Abstract
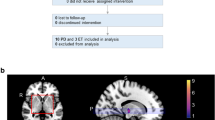
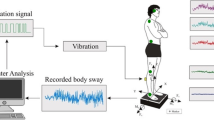
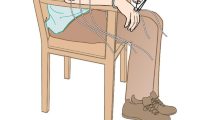
A preferred target for parkinsonian tremor alleviation is the ventrolateral (VL) thalamus. The goal of the present study is to determine how lesions involving the presumed cerebellar and pallidal recipient areas of the “motor” thalamus would alter the tremor and motor behavior of ten patients with Parkinson’s disease (PD). Tremor amplitude, power dispersion (a measure of sharpness of the power spectrum of tremor), and power distribution were quantified using a laser displacement sensor prior to, and a week after, VL thalamotomy. As well, the impact of surgery on tremor seen during movement was quantified in a manual-tracking (MT) task. Tremor-induced noise (a measure of the amount of tremor present during movement) and ERROR (difference between subject’s performance and target) were quantified. Finally, bradykinesia was assessed with a rapid alternating movement (RAM) task. Duration, range, and amplitude irregularity of wrist pronation–supination cycles were computed. Both motor tasks were quantified using a highly sensitive forearm rotational sensor. Healthy age-matched control subjects were also tested. Magnetic resonance images with an integrated atlas of thalamic nuclei were used to confirm lesion location. Results show that the lesions were centered upon the posterior portion of the ventral lateral (VLp) nucleus of the thalamus, included the posterior part of the ventral lateral anterior nucleus (VLa), and extended posteriorly to encroach upon the most rostral sector of the sensory ventral posterior nucleus (VPLa). VL thalamotomy significantly decreased tremor amplitude in all cases. Power dispersion was increased significantly so that it became similar to that of control subjects. Changes in power distribution indicate that thalamotomy selectively targeted PD tremor oscillations. Tremor detected during the MT task was also markedly decreased, becoming similar to that of controls. Patients also showed significant decrease in ERROR during MT. RAM duration and range were not significantly modified by the surgery, and patients’ performance remained impaired compared to healthy control subjects. Collectively, these results suggest that lesions involving the presumed “cerebellar” and “pallidal” recipient sectors of the motor thalamus do not worsen bradykinesia, suggesting that neural circuits other than the pallido-thalamo-cortical loop may be involved in slowness of movement in PD. A review of alternate pathways is presented.





Similar content being viewed by others
References
Atkinson JD, Collins L, Bertrand G, Peters TM, Pike BG, Sadikot AF (2002) Optimal location of thalamotomy lesions for tremor associated with Parkinson’s disease: a probabilistic analysis based on postoperative magnetic resonance imaging and integrated digital atlas. J Neurosurg 96:854–866
Aziz TZ, Davies L, Stein J, France S (1998) The role of descending basal ganglia connections to the brain stem in parkinsonian akinesia. Br J Neurosurg 12:245–249
Benabid AL (2003) Deep brain stimulation for Parkinson’s disease. Curr Opin Neurobiol 13:696–706
Berardelli A, Rothwell JC, Thompson PD, Hallet M (2001) Pathophysiology of bradykinesia in Parkinson’s disease. Brain 124:2131–2146
Bertrand G (1966a) Stimulation during stereotactic operations for dyskinesias. J Neurosurg 24:419–428
Bertrand G (1966b) Localization of lesions. J Neurosurg 24:446–448
Bertrand G, Jasper S, Wong A, Matthews G (1969) Microelectrode recording during stereotactic surgery. Clin Neurosurg 16:328–355
Beuter A, Edwards R, deGeoffroy A, Mergler D, Hundnell K (1999) Quantification of neuromotor function for detection of the effects of manganese. Neurotoxicology 20(2–3):355–366
