Abstract
The location of an object in peripersonal space can be represented with respect to our body (i.e., egocentric frame of reference) or relative to contextual features and other objects (i.e., allocentric frame of reference). In the current study, we sought to determine whether the frame, or frames, of visual reference supporting motor output is influenced by reach trajectories structured to maximize visual feedback utilization (i.e., controlled online) or structured largely in advance of movement onset via central planning mechanisms (i.e., controlled offline). Reaches were directed to a target embedded in a pictorial illusion (the induced Roelofs effect: IRE) and advanced knowledge of visual feedback was manipulated to influence the nature of reaching control as reported by Zelaznik et al. (J Mot Behav 15:217–236, 1983). When vision could not be predicted in advance of movement onset, trajectories showed primary evidence of an offline mode of control (even when vision was provided) and endpoints demonstrated amplified sensitivity to the illusory (i.e., allocentric) features of the IRE. In contrast, reaches performed with reliable visual feedback evidenced a primarily online mode of control and showed increased visuomotor resistance to the IRE. These findings suggest that the manner a reaching response is structured differentially influences the weighting of allocentric and egocentric visual information. More specifically, when visual feedback is unavailable or unpredictable, the weighting of allocentric visual information for the advanced planning of a reach trajectory is increased.



Similar content being viewed by others
Notes
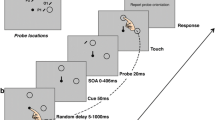
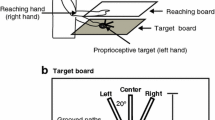
We elected to use the induced Roelofs effect because the illusory features of this stimulus are thought constructed by later visual processing systems (i.e., the ventral visual pathway; see Milner and Dyde 2003 for a discussion of this issue). Moreover, our IRE configuration was oriented in the anteroposterior direction (see Fig. 1) and thus differs from the mediolateral orientation used in most previous work (e.g., Bridgeman et al. 1997, 2000, Dassonville et al. 2004). Importantly, however, the IRE orientation used here has been shown to robustly influence reaching endpoints (Coello et al. 2003; Neely et al. 2007; see Neely 2005 for discussion of IRE orientations and visuomotor susceptibility to allocentric visual cues).
The fact that R 2 values at 25% of movement time did not differentiate between the feedback schedule and visual condition combinations is congruent with earlier work arguing that at this time point the spatiotemporal features of an initial movement impulse do not have sufficient time to unfold to predict ultimate movement endpoints (see Heath et al. 2004b).
Of course, the hypothesis proposed here is specific to situations in which reaching/grasping responses are directed to a target within a structured visual background and not restrictive experimental contexts wherein allocentric visual cues are not available to the performer; e.g., when a performer reaches to an isolated target (i.e., point of light) presented in an otherwise neutral or empty visual background.
Bridgeman et al’s (1997) original IRE experiment is frequently cited as providing direct evidence that the IRE does not influence visuomotor control. Careful examination of that experiment, however (see Experiment 1), shows that “5 subjects showed a highly significant Roelofs effect [F(2, 4) > 18, P < 0.01], whereas the other 5 showed no sign of an effect [F(2, 4) ≤ 3.16, P > 0.18]” (p. 460). Importantly, that finding combined with a more recent work (Neely et al. 2007) highlights the existence of a controversy surrounding the extent to which the IRE represents an exemplar illusion supporting the view that egocentric visual cues restrictively mediate visually derived reaches.
