Abstract
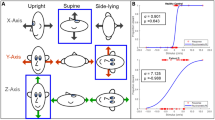
Despite the close interrelation between vestibular and visual processing (e.g., vestibulo-ocular reflex), surprisingly little is known about vestibular function in visually impaired people. In this study, we investigated thresholds of passive whole-body motion discrimination (leftward vs. rightward) in nine visually impaired participants and nine age-matched sighted controls. Participants were rotated in yaw, tilted in roll, and translated along the interaural axis at two different frequencies (0.33 and 2 Hz) by means of a motion platform. Superior performance of visually impaired participants was found in the 0.33 Hz roll tilt condition. No differences were observed in the other motion conditions. Roll tilts stimulate the semicircular canals and otoliths simultaneously. The results could thus reflect a specific improvement in canal–otolith integration in the visually impaired and are consistent with the compensatory hypothesis, which implies that the visually impaired are able to compensate the absence of visual input.

Similar content being viewed by others
Notes
“Blindness” is defined as a visual acuity <5 % or a visual acuity worse than 3/60 on the Snellen chart, respectively. A visual acuity between 3/60 and 6/60 is referred to as “severe visual impairment” (World Health Organization 2013).
A minimum reversal occurs when a participant provides the wrong answer and the stimulus level increases accordingly. A maximum reversal occurs when the subject correctly detects three subsequent motion stimuli after having made an error on the previous trial. See Figure 3 in Hartmann et al. (2014) for an example of a procedure.
References
Akbarian S, Grusser OJ, Guldin WO (1992) Thalamic connections of the vestibular cortical fields in the squirrel-monkey (Saimiri-sciureus). J Comp Neurol 326:423–441. doi:10.1002/Cne.903260308
Alary F, Goldstein R, Duquette M, Chapman CE, Voss P, Lepore F (2008) Tactile acuity in the blind: a psychophysical study using a two-dimensional angle discrimination task. Exp Brain Res 187:587–594. doi:10.1007/S00221-008-1327-7
Alary F, Duquette M, Goldstein R, Chapman CE, Voss P, La Buissonniere-Ariza V, Lepore F (2009) Tactile acuity in the blind: a closer look reveals superiority over the sighted in some but not all cutaneous tasks. Neuropsychologia 47:2037–2043. doi:10.1016/J.Neuropsychologia.2009.03.014
Angelaki DE, Yakusheva TA, Green AM, Dickman JD, Blazquez PM (2010) Computation of egomotion in the macaque cerebellar vermis. Cerebellum 9:174–182. doi:10.1007/s12311-009-0147-z
Benson AJ, Hutt ECB, Brown SF (1989) Thresholds for the perception of whole-body angular movement about a vertical axis. Aviat Space Environ Med 60:205–213
Bertolini G, Ramat S, Laurens J, Bockisch CJ, Marti S, Straumann D, Palla A (2011) Velocity storage contribution to vestibular self-motion perception in healthy human subjects. J Neurophysiol 105:209–223. doi:10.1152/Jn.00154.2010
Bisdorff AR, Wolsley CJ, Anastasopoulos D, Bronstein AM, Gresty MA (1996) The perception of body verticality (subjective postural vertical) in peripheral and central vestibular disorders. Brain 119:1523–1534. doi:10.1093/Brain/119.5.1523
Bland JM, Altman DG (1996) Transformations, means, and confidence intervals. Br Med J 312:1079. doi:10.1136/bmj.312.7038.1079
Bockisch C, Straumann D, Haslwanter T (2005) Human 3-D aVOR with and without otolith stimulation. Exp Brain Res 161:358–367. doi:10.1007/s00221-004-2080-1
Brandt T, Dieterich M (1999) The vestibular cortex—its locations, functions, and disorders. Ann NY Acad Sci 871:293–312. doi:10.1111/J.1749-6632.1999.Tb09193.X
Brandt T et al (2005) Vestibular loss causes hippocampal atrophy and impaired spatial memory in humans. Brain 128:2732–2741. doi:10.1093/brain/awh617
Bronstein AM, Grunfeld EA, Faldon M, Okada T (2008) Reduced self-motion perception in patients with midline cerebellar lesions. Neuroreport 19:691–693
Collignon O, Renier L, Bruyer R, Tranduy D, Veraart C (2006) Improved selective and divided spatial attention in early blind subjects. Brain Res 1075:175–182. doi:10.1016/J.Brainres.2005.12.079
