Abstract
Liraglutide is a glucagon-like peptide-1 analog for the treatment of type 2 diabetes. Major interference in plasma of human and animals and low fragment signal in tandem mass spectrometry are the main difficulties encountered in the bioanalysis of liraglutide. In this study, by combining differential mobility spectrometry (DMS) with multiple ion monitoring detection (MIM), a liquid chromatography differential mobility spectrometry tandem mass spectrometry with multiple ion monitoring detection (LC-DMS-MIM) method was developed for the quantitation of liraglutide in dog plasma. Mixed anion-exchange solid-phase extraction was used for sample preparation. The parameters of DMS were meticulously optimized to increase the signal-to-noise ratio of the analyte. The assay was linear in the range 1–100 ng/mL with good accuracy and precision. The lower limit of quantitation (LLOQ, the lowest standard on the calibration curve) of this method was 1 ng/mL. The research reveals that DMS is an effective tool for the elimination of interference in bioanalysis and that LC-DMS-MIM has better specificity and higher signal-to-noise ratio than classical liquid chromatography-tandem mass spectrometry (LC-MS/MS) for the bioanalysis of liraglutide.

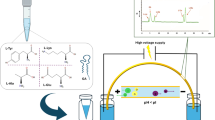
Process for the bioanalysis of liraglutide by liquid chromatography differential mobility spectrometry tandem mass spectrometry with multiple ion monitoring detection




Similar content being viewed by others
References
Nauck M, Stockmann F, Ebert R, Creutzfeldt W. Reduced incretin effect in type 2 (non-insulin-dependent) diabetes. Diabetologia. 1986;29(1):46–52.
Tuduri E, Lopez M, Dieguez C, Nadal A, Nogueiras R. Glucagon-like peptide 1 analogs and their effects on pancreatic islets. Trends Endocrinol Metab. 2016;27(5):304–18.
Petersen AB, Knop FK, Christensen M. Lixisenatide for the treatment of type 2 diabetes. Drugs Today. 2013;49(9):537–53.
Elbrond B, Jakobsen G, Larsen S, Agerso H, Jensen LB, Rolan P, Sturis J, Hatorp V, Zdravkovic M. Pharmacokinetics, pharmacodynamics, safety, and tolerability of a single-dose of NN2211, a long-acting glucagon-like peptide 1 derivative, in healthy male subjects. Diabetes Care. 2002;25(8):1398–404.
Agerso H, Jensen LB, Elbrond B, Rolan P, Zdravkovic M. The pharmacokinetics, pharmacodynamics, safety and tolerability of NN2211, a new long-acting GLP-1 derivative, in healthy men. Diabetologia. 2002;45(2):195–202.
Watson E, Jonker DM, Jacobsen LV, Ingwersen SH. Population pharmacokinetics of liraglutide, a once-daily human glucagon-like peptide-1 analog, in healthy volunteers and subjects with type 2 diabetes, and comparison to twice-daily exenatide. J Clin Pharmacol. 2010;50(8):886–94.
Ryan GJ, Foster KT, Jobe LJ. Review of the therapeutic uses of liraglutide. Clin Ther. 2011;33(7):793–811.
Nuffer WA, Trujillo JM. Liraglutide: a new option for the treatment of obesity. Pharmacotherapy. 2015;35(10):926–34.
Hall MJ, Adin CA, Borin-Crivellenti S, Rudinsky AJ, Rajala-Schultz P, Lakritz J, Gilor C. Pharmacokinetics and pharmacodynamics of the glucagon-like peptide-1 analog liraglutide in healthy cats. Domest Anim Endocrinol. 2015;51:114–21.
Moody DE, Laycock JD, Spanbauer AC, Crouch DJ, Foltz RL, Josephs JL, Amass L, Bickel WK. Determination of buprenorphine in human plasma by gas chromatography-positive ion chemical ionization mass spectrometry and liquid chromatography-tandem mass spectrometry. J Anal Toxicol. 1997;21(6):406–14.
Ceccato A, Klinkenberg R, Hubert P, Streel B. Sensitive determination of buprenorphine and its N-dealkylated metabolite norbuprenorphine in human plasma by liquid chromatography coupled to tandem mass spectrometry. J Pharm Biomed Anal. 2003;32(4–5):619–31.