Bhatia KP, Marsden CD (1994) The behavioural and motor consequences of focal lesions of the basal ganglia in man. Brain 117(Pt 4):859–876
Boecker H, Wills AJ, Ceballos-Baumann A, Samuel M, Thomas DG, Marsden CD, Brooks DJ (1997) Stereotactic thalamotomy in tremor-dominant Parkinson’s disease: an H2 (15)O PET motor activation study. Ann Neurol 41:108–111
Braak H, Del Tredici K, Rub U, de Vos RA, Jansen Steur EN, Braak E (2003) Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 24(2):197–211
Brown RG, Pluck G (2000) Negative symptoms: the pathology of motivation and goal-directed behaviour. Trends Neurosci 23(9):412–417
Van Buren JM, Li CL, Shapiro DY, Henderson WG, Sadowsky DA (1973) A qualitative and quantitative evaluation of parkinsonians three to six years following thalamotomy. Confin Neurol 35(4):202–235
Van Buskirk C, Wolbarcht ML, Stecher K Jr (1966) The non nervous causes of normal physiologic tremor. Neurology 16:217–220
DeLong MR (1990) Primate models of movement disorders of basal ganglia origin. Trends Neurosci 13(7):281–285
Duval C, Jones J (2005) Amplitude assessment of oscillations associated with high frequency components of physiological tremor: impact of loading and signal differentiation. Exp Brain Res 163(2):261–266
Duval C, Panisset M, Bertrand G, Sadikot AF (2000) Evidence that ventrolateral thalamotomy may eliminate the supraspinal component of both pathological and normal physiological tremors. Exp Brain Res 132(2):216–222
Duval C, Panisset M, Sadikot AF (2001) The relationship between physiological tremor and the performance of rapid alternating movements in healthy elderly subjects. Exp Brain Res 139:412–418
Duval C, Sadikot AF, Panisset M (2004) The detection of tremor during slow alternating movements performed by patients with early Parkinson’s disease. Exp Brain Res 154(3):395–398
Duval C, Strafella AP, Sadikot AF (2005) The impact of vetrolateral thalamotomy on high frequency components of tremor. Clin Neurophysiol 116(6):1391–1399
Elble RJ (1995) Mechanisms of physiological tremor and relationship to essential tremor. In: Findley LJ, Koller WC (eds) Handbook of tremor disorders. Dekker, New York, pp 51–62
Filion M (1979) Effects of interruption of the nigrostriatal pathway and of dopaminergic agents on the spontaneous activity of globus pallidus neurons in the awake monkey. Brain Res 178:425–441
Filion M, Tremblay L (1991) Abnormal spontaneous activity of globus pallidus neurons in monkey with MPTP-induced parkinsonism. Brain Res 547:142–151
Finnis KW, Starreveld YP, Parrent AG, Sadikot AF, Peters TM (2003) Three-dimensional database of subcortical electrophysiology for image-guided stereotactic functional neurosurgery. IEEE Trans Med Imaging 22(1):93–104
Hassler R (1982) Architectonic organization of the thalamic nuclei. In: Schaltenbrand G, Walker AR (eds) Stereotaxy of the human brain Anatomical Physiological and clinical application, 2nd edn. Theime, Stuttgart, pp 140–180
Hirai T, Jones EG (1989) A new parcellation of the human thalamus on the basis of histochemical staining. Brain Res Brain Res Rev 14(1):1–34
Hornykiewicz O, Kish SJ (1987) Biochemical pathophysiology of Parkinson’s disease. Adv Neurol 45:19–34
Ilinsky IA, Jouandet ML, Goldman-Rakic PS (1985) Organization of the nigrothalamocortical system in the rhesus monkey. J Comp Neurol 236(3):315–330
Jellinger KA (2002) Recent developments in the pathology of Parkinson’s disease. J Neural Transm Suppl 62:347–376
Kirk RE (1968) Experimental design: procedures for the behavioral sciences, 1st edn. Brooks-Cole, Monterey
Kojima J, Yamaji Y, Matsumura M, Nambu A, Inase M, Tokuno H, Takada M, Imai H (1997) Excitotoxic lesions of the pedunculopontine tegmental nucleus produce contralateral hemiparkinsonism in the monkey. Neurosci Lett 226(2):111–114
