Cannon B, Nedergaard J (2009) Thermogenesis challenges the adipostat hypothesis for body-weight control. Proc Nutr Soc 68:401–407
Nguyen AD, Lee NJ, Wee NKY, Zhang L, Enriquez RF, Khor EC, Nie T, Wu D, Sainsbury A, Baldock PA, Herzog H (2018) Uncoupling protein-1 is protective of bone mass under mild cold stress conditions. Bone 106:167–178
Shi YC, Lau J, Lin Z, Zhang H, Zhai L, Sperk G, Heilbronn R, Mietzsch M, Weger S, Huang XF, Enriquez RF, Baldock PA, Zhang L, Sainsbury A, Herzog H, Lin S (2013) Arcuate NPY controls sympathetic output and BAT function via a relay of tyrosine hydroxylase neurons in the PVN. Cell Metab 17:236–248
Patterson-Buckendahl P, Kvetnansky R, Fukuhara K, Cizza G, Cann C (1995) Regulation of plasma osteocalcin by corticosterone and norepinephrine during restraint stress. Bone 17:467–472
Serrat MA, King D, Lovejoy CO (2008) Temperature regulates limb length in homeotherms by directly modulating cartilage growth. Proc Natl Acad Sci USA 105:19348–19353
Robbins A, Tom C, Cosman MN, Moursi C, Shipp L, Spencer TM, Brash T, Devlin MJ (2018) Low temperature decreases bone mass in mice: implications for humans. Am J Phys Anthropol 167:557–568
Iwaniec UT, Philbrick KA, Wong CP, Gordon JL, Kahler-Quesada AM, Olson DA, Branscum AJ, Sargent JL, DeMambro VE, Rosen CJ, Turner RT (2016) Room temperature housing results in premature cancellous bone loss in growing female mice: implications for the mouse as a preclinical model for age-related bone loss. Osteoporos Int 27:3091–3101
Martin SA, Philbrick KA, Wong CP, Olson DA, Branscum AJ, Jump DB, Marik CK, DenHerder JM, Sargent JL, Turner RT, Iwaniec UT (2019) Thermoneutral housing attenuates premature cancellous bone loss in male C57BL/6J mice. Endocr Connect 8:1455–1467
Motyl KJ, Bishop KA, DeMambro VE, Bornstein SA, Le P, Kawai M, Lotinun S, Horowitz MC, Baron R, Bouxsein ML, Rosen CJ (2013) Altered thermogenesis and impaired bone remodeling in misty mice. J Bone Miner Res 28:1885–1897
Takeda S, Elefteriou F, Levasseur R, Liu X, Zhao L, Parker KL, Armstrong D, Ducy P, Karsenty G (2002) Leptin regulates bone formation via the sympathetic nervous system. Cell 111:305–317
Motyl KJ, Rosen CJ (2011) Temperatures rising: brown fat and bone. Discov Med 11:179–185
Baldock PA, Lin S, Zhang L, Karl T, Shi Y, Driessler F, Zengin A, Hormer B, Lee NJ, Wong IP, Lin EJ, Enriquez RF, Stehrer B, During MJ, Yulyaningsih E, Zolotukhin S, Ruohonen ST, Savontaus E, Sainsbury A, Herzog H (2014) Neuropeptide Y attenuates stress-induced bone loss through suppression of noradrenaline circuits. J Bone Miner Res 29:2238–2249
Baldock PA, Lee NJ, Driessler F, Lin S, Allison S, Stehrer B, Lin EJ, Zhang L, Enriquez RF, Wong IP, McDonald MM, During M, Pierroz DD, Slack K, Shi YC, Yulyaningsih E, Aljanova A, Little DG, Ferrari SL, Sainsbury A, Eisman JA, Herzog H (2009) Neuropeptide Y knockout mice reveal a central role of NPY in the coordination of bone mass to body weight. PLoS ONE 4:e8415
Baldock PA, Sainsbury A, Allison S, Lin E-JD, Couzens M, Boey D, Enriquez R, During M, Herzog H, Gardiner EM (2005) Hypothalamic control of bone formation: distinct actions of leptin and Y2 receptor pathways. J Bone Miner Res 20:1851–1857
Pierroz DD, Bonnet N, Bianchi EN, Bouxsein ML, Baldock PA, Rizzoli R, Ferrari SL (2012) Deletion of beta-adrenergic receptor 1, 2, or both leads to different bone phenotypes and response to mechanical stimulation. J Bone Miner Res 27:1252–1262
Bouxsein ML, Devlin MJ, Glatt V, Dhillon H, Pierroz DD, Ferrari SL (2009) Mice lacking beta-adrenergic receptors have increased bone mass but are not protected from deleterious skeletal effects of ovariectomy. Endocrinology 150:144–152
Gordon CJ (2012) Thermal physiology of laboratory mice: defining thermoneutrality. J Therm Biol 37:654–685
Ducy P, Amling M, Takeda S, Priemel M, Schilling AF, Beil FT, Shen J, Vinson C, Rueger JM, Karsenty G (2000) Leptin inhibits bone formation through a hypothalamic relay: a central control of bone mass. Cell 100:197–207