Cousins S et al (2013) Vestibular perception following acute unilateral vestibular lesions. PLoS One. doi:10.1371/journal.pone.0061862
Cutfield NJ, Scott G, Waldman AD, Sharp DJ, Bronstein AM (2014) Visual and proprioceptive interaction in patients with bilateral vestibular loss. Neuroimage Clin 4:274–282. doi:10.1016/j.nicl.2013.12.01
Dieterich M, Brandt T (1999) Episodic vertigo related to migraine (90 cases): vestibular migraine? J Neurol 246:883–892
Falconer CJ, Mast FW (2012) Balancing the mind. Exp Psychol 59:332–339. doi:10.1027/1618-3169/a000161
Fieger A, Roder B, Teder-Salejarvi W, Hillyard SA, Neville HJ (2006) Auditory spatial tuning in late-onset blindness in humans. Cogn Neurosci 18:149–157. doi:10.1162/089892906775783697
Fortin M et al (2008) Wayfinding in the blind: larger hippocampal volume and supranormal spatial navigation. Brain 131:2995–3005. doi:10.1093/Brain/Awn250
Fuller PM, Jones TA, Jones SM, Fuller CA (2002) Neurovestibular modulation of circadian and homeostatic regulation: vestibulohypothalamic connection? Proc Natl Acad Sci USA 99:15723–15728. doi:10.1073/Pnas.242251499
Gianna C, Heimbrand S, Gresty M (1996) Thresholds for detection of motion direction during passive lateral whole-body acceleration in normal subjects and patients with bilateral loss of labyrinthine function. Brain Res Bull 40:443–447. doi:10.1016/0361-9230(96)00140-2
Goldreich D, Kanics IM (2003) Tactile acuity is enhanced in blindness. J Neurosci 23:3439–3445
Good P (2011) A practitioner’s guide to resampling for data analysis, data mining, and modeling. Chapman & Hall/CRC
Gori M, Sandini G, Martinoli C, Burr D (2010) Poor haptic orientation discrimination in nonsighted children may reflect disruption of cross-sensory calibration. Curr Biol 20:223–225. doi:10.1016/J.Cub.2009.11.069
Gougoux F, Lepore F, Lassonde M, Voss P, Zatorre RJ, Belin P (2004) Pitch discrimination in the early blind. Nature 430:309. doi:10.1038/430309a
Grabherr L, Nicoucar K, Mast FW, Merfeld DM (2008) Vestibular thresholds for yaw rotation about an earth-vertical axis as a function of frequency. Exp Brain Res 186:677–681. doi:10.1007/s00221-008-1350-8
Grabherr L, Cuffel C, Guyot J-P, Mast FW (2011) Mental transformation abilities in patients with unilateral and bilateral vestibular loss. Exp Brain Res 209:205–214. doi:10.1007/s00221-011-2535-0
Guldin WO, Akbarian S, Grusser OJ (1992) Corticocortical connections and cytoarchitectonics of the primate vestibular cortex—a study in squirrel-monkeys (saimiri-sciureus). J Comp Neurol 326:375–401. doi:10.1002/Cne.903260306
Haburcakova C, Lewis RF, Merfeld DM (2012) Frequency dependence of vestibuloocular reflex thresholds. J Neurophysiol 107:973–983. doi:10.1152/Jn.00451.2011
Hallemans A, Ortibus E, Meire F, Aerts P (2010) Low vision affects dynamic stability of gait. Gait Posture 32:547–551. doi:10.1016/J.Gaitpost.2010.07.018
Hartmann M, Grabherr L, Mast FW (2012) Moving along the mental number line: interactions between whole-body motion and numerical cognition. J Exp Psychol Hum Percept Perform 38:1416–1427. doi:10.1037/a0026706
Hartmann M, Furrer S, Herzog MH, Merfeld DM, Mast FW (2013) Self-motion perception training: thresholds improve in the light but not in the dark. Exp Brain Res 226:231–240. doi:10.1007/S00221-013-3428-1
Hartmann M, Haller K, Moser I, Hossner E-J, Mast FW (2014) Direction detection thresholds of passive self-motion in artistic gymnasts. Exp Brain Res 232:1249–1258. doi:10.1007/s00221-014-3841-0
Hesterberg TC, Moore DS, Monaghan S, Clipson A, Epstein R (2005) Bootstrap methods and permutation tests. In: Moore DS, McCabe GP (eds) Introduction to the practice of statistics. WH Freeman & Co, New York
Hötting K, Röder B (2009) Auditory and auditory-tactile processing in congenitally blind humans. Hear Res 258:165–174. doi:10.1016/J.Heares.2009.07.012
International Council of Ophthalmology (2002) Visual standards. Aspects and ranges of vision loss with emphasis on population surveys. Report prepared for the International Council of Opthalmology at the 29th International Congress of Ophthalmology. Sydney, Australia, April 2002. http://www.icoph.org/downloads/visualstandardsreport.pdf. Accessed 27 Aug 2014