Köster B, Lauk M, Timmer J, Winter T, Guschlbauer B, Glocker FX, Danek A, Deuschl G, Lucking CH (1998) Central mechanisms in human enhanced physiological tremor. Neurosci Lett 241(2–3):135–138
Krayenbühl H, Siegfried J, Yasargil MG (1963) Résultats tardifs des opérations stéréotaxiques dans le traitement de la maladie de Parkinson. Rev Neurol (Paris) 108:485–494
Lee MS, Marsden CD (1994) Movement disorders following lesions of the thalamus or subthalamic region. Mov Disord 9(5):493–507
Lozano AM, Lang AE, Galvez-Jimenez N, Miyasaki J, Duff J, Hutchinson WD, Dostrovsky JO (1995) Effect of GPi Pallidotomy on motor function in Parkinson’s disease. Lancet 346:1383–1387
Van Manen J, Speelman JD, Tans RJJ (1984) Indications for surgical treatment of Parkinson’s disease after levodopa therapy. Clin Neurol Neurosurg 86:207–212
Marsden CD (1984) Origins of normal and pathological tremor. In: Findley LJ, Calpiledo R (eds) Movement disorders: tremor. Macmillan, London, pp 165–182
Marsden CD, Meadows JC, Lange GW, Watson RS (1969) The role of ballistocardiac impulse in the genesis of physiological tremor. Brain 92:647–662
Miller WC, DeLong MR (1988) Parkinsonian symptomatology. An anatomical and physiological analysis. Ann N Y Acad Sci 515:287–302
Mink JW (1996) The basal ganglia: focused selection and inhibition of competing motor programs. Prog Neurobiol 50(4):381–425
Mogenson GJ, Yang CR (1991) The contribution of basal forebrain to limbic-motor integration and the mediation of motivation to action. In: Napier TC, Kalivas PW, Hanin I (eds) The basal forebrain: anatomy to function. Advances in experimental medicine and biology, vol 295. Plenum Press, New York, p 267
Nagaseki Y, Shibazaki T, Hirai T, Kawashima Y, Hirato M, Wada H, Miyazaki M, Ohye C (1986) Long-term follow-up results of selective VIM-thalamotomy. J Neurosurg 65:296–302
Obrist WD, Thompson HK Jr, Wang HS, Wilkinson WE (1975) Regional cerebral blood flow estimated by 133-xenon inhalation. Stroke 6(3):245–256
Ohye C, (2000) Use of selective thalamotomy for various kinds of movement disorders, based on basic studies. Stereotact Funct Neurosurg 75:54–65
Ohye C, Hirai T, Miyazaki M, Shibazaki T, Nakajima H (1982) VIM thalamotomy for the treatment of various kinds of tremor. Appl Neurophysiol 45:275–280
Ohye C, Shibasaki T, Hirai T, Kawashima Y, Hirato M, Matsumura M (1985) Plastic change of thalamic organization in patients with tremor after stroke. Appl Neurophysiol 48:288–292
Okada M, Okada M (1983) A method for quantification of alternate pronation and supination of forearms. Comput Biomed Res 16(1):59–78
Okun MS, Vitek JL (2004) Lesion therapy for Parkinson’s disease and other movement disorders: update and controversies. Mov Disord 19(4):375–389
Pahapill PA, Lozano AM (2000) The pedunculopontine nucleus and Parkinson’s disease. Brain 123(Pt 9):1767–1783
Paré D, Curro’Dossi R, Steriade M (1990) Neural basis of the parkinsonian resting tremor: a hypothesis and its implications for treatment. Neuroscience 35:217–226
Parent A, Cossette M (2001) Extrastriatal dopamine and Parkinson’s disease. Adv Neurol 86:45–54
Paus T (2001) Primate anterior cingulate cortex: where motor control, drive and cognition interface. Nat Rev Neurosci 2(6):417–424
Perret E (1968) Simple motor performance of patients with Parkinson’s disease before and after surgical lesion in the thalamus. J Neurol Neurosurg Psychiatry 31:284–290
Perret E, Eggenberger E, Siegfried J (1970) Simple and complex finger movement performance of patients with Parkinsonism before and after a unilateral stereotaxic thalamotomy. J Neurol Neurosurg Psychiatry 33(1):16–21
Sadikot AF, Parent A, Smith Y, Bolam JP (1992) Efferent connections of the centromedian and parafascicular thalamic nuclei in the squirrel monkey: a light and electron microscopic study of the thalamostriatal projection in relation to striatal heterogeneity. J Comp Neurol 320:228–242