Fu Y, Xia YQ, Flarakos J, Tse FL, Miller JD, Jones EB, Li W. Differential mobility spectrometry coupled with multiple ion monitoring in regulated LC-MS/MS bioanalysis of a therapeutic cyclic peptide in human plasma. Anal Chem. 2016;88(7):3655–61.
Levin DS, Miller RA, Nazarov EG, Vouros P. Rapid separation and quantitative analysis of peptides using a new nanoelectrospray-differential mobility spectrometer-mass spectrometer system. Anal Chem. 2006;78(15):5443–52.
Beach DG, Kerrin ES, Quilliam MA. Selective quantitation of the neurotoxin BMAA by use of hydrophilic-interaction liquid chromatography-differential mobility spectrometry-tandem mass spectrometry (HILIC-DMS-MS/MS). Anal Bioanal Chem. 2015;407(28):8397–409.
Liu C, Le Blanc JC, Shields J, Janiszewski JS, Ieritano C, Ye GF, Hawes GF, Hopkins WS, Campbell JL. Using differential mobility spectrometry to measure ion solvation: an examination of the roles of solvents and ionic structures in separating quinoline-based drugs. Analyst. 2015;140(20):6897–903.
Kolakowski BM, Mester Z. Review of applications of high-field asymmetric waveform ion mobility spectrometry (FAIMS) and differential mobility spectrometry (DMS). Analyst. 2007;132(9):842–64.
Ge L, Yong JW, Tan SN, Yang XH, Ong ES. Analysis of cytokinin nucleotides in coconut (Cocos nucifera L.) water using capillary zone electrophoresis-tandem mass spectrometry after solid-phase extraction. J Chromatogr a. 2006;1133(1–2):322–31.
Josefsson M, Sabanovic A. Sample preparation on polymeric solid phase extraction sorbents for liquid chromatographic-tandem mass spectrometric analysis of human whole blood - a study on a number of beta-agonists and beta-antagonists. J Chromatogr a. 2006;1120(1–2):1–12.
Meece J. Pharmacokinetics and pharmacodynamics of liraglutide, a long-acting, potent glucagon-like peptide-1 analog. Pharmacotherapy. 2009;29(12 Pt 2):33s–42s.
Such-Sanmartin G, Bache N, Callesen AK, Rogowska-Wrzesinska A, Jensen ON. Targeted mass spectrometry analysis of the proteins IGF1, IGF2, IBP2, IBP3 and A2GL by blood protein precipitation. J Proteome. 2015;113:29–37.
De Bock M, de Seny D, Meuwis MA, Servais AC, Minh TQ, Closset J, Chapelle JP, Louis E, Malaise M, Merville MP, Fillet M. Comparison of three methods for fractionation and enrichment of low molecular weight proteins for SELDI-TOF-MS differential analysis. Talanta. 2010;82(1):245–54.
Malm-Erjefalt M, Bjornsdottir I, Vanggaard J, Helleberg H, Larsen U, Oosterhuis B, van Lier JJ, Zdravkovic M, Olsen AK. Metabolism and excretion of the once-daily human glucagon-like peptide-1 analog liraglutide in healthy male subjects and its in vitro degradation by dipeptidyl peptidase IV and neutral endopeptidase. Drug Metab Dispos. 2010;38(11):1944–53.
Porta T, Varesio E, Hopfgartner G. Gas-phase separation of drugs and metabolites using modifier-assisted differential ion mobility spectrometry hyphenated to liquid extraction surface analysis and mass spectrometry. Anal Chem. 2013;85(24):11771–9.
Purves RW. Enhancement of biological mass spectrometry by using separations based on changes in ion mobility (FAIMS and DMS). Anal Bioanal Chem. 2013;405(1):35–42.
Kellie JF, Kehler JR, Szapacs ME. Application of high-resolution MS for development of peptide and large-molecule drug candidates. Bioanalysis. 2016;8(3):169–77.
Acknowledgments
The authors gratefully acknowledge financial support from the National Natural Science Foundation of China (Grant Nos. 81430087, 81473142, 81673396, and 81673502). We are also grateful to SCIEX application engineers Huaidong Yu and Chao Zhang for technical support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 288 kb)
Rights and permissions
About this article
Cite this article
Meng, X., Xu, H., Zhang, Z. et al. Differential mobility spectrometry tandem mass spectrometry with multiple ion monitoring for the bioanalysis of liraglutide. Anal Bioanal Chem 409, 4885–4891 (2017). https://doi.org/10.1007/s00216-017-0431-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-017-0431-6