References
Adamovich SV, Berkinblit MB, Fookson O, Poizner H (1999) Pointing in 3D space to remembered targets. II. Effects of movement speed toward kinesthetically defined targets. Exp Brain Res 125:200–210
Beggs WD, Howarth CI (1970) Movement control in a repetitive motor task. Nature 225:752–753
Binsted G, Brownell K, Vorontsova Z, Heath M, Saucier D (2007) Visuomotor system uses target information unavailable to conscious awareness. Proc Natl Acad Sci USA 104:12669–12672
Binsted G, Heath M (2004) Can the motor system use a stored representation to control movement? Behav Brain Sci 27:25–27
Bridgeman B, Gemmer A, Forsman T, Huemer V (2000) Processing spatial information in the sensorimotor branch of the visual system. Vision Res 40:3539–3352
Bridgeman B, Peery S, Anand S (1997) Interaction of cognitive and sensorimotor maps of visual space. Percept Psychophys 59:456–469
Carlton LG (1981) Processing visual feedback information for movement control. J Exp Psychol Hum Percept Perform 7:1019–1030
Coello Y, Grealy MA (1997) Effect of size and frame of visual field on the accuracy of an aiming movement. Perception 26:287–300
Coello Y, Richaud S, Magne P, Rossetti Y (2003) Vision for spatial perception and vision for action: a dissociation between the left–right and near–far dimensions. Neuropsychologia 41:622–633
Conti P, Beaubaton D (1980) Role of structured visual field and visual reafference in accuracy of pointing movements. Percept Mot Skills 50:239–244
Culham JC, Danckert SL, DeSouza JF, Gati JS, Menon RS, Goodale MA (2003) Visually guided grasping produces fMRI activation in dorsal but not ventral stream brain areas. Exp Brain Res 153:180–189
Daprati E, Gentilucci M (1997) Grasping an illusion. Neuropsychologia 35:1577–1582
Dassonvile P, Bridgeman B, Bala JK, Thiem P, Sampanes A (2004) The induced Roelofs effect: two visual systems or the shift of a single reference frame? Vision Res 44:603–611
Diedrichsen J, Werner S, Schmidt T, Trommershäuser J (2004) Immediate spatial distortions of pointing movements induced by visual landmarks. Percept Psychophys 66:89–103
Elliott D (1988) The influence of visual target and limb information on manual aiming. Can J Psychol 42:57–68
Elliott D, Allard F (1985) The utilization of visual feedback information during rapid pointing movements. Q J Exp Psychol 37:407–425
Elliott D, Binsted G, Heath M (1999a) The control of goal-directed limb movements: correcting errors in the trajectory. Hum Mov Sci 18:121–136
Elliott D, Heath M, Binsted G, Ricker KL, Roy EA, Chua R (1999b) Goal-directed aiming: correcting a force-specification error with the right and left hands. J Mot Behav 31:309–324
Elliott D, Helsen WF, Chua R (2001) A century later: Woodworth’s (1899) two-component model of goal-directed aiming. Psychol Bull 127:342–57
Elliott D, Lee TD (1995) The role of target information on manual-aiming bias. Psychol Res 58:2–9
Elliott D, Madalena J (1987) The influence of premovement visual information on manual aiming. Q J Exp Psychol 39A:541–559
Gentilucci M, Chieffi S, Daprati E, Saetti MC, Toni I (1996) Visual illusion and action. Neuropsychologia 34:369–376
Glazebrook CM, Dhillon VP, Keetch KM, Lyons J, Amazeen E, Weeks DJ, Elliott D (2005) Perception-action and the Muller-Lyer illusion: amplitude or endpoint bias? Exp Brain Res 160:71–78
Goodale MA, Milner AD (1992) Separate visual pathways for perception and action. Trends Neurosci 15:20–25
Goodale MA, Westwood DA (2004) An evolving view of duplex vision: separate but interacting cortical pathways for perception and action. Curr Opin Neurobiol 14:203–211
Goodale MA, Westwood DA, Milner AD (2004) Two distinct modes of control for object-directed action. Prog Brain Res 144:131–144
Heath M (2005) Role of limb and target vision in the online control of memory-guided reaches. Motor Control 9:281–311
Heath M, Neely K, Binsted G (2007) Allocentric visual cues influence online limb adjustments. Motor Control 11:54–70