Shink E, Sidibe M, Smith Y (1997) Efferent connections of the internal globus pallidus in the squirrel monkey: II Topography and synaptic organization of pallidal efferents to the pedunculopontine nucleus. J Comp Neurol 382(3):348–363
Sidibe M, Bevan MD, Bolam JP, Smith Y (1997) Efferent connections of the internal globus pallidus in the squirrel monkey: I Topography and synaptic organization of the pallidothalamic projection. J Comp Neurol 382(3):323–347
van Someren EJ, van Gool WA, Vonk BF, Mirmiran M, Speelman JD, Bosch DA, Swaab DF (1993) Ambulatory monitoring of tremor and other movements before and after thalamotomy: a new quantitative technique. J Neurol Sci 117(1–2):16–23
Stepniewska I, Sakai ST, Qi HX, Kaas JH (2003) Somatosensory input to the ventrolateral thalamic region in the macaque monkey: potential substrate for parkinsonian tremor. J Comp Neurol 455(3):378–395
Stiles RN, Randall JE (1967) Mechanical factors in human tremor frequency. J Appl Physiol 23(3):324–330
St-Jean P, Sadikot AF, Collins L, Clonda D, Kasrai R, Evans AC, Peters TM (1998) Automated atlas integration and interactive three-dimensional visualization tools for planning and guidance in functional neurosurgery. IEEE Trans Med Imaging 17(5):672–680
Strafella AP, Dagher A, Sadikot AF (2003) Cerebral blood flow changes induced by subthalamic stimulation in Parkinson’s disease. Neurology 60(6):1039–1042
Velasco F, Velasco M, Ogarrio C, Olvera A (1986) Neglect induced by thalamotomy in humans: a quantitative appraisal of the sensory and motor deficits. Neurosurgery 19(5):744–751
Vogt BA, Pandya DN, Rosene DL (1987) Cingulate cortex of the rhesus monkey: I Cytoarchitecture and thalamic afferents. J Comp Neurol 262(2):256–270
Volkmann J, Joliot M, Mogilner A, Ioannides AA, Lado F, Fazzini E, Ribary U, Llinas R (1996) Central motor loop oscillations in parkinsonian resting tremor revealed by magnetoencephalography. Neurology 46(5):1359–1370
Walker AE (1982) Stereotaxic surgery for tremor. In: Schaltenbrand G, Walker AE (eds) Stereotaxy for the human brain: anatomical Physiological and clinical applications. Thieme, Stuttgart, pp 515–521
Wichmann T, DeLong MR (2003) Pathophysiology of Parkinson’s disease: the MPTP primate model of the human disorder. Ann N Y Acad Sci 991:199–213
Wichmann T, Bergman H, Starr PA, Subramanian T, Watts RL, DeLong MR (1999) Comparison of MPTP-induced changes in spontaneous neuronal discharge in the internal pallidal segment and in the substantia nigra pars reticulata in primates. Exp Brain Res 125(4):397–409
Yap CB, Boshes B (1967) The frequency and pattern of normal tremor. Electroencephalogr Clin Neurophysiol 22:197–203
Zweig RM, Jankel WR, Hedreen JC, Mayeux R, Price DL (1989) The pedunculopontine nucleus in Parkinson’s disease. Ann Neurol 26:41–46
Acknowledgments
Christian Duval is supported by the Natural Sciences and Engineering Research Council of Canada (NSERC), Canada Foundation for Innovation (CFI), Ontario Innovation Trust (OIT), and Parkinson Society Canada. Antonio P. Strafella and Abbas F. Sadikot are supported by Operating Grants from the Canadian Institute for Health Research (CIHR, AS, AFS) and the Parkinson Society of Canada (AFS). Abbas F. Sadikot is supported by a Senior Scientist Award of the Fonds de Recherche en Santé du Quebec (FRSQ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duval, C., Panisset, M., Strafella, A.P. et al. The impact of ventrolateral thalamotomy on tremor and voluntary motor behavior in patients with Parkinson’s disease. Exp Brain Res 170, 160–171 (2006). https://doi.org/10.1007/s00221-005-0198-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-005-0198-4