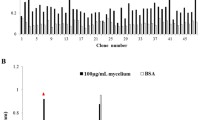
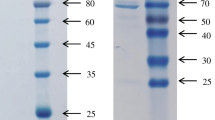
Abstract
A toxin-free enzyme-linked immunosorbent assay (ELISA) for aflatoxins was developed using an anti-idiotype nanobody VHH 2–5 as surrogate standard. Anti-idiotype nanobody VHH 2–5 was generated by immunizing an alpaca with anti-aflatoxin monoclonal antibody 1C11. This assay was used to detect aflatoxins in agro-products after a simple extraction with 75 % methanol/H2O. Aflatoxin concentration was calculated by a two-step approach: the concentration of VHH 2–5 was first obtained by a four-parameter logistic regression from the detected absorbance value at 450 nm, and then converted to aflatoxin concentration by a linear equation. The assay exhibits a limit of detection (LOD) of 0.015 ng mL−1, which is better than or comparable with conventional immunoassays. The performance of our VHH surrogate-based ELISA was further validated with a high-performance liquid chromatography (HPLC) method for total aflatoxins determination in 20 naturally contaminated peanut samples, displaying a good correlation (R 2 = 0.988). In conclusion, the proposed assay represents a first example applying an anti-idiotype VHH antibody as a standard surrogate in ELISA. With the advantages of high stability and ease of production, the VHH antibody-based standard surrogate can be extended in the future to immunoassays for other highly toxic compounds.

ᅟ






Similar content being viewed by others
References
Hammock BD, Mumma RO. Potential of immunochemical technology for pesticide analysis. In: Harvey Jr J, Zeig G, editors. Recent pesticide analytical methodology. Washington DC: American Chemical Society; 1980.
Groopman JD, Trudel LJ, Donahue PR, Marshak-Rothstein A, Wogan GN. High-affinity monoclonal antibodies for aflatoxins and their application to solid-phase immunoassays. Proc Natl Acad Sci U S A. 1984;81:7728–31.
Tang D, Yu Y, Niessner R, Miró M, Knopp D. Magnetic bead-based fluorescence immunoassay for aflatoxin B1 in food using biofunctionalized rhodamine B-doped silica nanoparticles. Analyst. 2010;135:2661–7.
Ngundi MM, Shriver-Lake LC, Moore MH, Lassman ME, Ligler FS, Taitt CR. Array biosensor for detection of ochratoxin A in cereals and beverages. Anal Chem. 2005;77:148–54.
Hervás M, López MA, Escarpa A. Integrated electrokinetic magnetic bead-based electrochemical immunoassay on microfluidic chips for reliable control of permitted levels of zearalenone in infant foods. Analyst. 2011;136:2131–8.
Meneely JP, Sulyok M, Baumgartner S, Krska R, Elliott CT. A rapid optical immunoassay for the screening of T-2 and HT-2 toxin in cereals and maize-based baby food. Talanta. 2010;81:630–6.
Guo Y, Sanders M, Galvita A, Heyerick A, Deforce D, Bracke M, et al. Heterologous screening of hybridomas for the development of broad-specific monoclonal antibodies against deoxynivalenol and its analogues. World Mycotoxin J. 2014;7:1–9.
Yeung JM, Prelusky DB, Savard ME, Dang BDM, Robinson LA. Sensitive immunoassay for fumonisin B1 in corn. J Agr Food Chem. 1996;44:3582–6.
Reiter EV, Cichna-Markl M, Chung D, Shim W, Zentek J, Razzazi-Fazeli E. Determination of ochratoxin A in grains by immune-ultrafiltration and HPLC-fluorescence detection after postcolumn derivatisation in an electrochemical cell. Anal Bioanal Chem. 2011;400:2615–22.
Zhao Z, Liu N, Yang L, Deng Y, Wang J, Song S, et al. Multi-mycotoxin analysis of animal feed and animal-derived food using LC-MS/MS system with timed and highly selective reaction monitoring. Anal Bioanal Chem. 2015;407:7359–68.
Thirumala-Cevi K, Miller JS, Reddy G, Reddy DVR. Phage-displayed peptides that mimic afatoxin B1 in serological reactivity. J Appl Microbiol. 2001;90:330–6.
Yuan Q, Pestka JJ, Hespenheide BM, Kuhn LA, Linz JE, Hart LP. Identification of mimotope peptides which bind to the mycotoxin deoxynivalenol-specific monoclonal antibody. Appl Environ Microbiol. 1999;65:3279–86.
Liu R, Yu Z, He Q, Xu Y. An immunoassay for ochratoxin A without the mycotoxin. Food Control. 2007;17:872–7.
He Z, He Q, Xu Y, Li Y, Liu X, Chen B, et al. Ochratoxin A mimotope from second-generation peptide library and its application in immunoassay. Anal Chem. 2013;85:10304–11.
He Q, Xu Y, Zhang C, Li Y, Huang Z. Phage-borne peptidomimetics as immunochemical reagent in dot-immunoassay for mycotoxin zearalenone. Food Control. 2014;39:56–61.