Bal NC, Singh S, Reis FCG, Maurya SK, Pani S, Rowland LA, Periasamy M (2017) Both brown adipose tissue and skeletal muscle thermogenesis processes are activated during mild to severe cold adaptation in mice. J Biol Chem 292:16616–16625
Pant M, Bal NC, Periasamy M (2016) Sarcolipin: a key thermogenic and metabolic regulator in skeletal muscle. Trends Endocrinol Metab 27:881–892
Li L, Li B, Li M, Speakman JR (2019) Switching on the furnace: regulation of heat production in brown adipose tissue. Mol Aspects Med 68:60–73
Kuo LE, Kitlinska JB, Tilan JU, Li L, Baker SB, Johnson MD, Lee EW, Burnett MS, Fricke ST, Kvetnansky R, Herzog H, Zukowska Z (2007) Neuropeptide Y acts directly in the periphery on fat tissue and mediates stress-induced obesity and metabolic syndrome. Nat Med 13:803–811
Small L, Gong H, Yassmin C, Cooney GJ, Brandon AE (2018) Thermoneutral housing does not influence fat mass or glucose homeostasis in C57BL/6 mice. J Endocrinol 239:313–324
Overton JM (2010) Phenotyping small animals as models for the human metabolic syndrome: thermoneutrality matters. Int J Obes 34(Suppl 2):S53–S58
Maher RL, Barbash SM, Lynch DV, Swoap SJ (2015) Group housing and nest building only slightly ameliorate the cold stress of typical housing in female C57BL/6J mice. Am J Physiol Regul Integr Comp Physiol 308:R1070–1079
Harshaw C, Culligan JJ, Alberts JR (2014) Sex differences in thermogenesis structure behavior and contact within huddles of infant mice. PLoS ONE 9:e87405
Kaikaew K, Steenbergen J, Themmen APN, Visser JA, Grefhorst A (2017) Sex difference in thermal preference of adult mice does not depend on presence of the gonads. Biol Sex Differ 8:24
Patel HR, Qi Y, Hawkins EJ, Hileman SM, Elmquist JK, Imai Y, Ahima RS (2006) Neuropeptide Y deficiency attenuates responses to fasting and high-fat diet in obesity-prone mice. Diabetes 55:3091–3098
Segal-Lieberman G, Trombly DJ, Juthani V, Wang X, Maratos-Flier E (2003) NPY ablation in C57BL/6 mice leads to mild obesity and to an impaired refeeding response to fasting. Am J Physiol Endocrinol Metab 284:E1131–1139
Wee NKY, Sinder BP, Novak S, Wang X, Stoddard C, Matthews BG, Kalajzic I (2019) Skeletal phenotype of the neuropeptide Y knockout mouse. Neuropeptides 73:78–88
Erickson JC, Clegg KE, Palmiter RD (1996) Sensitivity to leptin and susceptibility to seizures of mice lacking neuropeptide Y. Nature 381:415–421
Karl T, Duffy L, Herzog H (2008) Behavioural profile of a new mouse model for NPY deficiency. Eur J Neurosci 28:173–180
Wee NKY, Enriquez RF, Nguyen AD, Horsnell H, Kulkarni R, Khor EC, Herzog H, Baldock PA (2018) Diet-induced obesity suppresses cortical bone accrual by a neuropeptide Y-dependent mechanism. Int J Obes 42:1925
Acknowledgements
NW was supported by an Australian Postgraduate Award (APA) administered by UNSW Australia. We would also like to thank the Biological Testing Facility at the Garvan Institute for assistance with the animal work.
Author information
Authors and Affiliations
Contributions
Conception and design of study: NKW, HH, PAB. Acquisition of data: NKW, ADN, RFE. Analysis and interpretation: NKW, LZ, HH, PAB. Drafting and revising of the manuscript: NKW, HH, PAB. Final manuscript approved: NKW, ADN, RFE, LZ, HH, PAB.
Corresponding author
Ethics declarations
Conflict of interests
Natalie K. Y. Wee, Amy D. Nguyen, Ronaldo F. Enriquez, Lei Zhang, Herbert Herzog and Paul A. Baldock declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
All experiments involving animals were approved by the Garvan Institute/St Vincent’s Animal Ethics Committee and conducted in accordance with the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wee, N.K.Y., Nguyen, A.D., Enriquez, R.F. et al. Neuropeptide Y Regulation of Energy Partitioning and Bone Mass During Cold Exposure. Calcif Tissue Int 107, 510–523 (2020). https://doi.org/10.1007/s00223-020-00745-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-020-00745-9