Heath M, Rival C (2005) Role of the visuomotor system in on-line attenuation of a premovement illusory bias in grip aperture. Brain Cogn 57:111–114
Heath M, Rival C, Binsted G (2004a) Can the motor system resolve a premovement bias in grip aperture? Online analysis of grasping the Müller-Lyer illusion. Exp Brain Res 158:378–384
Heath M, Rival C, Neely K (2006a) Visual feedback schedules influence visuomotor resistance to the Müller-Lyer figures. Exp Brain Res 168:348–56
Heath M, Rival C, Neely K, Krigolson O (2006b) Müller-Lyer illusions influence the online reeorganziation of visually guided grasping movements. Exp Brain Res 169:473–481
Heath M, Rival C, Westwood DA, Neely K (2005) Time course analysis of closed- and open-loop grasping of the Müller-Lyer illusion. J Mot Behav 37:179–185
Heath M, Westwood DA (2003) Can a visual representation support the online control of memory-dependent reaching? Evidence from a variable spatial mapping paradigm. Motor Control 7:346–361
Heath M, Westwood DA, Binsted G (2004b) The control of memory-guided reaching movements in peripersonal space. Motor Control 8:76–106
Henry FM (1986) Development of the motor memory trace and control program. J Mot Behav 18:77–100
Henry FM, Rogers DE (1960) Increased response latency for complication movements and a “memory drum” theory of neuromotor reaction. Res Q Exerc Sport 31:448–458
Held R, Gottlieb N (1958) Technique for studying adaptation to disarranged hand–eye coordination. Percept Mot Skills 8:83–86
Hu Y, Goodale MA (2000) Grasping after a delay shifts size-scaling from absolute to relative metrics. J Cogn Neurosci 12:856–868
Hu Y, Eagleson R, Goodale MA (1999) The effects of delay on the kinematics of grasping. Exp Brain Res 126:109–116
Jakobson LS, Goodale MA (1991) Factors affecting higher-order movement planning: a kinematic analysis of human prehension. Exp Brain Res 86:199–208
James TW, Culham J, Humphrey GK, Milner AD, Goodale MA (2003) Ventral occipital lesions impair object recognition but not object-directed grasping: an fMRI study. Brain 126:2463–2475
Khan MA, Elliott D, Coull J, Chua R, Lyons J (2002) Optimal control strategies under different feedback schedules: kinematic evidence. J Mot Behav 32:45–57
Keetch KM, Glazebrook CM, Lyons J, Lam MY, Weeks DJ, Elliott D (2006) The effect of response uncertainty on illusory biases of perception and action. Neurosci Lett 406:117–121
Keele SW (1968) Movement control in skilled motor performance. Psychol Bull 70:387–403
Klapp ST (1975) Feedback versus motor programming in the control of aimed movements. J Exp Psychol Hum Percept Perform 104:161–169
Krigolson O, Clark N, Heath M, Binsted G (2007) The proximity of visual landmarks impacts reaching performance. Spat Vis 20:317–336
Krigolson O, Heath M (2004) Background visual cues and memory-guided reaching. Hum Mov Sci 23:861–877
Krigolson O, Heath M (2006) A lower visual field advantage for endpoint stability but no advantage for online movement precision. Exp Brain Res 170:127–135
McIntosh RD, McClements KI, Schindler I, Cassidy TP, Birchall D, Milner AD (2004) Avoidance of obstacles in the absence of visual awareness. Proc Biol Sci 7:15–20
McIntyre J, Stratta F, Lacquaniti F (1997) Viewer-centered frame of reference for pointing to memorized targets in three-dimensional space. J Neurophysiol 78:1601–1618
Mendoza JE, Elliott D, Meegan DV, Lyons JL, Welsh TN (2006) The effect of the Muller-Lyer illusion on the planning and control of manual aiming movements. J Exp Psychol Hum Percept Perform 32:413–422
Mendoza J, Hansen S, Glazebrook CM, Keetch KM, Elliott D (2005) Visual illusions affect both movement planning and on-line control: a multiple cue position on bias and goal-directed action. Hum Mov Sci 24:760–773
Merigan WH, Maunsell JH (1993) How parallel are the primate visual pathways? Annu Rev Neurosci 16:369–402
Messier J, Kalaska JF (1999) Comparison of variability of initial kinematics and endpoints of reaching movements. Exp Brain Res 125:139–152
