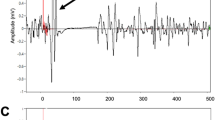
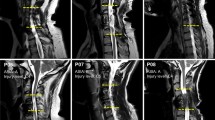
Abstract
Transcranial magnetic stimulation (TMS) is used to investigate corticomotor neurophysiology associated with functional recovery in individuals with spinal cord injury (SCI). There is insufficient evidence about test–retest measurement properties of TMS in SCI. Therefore, we investigated test–retest agreement and reliability of TMS metrics representing corticomotor excitability, output, gain, map (representation), and inhibition in individuals with cervical SCI. We collected TMS metrics from biceps and triceps muscles because of the relevance of this proximal muscle pair to the cervical SCI population. Twelve individuals with chronic C3–C6 SCI participated in two TMS sessions separated by ≥ 2 weeks. Measurement agreement was evaluated using t tests, Bland–Altman limits of agreement and relative standard error of measurement (SEM%), while reliability was investigated using intra-class correlation coefficient (ICC) and concordance correlation coefficient (CCC). We calculated the smallest detectable change for all TMS metrics. All TMS metrics except antero-posterior map coordinates and corticomotor inhibition were in agreement upon repeated measurement though limits of agreement were generally large. Measures of corticomotor excitability, output and medio-lateral map coordinates had superior agreement (SEM% < 10). Metrics representing corticomotor excitability, output, and inhibition had good-to-excellent reliability (ICC/CCC > 0.75). The smallest detectable change for TMS metrics was generally high for a single individual, but this value reduced substantially with increase in sample size. We recommend use of corticomotor excitability and recruitment curve area owing to their superior measurement properties. A modest group size (20 or above) yields more stable measurements, which may favor use of TMS metrics in group level modulation after SCI.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anderson KD (2004) Targeting recovery: priorities of the spinal cord-injured population. J Neurotrauma 21:1371–1383. https://doi.org/10.1089/neu.2004.21.1371
Beaulieu L-D, Flamand VH, Masse-Alarie H, Schneider C (2017) Reliability and minimal detectable change of transcranial magnetic stimulation outcomes in healthy adults: a systematic review. Brain Stimul 10:196–213
Berchtold A (2016) Test–retest: agreement or reliability? Methodol Innov 9:2059799116672875
Brasil-Neto JP, McShane LM, Fuhr P et al (1992) Topographic mapping of the human motor cortex with magnetic stimulation: factors affecting accuracy and reproducibility. Electroencephalogr Clin Neurophysiol Potentials Sect 85:9–16
Brouwer B, Hopkins-Rosseel DH (1997) Motor cortical mapping of proximal upper extremity muscles following spinal cord injury. Spinal Cord 35:205–212
Carson RG, Nelson BD, Buick AR et al (2013) Characterizing changes in the excitability of corticospinal projections to proximal muscles of the upper limb. Brain Stimul 6:760–768
Chen R, Lozano AM, Ashby P (1999) Mechanism of the silent period following transcranial magnetic stimulation evidence from epidural recordings. Exp Brain Res 128:539–542
Cunningham DA, Janini D, Wyant A et al (2016) Post-exercise depression following submaximal and maximal isometric voluntary contraction. Neuroscience 326:95–104
de Vet HCW, Terwee CB, Knol DL, Bouter LM (2006) When to use agreement versus reliability measures. J Clin Epidemiol 59:1033–1039
Devanne H, Lavoie BA, Capaday C (1997) Input-output properties and gain changes in the human corticospinal pathway. Exp Brain Res 114:329–338
Edwards DJ, Cortes M, Rykman-Peltz A et al (2019) Clinical improvement with intensive robot-assisted arm training in chronic stroke is unchanged by supplementary tDCS. Restor Neurol 37:167–180
Freund P, Rothwell J, Craggs M et al (2011) Corticomotor representation to a human forearm muscle changes following cervical spinal cord injury. Eur J Neurosci 34:1839–1846
Fuhr P, Agostino R, Hallett M (1991) Spinal motor neuron excitability during the silent period after cortical stimulation. Electroencephalogr Clin Neurophysiol 81:257–262
Gomes-Osman J, Field-Fote EC (2015a) Improvements in hand function in adults with chronic tetraplegia following a multi-day 10Hz rTMS intervention combined with repetitive task practice. J Neurol Phys Ther 39:23
Gomes-Osman J, Field-Fote EC (2015b) Cortical vs. afferent stimulation as an adjunct to functional task practice training: a randomized, comparative pilot study in people with cervical spinal cord injury. Clin Rehabil 29:771–782
Graves DE, Frankiewicz RG, Donovan WH (2006) Construct validity and dimensional structure of the ASIA motor scale. J Spinal Cord Med 29:39–45
Green JB, Sora E, Bialy Y et al (1999) Cortical motor reorganization after paraplegia: an EEG study. J Neurol 53:736