Karmali F, Lim K, Merfeld DM (2014) Visual and vestibular perceptual thresholds each demonstrate better precision at specific frequencies and also exhibit optimal integration. J Neurophysiol 111:2393–2403. doi:10.1152/Jn.00332.2013
King S, Wang J, Priesol AJ, Lewis RF (2014) Central integration of canal and otolith signals is abnormal in vestibular migraine. Front Neurol 5:233. doi:10.3389/fneur.2014.00233
Kömpf D, Piper H-F (1987) Eye movements and vestibulo-ocular reflex in the blind. J Neurol 234:337–341
Leek MR (2001) Adaptive procedures in psychophysical research. Percept Psychophys 63:1279–1292. doi:10.3758/Bf03194543
Lempert T et al (2012) Vestibular migraine: diagnostic criteria. J Vestib Res 22:167–172. doi:10.3233/Ves-2012-0453
Lewis RF, Priesol AJ, Nicoucar K, Lim K, Merfeld DM (2011a) Abnormal motion perception in vestibular migraine. Laryngoscope 121:1124–1125. doi:10.1002/Lary.21723
Lewis RF, Priesol AJ, Nicoucar K, Lim K, Merfeld DM (2011b) Dynamic tilt thresholds are reduced in vestibular migraine. J Vestib Res 21:323–330. doi:10.3233/Ves-2011-0422
Machado ML, Lelong-Boulouard V, Philoxene B, Davis A, Denise P, Besnard S (2014) Vestibular loss promotes procedural response during a spatial task in rats. Hippocampus 24:591–597. doi:10.1002/Hipo.22251
MacNeilage PR, Banks MS, DeAngelis GC, Angelaki DE (2010) Vestibular heading discrimination and sensitivity to linear acceleration in head and world coordinates. J Neurosci 30:9084–9094. doi:10.1523/Jneurosci.1304-10.2010
Martin T et al (2015) Vestibular loss disrupts daily rhythm in rats. J Appl Physiol (1985) 118:310–318. doi:10.1152/japplphysiol.00811.2014
Mast FW, Merfeld DA, Kosslyn SA (2006) Visual mental imagery during caloric vestibular stimulation. Neuropsychologia 44:101–109. doi:10.1016/J.Neuropsychologia.2005.04.005
Merfeld DM (2011) Signal detection theory and vestibular thresholds: I. Basic theory and practical considerations. Exp Brain Res 210:389–405. doi:10.1007/S00221-011-2557-7
Mittelstaedt H (1996) Somatic graviception. Biol Psychol 42:53–74. doi:10.1016/0301-0511(95)05146-5
Niemeyer W, Starlinger I (1981) Do the blind hear better—investigations on auditory processing in congenital or early acquired blindness. II. Central functions. Audiology 20:510–515
Occelli V, Spence C, Zampini M (2013) Auditory, tactile, and audiotactile information processing following visual deprivation. Psychol Bull 139:189–212. doi:10.1037/A0028416
Okada T, Grunfeld E, Shallo-Hoffmann J, Bronstein AM (1999) Vestibular perception of angular velocity in normal subjects and in patients with congenital nystagmus. Brain 122:1293–1303. doi:10.1093/Brain/122.7.1293
Pasqualotto A, Proulx MJ (2012) The role of visual experience for the neural basis of spatial cognition. Neurosci Biobehav Rev 36:1179–1187. doi:10.1016/J.Neubiorev.2012.01.008
Postma A, Zuidhoek S, Noordzij ML, Kappers AML (2008) Haptic orientation perception benefits from visual experience: evidence from early-blind, late-blind, and sighted people. Percept Psychophys 70:1197–1206. doi:10.3758/Pp.70.7.1197
Priesol AJ, Valko Y, Merfeld DM, Lewis RF (2014) Motion perception in patients with idiopathic bilateral vestibular hypofunction. Otolaryngol Head Neck Surg 150:1040–1042. doi:10.1177/0194599814526557
Raphan T, Matsuo V, Cohen B (1979) Velocity storage in the vestibulo-ocular reflex arc (VOR). Exp Brain Res 35:229–248
Ray CT, Horvat M, Croce R, Mason RC, Wolf SL (2008) The impact of vision loss on postural stability and balance strategies in individuals with profound vision loss. Gait Posture 28:58–61. doi:10.1016/J.Gaitpost.2007.09.010
Röder B, Rösler F (2003) Memory for environmental sounds in sighted, congenitally blind and late blind adults: evidence for cross-modal compensation. Int J Psychophysiol 50:27–39. doi:10.1016/S0167-8760(03)00122-3
Röder B, Rösler F, Spence C (2004) Early vision impairs tactile perception in the blind. Curr Biol 14:121–124. doi:10.1016/J.Cub.2003.12.054
Rombaux P, Huart C, De Volder AG, Cuevas I, Renier L, Duprez T, Grandin C (2010) Increased olfactory bulb volume and olfactory function in early blind subjects. Neuroreport 21:1069–1073. doi:10.1097/Wnr.0b013e32833fcb8a