Meyer DA, Abrams RA, Kornblum S, Wright CE, Smith JEK (1988) Optimality in human motor performance: ideal control of rapid aimed movements. Psychol Rev 95:340–370
Milner AD, Goodale MA (1995) The visual brain in action. Oxford University Press, Oxford
Milner D, Dyde R (2003) Why do some perceptual illusions affect visually guided action, when others don’t? Trends Cogn Sci 7:10–11
Neely KA (2005) The induced Roelofs effect: evidence for an interaction between allocentric and egocentric visual information. Unpublished master’s thesis, Indiana University, Bloomington
Neely KA, Binsted G, Heath M (2007) Allocentric and egocentric visual cues influence the specification of movement distance and direction. J Mot Behav (in press)
Obhi SS, Goodale MA (2005) The effects of landmarks on the performance of delayed and real-time pointing movements. Exp Brain Res 167:335–344
Pisella L, Grea H, Tilikete C, Vighetto A, Desmurget M, Rode G, Boisson D, Rossetti Y (2000) An ‘automatic pilot’ for the hand in human posterior parietal cortex: toward reinterpreting optic ataxia. Nat Neurosci 3:729–736
Plamondon R (1995) A kinematic theory of rapid human movements. Part II. Movement time and control. Biol Cybern 72:309–720
Proteau L, Marteniuk RG, Levesque L (1992) A sensorimotor basis for motor learning: evidence indicating specificity of practice. Q J Exp Psychol 44A:557–575
Redon C, Hay L (2005) Role of visual context and oculomotor conditions in pointing accuracy. Neuroreport 16:2065–2067
Schluter ND, Rushworth MF, Mills KR, Passingham RE (1999) Signal-, set-, and movement-related activity in the human premotor cortex. Neuropsychologia 37:233–243
Schmidt RA, Zelaznik H, Hawkins B, Frank JS, Quinn JT (1979) Motor-output variability: a theory for the accuracy of rapid motor acts. Psychol Rev 47:415–451
Schindler I, Rice NJ, McIntosh RD, Rossetti Y, Vighetto A, Milner AD (2004) Automatic avoidance of obstacles is a dorsal stream function: evidence from optic ataxia. Nat Neurosci 7:779–784
Smeets JBJ, Brenner E, de Grave DDJ, Cuijpers RH (2002) Illusions in action: consequences of inconsistent processing of spatial attributes. Exp Brain Res 147:135–144
Smyrnis N, Taira M, Ashe J, Georgopoulos AP (1992) Motor cortical activity in a memorized delay task. Exp Brain Res 92:139–151
Ungerleider LG, Courtney SM, Haxby JV (1998) A neural system for human visual working memory. Proc Natl Acad Sci USA 95:883–90
Velay JL, Beaubaton D (1986) Influence of visual context on pointing movement accuracy. Cahiers de Psychologie Cognitive 6:447–456
Westwood DA, Heath M, Roy EA (2000) The effect of a pictorial illusion on closed-loop and open-loop prehension. Exp Brain Res 134:456–463
Westwood DA, Heath M, Roy EA (2001) The accuracy of reaching movements in brief delay conditions. Can J Exp Psychol 55:304–10
Westwood DA, Heath M, Roy EA (2003) No evidence for accurate visuomotor memory: systematic and variable error in memory-guided reaching. J Mot Behav 35:127–133
Westwood DA, Goodale MA (2003) Perceptual illusion and the real-time control of action. Spat Vis 16:243–254
Whitney D, Westwood DA, Goodale MA (2003) The influence of visual motion on fast reaching movements to a stationary object. Nature 423:869–873
Woodworth RS (1899) The accuracy of voluntary movement. Psychol Rev 3:1–114
Zelaznik HZ, Hawkins B, Kisselburgh L (1983) Rapid visual feedback processing in single-aiming movements. J Mot Behav 15:217–236
Acknowledgments
Natural Sciences and Engineering Research Council of Canada Discovery Grants (MH and GB) and a University of Western Ontario Major Academic Development Fund Award (MH) supported this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Neely, K.A., Tessmer, A., Binsted, G. et al. Goal-directed reaching: movement strategies influence the weighting of allocentric and egocentric visual cues. Exp Brain Res 186, 375–384 (2008). https://doi.org/10.1007/s00221-007-1238-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-007-1238-z