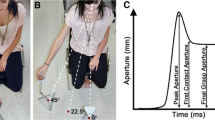
Abstract
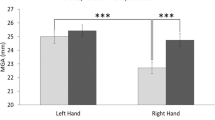
Studies have suggested a left-hemisphere specialization for visually guided grasp-to-eat actions by way of task-dependent kinematic asymmetries (i.e., smaller maximum grip apertures for right-handed grasp-to-eat movements than for right-handed grasp-to-place movements or left-handed movements of either type). It is unknown, however, whether this left-hemisphere/right-hand kinematic advantage is reliant on the dorsal “vision-for-action” visual stream. The present study investigates the kinematic differences between grasp-to-eat and grasp-to place actions performance during closed-loop (i.e., dorsally mediated) and open-loop delay (i.e., ventrally mediated) conditions. Twenty-one right-handed adult participants were asked to reach to grasp small food items to (1) eat them, or (2) place them in a container below the mouth. Grasps were performed in both closed-loop and open-loop delay conditions, in separate sessions. We show that participants displayed the right-hand grasp-to-eat kinematic advantage in the closed-loop condition, but not in the open-loop delay condition. As no task-dependent kinematic differences were found in ventrally mediated grasps, we posit that the left-hemisphere/right-hand advantage is dependent on dorsal stream processing.


Similar content being viewed by others
Notes
During pilot testing, the open-loop delay condition viewing period was set at 1000 ms to match that of our closed-loop condition. However, participants expressed frustration at this seemingly short viewing period and blamed subsequent errors on its brevity; specifically, they would often knock the target off the pedestal, perform the movement with excessive slowness, and/or express that they were unable to even identify the target before the viewing period would end. We concluded that the viewing period was too brief, and added 500 ms. This elongated viewing period eliminated the majority of both trial errors and participant frustrations. While the difference in the viewing period length may have had a significant effect on reaction times, we consider this a necessary trade-off in exchange for consistently successful movements in the open-loop delay condition. In light of this confound, and because our participants were specifically instructed to “move at a comfortable pace, with an emphasis on accuracy, not speed,” we do not include analyses on reaction time in the current report.
“Secondary” referring here to the portion of the grasping movement occurring after target acquisition; we would argue that the grasp-to-throw movement, from initiation of the grasp, to grasp onset and target acquisition, to follow-through and target release, is a single unified movement, at least with respect to neural control.
References
Annett J, Annett M, Hudson P, Turner A (1979) The control of movement in the preferred and non-preferred hands. Q J Exp Psychol 31(4):641–652
Ansuini C, Giosa L, Turella L, Altoè G, Castiello U (2008) An object for an action, the same object for other actions: effects on hand shaping. Exp Brain Res 185(1):111–119
Ansuini C, Grigis K, Massaccesi S, Castiello U (2009) Breaking the flow of an action. Exp Brain Res 192(2):287–292
Armbrüster C, Spijkers W (2006) Movement planning in prehension: do intended actions influence the initial reach and grasp movement? Mot Control 10(4):311–329
Berthier NE, Clifton RK, Gullapalli V, McCall DD, Robin D (1996) Visual information and object size in the control of reaching. J Mot Behav 28:187–197
Bootsma RJ, Marteniuk RG, MacKenzie CL, Zaal FT (1994) The speed-accuracy trade-off in manual prehension: effects of movement amplitude, object size and object width on kinematic characteristics. Exp Brain Res 98(3):535–541
Boulinguez P, Nougier V, Velay J-L (2001) Manual asymmetries in reaching movement control. I: study of right-handers. Cortex 37(1):101–122
Carnahan H (1998) Manual asymmetries in response to rapid target movement. Brain Cogn 37(2):237–253
Carson RG, Chua R, Elliott D, Goodman D (1990) The contribution of vision to asymmetries in manual aiming. Neuropsychologia 28(11):1215–1220
Carson RG, Goodman D, Chua R, Elliott D (1993) Asymmetries in the regulation of visually guided aiming. J Mot Behav 25(1):21–32
Castiello U, Bennett K, Stelmach G (1993) Reach to grasp: the natural response to perturbation of object size. Exp Brain Res 94:163–178
Castiello U, Bennett K, Chambers H (1998) Reach to grasp: the response to a simultaneous perturbation of object position and size. Exp Brain Res 120(1):31–40
Cavill S, Bryden P (2003) Development of handedness: comparison of questionnaire and performance-based measures of preference. Brain Cogn 53(2):149–151
Duff SV, Sainburg RL (2007) Lateralization of motor adaptation reveals independence in control of trajectory and steady-state position. Exp Brain Res 179(4):551–561
Elliott D, Roy EA, Goodman D, Carson RG, Chua R, Maraj BK (1993) Asymmetries in the preparation and control of manual aiming movements. Can J Exp Psychol Revue canadienne de psychologie expérimentale 47(3):570
Ferri F, Campione GC, Dalla Volta R, Gianelli C, Gentilucci M (2010) To me or to you? When the self is advantaged. Exp Brain Res 203(4):637–646