Sack RL, Brandes RW, Kendall AR, Lewy AJ (2000) Entrainment of free-running circadian rhythms by melatonin in blind people. N Engl J Med 343:1070–1077. doi:10.1056/Nejm200010123431503
Sanabria-Bohorquez SM, De Volder AG, Arno P, Sibomana M, Coppens A, Michel C, Veraart C (2001) Decreased benzodiazepine receptor density in the cerebellum of early blind human subjects. Brain Res 888:203–211. doi:10.1016/S0006-8993(00)03049-3
Schautzer F, Hamilton D, Kalla R, Strupp M, Brandt T (2003) Spatial memory deficits in patients with chronic bilateral vestibular failure. Ann NY Acad Sci 1004:316–324. doi:10.1196/annals.1303.029
Schwenn O, Hundorf I, Moll B, Pitz S, Mann W (2002) Do blind persons have a better sense of smell than normal sighted people? Klin Monatsbl Augenheilkd 219:649–654. doi:10.1055/s-2002-35167
Seemungal BM, Gunaratne IA, Fleming IO, Gresty MA, Bronstein AM (2004) Perceptual and nystagmic thresholds of vestibular function in yaw. J Vestib Res 14:461–466
Seemungal BM, Glasauer S, Gresty MA, Bronstein AM (2007) Vestibular perception and navigation in the congenitally blind. J Neurophysiol 97:4341–4356. doi:10.1152/Jn.01321.2006
Sherman KR, Keller EL (1986) Vestibulo-ocular reflexes of adventitiously and congenitally blind adults. Invest Ophthalmol Vis Sci 27:1154–1159
Soyka F, Giordano PR, Beykirch K, Bulthoff HH (2011) Predicting direction detection thresholds for arbitrary translational acceleration profiles in the horizontal plane. Exp Brain Res 209:95–107. doi:10.1007/S00221-010-2523-9
Starlinger I, Niemeyer W (1981) Do the blind hear better—investigations on auditory processing in congenital or early acquired blindness. 1. Peripheral functions. Audiology 20:503–509
Sterr A, Green L, Elbert T (2003) Blind Braille readers mislocate tactile stimuli. Biol Psychol 63:117–127. doi:10.1016/S0301-0511(03)00051-6
Stevens AA, Weaver K (2005) Auditory perceptual consolidation in early-onset blindness. Neuropsychologia 43:1901–1910. doi:10.1016/J.Neuropsychologia.2005.03.007
Taylor MM, Creelman CD (1967) Pest—efficient estimates on probability functions. J Acoust Soc Am 41:782. doi:10.1121/1.1910407
Ungar S, Blades M, Spencer C (1997) Teaching visually impaired children to make distance judgments from a tactile map. J Vis Impair Blind 91:163–174
Valko Y, Lewis RF, Priesol AJ, Merfeld DM (2012) Vestibular labyrinth contributions to human whole-body motion discrimination. J Neurosci 32:13537–13542. doi:10.1523/Jneurosci.2157-12.2012
Vecchi T, Tinti C, Cornoldi C (2004) Spatial memory and integration processes in congenital blindness. Neuroreport 15:2787–2790
Voss P, Lassonde M, Gougoux F, Fortin M, Guillemot JP, Lepore F (2004) Early- and late-onset blind individuals show supra-normal auditory abilities in far-space. Curr Biol 14:1734–1738. doi:10.1016/J.Cub.2004.09.051
Wan CY, Wood AG, Reutens DC, Wilson SJ (2010) Early but not late-blindness leads to enhanced auditory perception. Neuropsychologia 48:344–348. doi:10.1016/j.neuropsychologia.2009.08.016
Wearne S, Raphan T, Cohen B (1998) Control of spatial orientation of the angular vestibuloocular reflex by the nodulus and uvula. J Neurophysiol 79:2690–2715
World Health Organization (2013) ICD update and revision platform: change the definition of blindness. http://www.who.int/blindness/en/index.html. Accessed 27 Aug 2014
Yates JT, Johnson R, Starz W (1972) Loudness perception of the blind. Audiology 11:368–376
Zwiers MP, Van Opstal AJ, Cruysberg JRM (2001) A spatial hearing deficit in early-blind humans. J Neurosci 21:art. no.-RC142
Acknowledgments
We would like to thank Vanda Lory for technical assistance and both Patrick Kilchör and Katharina Engelhart for help in recruiting visually impaired participants. This research was supported by the Swiss National Science Foundation (Sinergia Grant “Balancing Self and Body”, CRSII1_125135) and a postdoctoral fellowship awarded to Luzia Grabherr.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moser, I., Grabherr, L., Hartmann, M. et al. Self-motion direction discrimination in the visually impaired. Exp Brain Res 233, 3221–3230 (2015). https://doi.org/10.1007/s00221-015-4389-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-015-4389-3