Groppa S, Oliviero A, Eisen A et al (2012) A practical guide to diagnostic transcranial magnetic stimulation: report of an IFCN committee. Clin Neurophysiol 123:858–882
Hoffman LR, Field-Fote EC (2007) Cortical reorganization following bimanual training and somatosensory stimulation in cervical spinal cord injury: a case report. Phys Ther 87:208–223
Koo TK, Li MY (2016) A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15:155–163
Leao MTD, Wiesinger L, Ziemann U et al (2020) Rapid motor cortical reorganization following subacute spinal cord dysfunction. Brain Stimul 13:783–785
Levy WJ Jr, Amassian VE, Traad M, Cadwell J (1990) Focal magnetic coil stimulation reveals motor cortical system reorganized in humans after traumatic quadriplegia. Brain Res 510:130–134
Malcolm MP, Triggs WJ, Light KE et al (2006) Reliability of motor cortex transcranial magnetic stimulation in four muscle representations. Clin Neurophysiol 117:1037–1046
Marino RJ, Jones L, Kirshblum S et al (2008) Reliability and repeatability of the motor and sensory examination of the international standards for neurological classification of spinal cord injury. J Spinal Cord Med 31:166–170
Mokkink LB, Terwee CB, Patrick DL et al (2010) The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J Clin Epidemiol 63:737–745
Oudega M, Perez MA (2012) Corticospinal reorganization after spinal cord injury. J Physiol 590:3647–3663
Palmer E, Ashby P (1992) Corticospinal projections to upper limb motoneurones in humans. J Physiol 448:397–412
Potter-Baker KA, Janini DP, Frost FS et al (2016) Reliability of TMS metrics in patients with chronic incomplete spinal cord injury. Spinal Cord 54:980
Rossi S, Hallett M, Rossini PM et al (2009) Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol 120:2008–2039
Rossini PM, Burke D, Chen R et al (2015) Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: basic principles and procedures for routine clinical and research application. An updated report from an IFCN Committee. Clin Neurophysiol 126:1071–1107
Sankarasubramanian V, Roelle SM, Bonnett CE et al (2015) Reproducibility of transcranial magnetic stimulation metrics in the study of proximal upper limb muscles. J Electromyogr 25:754–764
Schambra HM, Ogden RT, Martínez-Hernández I et al (2015) The reliability of repeated TMS measures in older adults and in patients with subacute and chronic stroke. Front Cell Neurosci 9:335
Smith HC, Savic G, Frankel HL et al (2000) Corticospinal function studied over time following incomplete spinal cord injury. Spinal Cord 38:292–300
Streletz LJ, Belevich JKS, Jones SM et al (1995) Transcranial magnetic stimulation: cortical motor maps in acute spinal cord injury. Brain Topogr 7:245–250
van Kuijk AA, Pasman JW, Hendricks HT et al (2009) Predicting hand motor recovery in severe stroke: the role of motor evoked potentials in relation to early clinical assessment. NeuroRehabilitation 23:45–51
Van Kuijk AAA, Bakker CD, Hendriks JCM et al (2014) Definition dependent properties of the cortical silent period in upper-extremity muscles, a methodological study. J Neuroeng Rehabil 11:1–9
Acknowledgements
We thank Yin-Liang Lin for assistance with data collection, Daniel Janini for the initial analysis, and Patrick Chabra, Corin Bonnett, and Sarah Roelle for the recruitment efforts. We also thank all research participants for their valuable time and efforts.
Funding
The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014 is the awarding and administering acquisition office. This work was supported by the USAMRAA through the Spinal Cord Injury Research Programs under Award Nos. W81XWH1810530 and W81XWH1110707 to EP. Opinions, interpretations, conclusions and recommendations are those of the author and are not necessarily endorsed by the Department of Defense or U.S. Army). This work was also supported by Conquer Paralysis Now grant (CPN1512) to EP and Cleveland Clinic Research Programs Committee award (2016-195) to KPB.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all the authors, the corresponding author states that there is no conflict of interest.
Ethical approval
The study was approved by the Institutional Review Board of the Cleveland Clinic and the Human Research Protections Office of the U.S. Department of Defense.
Consent to participate
All the participants provided written informed consent.
Additional information
Communicated by Winston D Byblow.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Arora, T., Potter-Baker, K., O’Laughlin, K. et al. Measurement error and reliability of TMS metrics collected from biceps and triceps in individuals with chronic incomplete tetraplegia. Exp Brain Res 239, 3077–3089 (2021). https://doi.org/10.1007/s00221-021-06160-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-021-06160-2