Flindall JW, Gonzalez CL (2013) On the evolution of handedness: evidence for feeding biases. PLoS One 8(11):e78967
Flindall JW, Gonzalez CL (2014) Eating interrupted: the effect of intent on hand-to-mouth actions. J Neurophysiol 112(8):2019–2025
Flindall JW, Gonzalez CL (2016) The destination defines the journey: an examination of the kinematics of hand-to-mouth movements. J Neurophysiol 116(5):2105–2113
Flindall JW, Gonzalez CL (2017) The inimitable mouth: task-dependent kinematic differences are independent of terminal precision. Exp Brain Res 235(6):1945–1952
Flindall JW, Doan JB, Gonzalez CL (2014) Manual asymmetries in the kinematics of a reach-to-grasp action. Laterality Asymmetries Body Brain Cogn 19(4):489–507
Flindall JW, Stone KD, Gonzalez CL (2015) Evidence for right-hand feeding biases in a left-handed population. Laterality Asymmetries Body Brain Cogn 20(3):287–305
Goodale M, Milner AD (1992) Separate visual pathways for perception and action. Trends Neurosci 15(1):20–25
Goodale M, Jakobson L, Keillor J (1994) Differences in the visual control of pantomimed and natural grasping movements. Neuropsychologia 32(10):1159–1178
Goodale M, Westwood DA, Milner AD (2004) Two distinct modes of control for object-directed action. Prog Brain Res 144:131–144
Grosskopf A, Kuhtz-Buschbeck JP (2006) Grasping with the left and right hand: a kinematic study. Exp Brain Res 168(1–2):230–240
Heath M, Binsted G (2007) Visuomotor memory for target location in near and far reaching spaces. J Mot Behav 39(3):169–177
Heath M, Westwood DA (2003) Can a visual representation support the online control of memory-dependent reaching? Evidence from a variable spatial mapping paradigm. Mot Control 7(4):349–365
Hesse C, Franz VH (2010) Grasping remembered objects: exponential decay of the visual memory. Vis Res 50(24):2642–2650
Hopkins WD, de Waal FB (1995) Behavioral laterality in captive bonobos (Pan paniscus): replication and extension. Int J Primatol 16(3):261–276
Hopkins WD, Bennett AJ, Bales SL, Lee J, Ward JP (1993) Behavioral laterality in captive bonobos (Pan paniscus). J Comp Psychol 107(4):403
Hopkins WD, Russell JL, Hook M, Braccini S, Schapiro SJ (2005) Simple reaching is not so simple: association between hand use and grip preferences in captive chimpanzees. Int J Primatol 26(2):259–277
Hopkins WD, Phillips KA, Bania A, Calcutt SE, Gardner M, Russell J, Schapiro SJ et al (2011) Hand preferences for coordinated bimanual actions in 777 great apes: implications for the evolution of handedness in hominins. J Hum Evol 60(5):605–611
Hu Y, Goodale M (2000) Grasping after a delay shifts size-scaling from absolute to relative metrics. J Cogn Neurosci 12(5):856–868
Hu Y, Eagleson R, Goodale M (1999) The effects of delay on the kinematics of grasping. Exp Brain Res 126(1):109–116
Marteniuk R, MacKenzie C, Jeannerod M, Athenes S, Dugas C (1987) Constraints on human arm movement trajectories. Can J Psychol Revue canadienne de psychologie 41(3):365
Milner AD, Goodale M (2006) The visual brain in action. Oxford University Press, Oxford
Milner AD, Goodale M (2008) Two visual systems re-viewed. Neuropsychologia 46(3):774–785
Mutha PK, Haaland KY, Sainburg RL (2013) Rethinking motor lateralization: specialized but complementary mechanisms for motor control of each arm. PLoS One 8(3):e58582
Naish KR, Reader AT, Houston-Price C, Bremner AJ, Holmes NP (2013) To eat or not to eat? Kinematics and muscle activity of reach-to-grasp movements are influenced by the action goal, but observers do not detect these differences. Exp Brain Res 225(2):261–275
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9(1):97–113
Paulignan Y, Frak VG, Toni I, Jeannerod M (1997) Influence of object position and size on human prehension movements. Exp Brain Res 114:226–234
Roy EA, Kalbfleisch L, Elliott D (1994) Kinematic analyses of manual asymmetries in visual aiming movements. Brain Cogn 24(2):289–295
Schaefer SY, Haaland KY, Sainburg RL (2009) Hemispheric specialization and functional impact of ipsilesional deficits in movement coordination and accuracy. Neuropsychologia 47(13):2953–2966
Shabbott BA, Sainburg RL (2008) Differentiating between two models of motor lateralization. J Neurophysiol 100(2):565–575
Stone KD, Bryant DC, Gonzalez CLR (2013) Hand use for grasping in a bimanual task: evidence for different roles? Exp Brain Res 224(3):455–467
Tomlinson T, Sainburg R (2012) Dynamic dominance persists during unsupported reaching. J Mot Behav 44(1):13–25
Tretriluxana J, Gordon J, Winstein CJ (2008) Manual asymmetries in grasp pre-shaping and transport–grasp coordination. Exp Brain Res 188(2):305–315
Veazie PJ (2006) When to combine hypotheses and adjust for multiple tests. Health Serv Res 41(3p1):804–818
Wang J, Sainburg RL (2007) The dominant and nondominant arms are specialized for stabilizing different features of task performance. Exp Brain Res 178(4):565–570
Westwood DA, Goodale M (2003) Perceptual illusion and the real-time control of action. Spat Vis 16(3):243–254
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beke, C., Flindall, J.W. & Gonzalez, C.L.R. Kinematics of ventrally mediated grasp-to-eat actions: right-hand advantage is dependent on dorsal stream input. Exp Brain Res 236, 1621–1630 (2018). https://doi.org/10.1007/s00221-018-5242-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-018-5242-2