Jerne NK. Towards a network theory of the immune system. Ann Immunol (Paris). 1974;125C:373–89.
Hsu KH, Chu FS. Anti-idiotype and anti-anti-idiotype antibodies generated from polyclonal antibodies against aflatoxin B1. Food Agric Immunol. 1995;7:139–51.
Guan D, Li P, Cui Y, Zhang Q, Zhang W. A competitive immunoassay with a surrogate calibrator curve for aflatoxin M1 in milk. Anal Chim Acta. 2011;703:64–9.
Yu FY, Chu FS. Production and characterization of a monoclonal anti-anti-idiotype antibody against fumonisin B1. J Agric Food Chem. 1999;47:4815–20.
Chanh TC, Huot RI, Schick MR, Hewetson JF. Anti-idiotypic antibodies against a monoclonal antibody specific for the trichothecene mycotoxin T-2. Toxicol Appl Pharmacol. 1989;100:201–7.
Van Houwelingen A, De Saeger S, Rusanova T, Waalwijk C, Beekwilder J. Generation of recombinant alpaca VHH antibody fragments for the detection of the mycotoxin ochratoxin A. World Mycotoxin J. 2008;1:407–17.
Hamers-Casterman C, Atarhouch T, Muyldermans S, Robinson G, Hamers C, Songa EB, et al. Naturally occurring antibodies devoid of light chains. Nature. 1993;363:446–8.
Desmyter A, Transue TR, Ghahroudi MA, Thi MH, Poortmans F, Hamers R, et al. Crystal structure of a camel single-domain VH antibody fragment in complex with lysozyme. Nat Struct Biol. 1996;3:803–11.
Cortez-Retamozo V, Lauwereys M, Hassanzadeh Gh. G, Gobert M, Conrath K, Muyldermans S, et al. Efficient tumor targeting by single-domain antibody fragments of camels. Int J Cancer. 2002;98:456–62.
Doyle PJ, Arbabi-ghahroudi M, Gaudette N, Furzer G, Savard ME, Gleddie S, et al. Cloning, expression, and characterization of a single-domain antibody fragment with affinity for 15-acetyl-deoxynivalenol. Mol Immunol. 2008;45:3703–13.
Kim HJ, McCoy MR, Majkova Z, Dechant JE, Gee SJ, Tabares-da Rosa S, et al. Isolation of alpaca anti-hapten heavy chain single domain antibodies for development of sensitive immunoassay. Anal Chem. 2012;84:1165–71.
Ladenson RC, Crimmins DL, Landt Y, Ladenson JH. Isolation and characterization of a thermally stable recombinant anti-caffeine heavy-chain antibody fragment. Anal Chem. 2006;78:4501–8.
Tabares-da Rosa S, Rossotti M, Carleiza C, Carri F, Pritsch O, Ahn KC, et al. Competitive selection from single domain antibody libraries allows isolation of high-affinity antihapten antibodies that are not favored in the llama immune response. Anal Chem. 2011;83:7213–20.
Zarebski LM, Urrutia M, Goldbaum FA. Llama single domain antibodies as a tool for molecular mimicry. J Mol Biol. 2005;349:814–24.
Xu Y, Xiong L, Li Y, Xiong Y, Tu Z, Fu J, et al. Anti-idiotypic nanobody as citrinin mimotope from a naïve alpaca heavy chain single domain antibody library. Anal Bioanal Chem. 2015;407:5333–41.
Li J, Xia L, Su Y, Liu H, Xia X, Lu Q, et al. Molecular imprint of enzyme active site by camel nanobodies rapid and efficient approach to produce abzymes with alliinase activity. J Biol Chem. 2012;287:13713–21.
Shephard GS. Aflatoxin analysis at the beginning of the twenty-first century. Anal Bioanal Chem. 2009;395:1215–24.
Li P, Zhang Q, Zhang W. Immunoassays for aflatoxins. Trends Anal Chem. 2009;28:1115–26.
Wang Y, Li P, Majkova Z, Bever CRS, Kim HJ, Zhang Q, et al. Isolation of alpaca anti-idiotypic heavy-chain single-domain antibody for the aflatoxin immunoassay. Anal Chem. 2013;85:8298–303.
Zhang D, Li P, Zhang Q, Zhang W, Huang Y, Ding X, et al. Production of ultrasensitive generic monoclonal antibodies against major aflatoxins using a modified two-step screening procedure. Anal Chim Acta. 2009;636:63–9.
Guan D, Li P, Zhang Q, Zhang W, Zhang D, Jiang J. An ultra-sensitive monoclonal antibody-based competitive enzyme immunoassay for aflatoxin M1 in milk and infant milk products. Food Chem. 2011;125:1359–64.
Li X, Li P, Zhang Q, Zhang Z, Li R, Zhang W, et al. A sensitive immunoaffinity column-linked indirect competitive ELISA for ochratoxin A in cereal and oil products based on a new monoclonal antibody. Food Anal Method. 2013;6:1433–40.
Tang X, Li X, Li P, Zhang Q, Li R, Zhang W, et al. Development and application of an immunoaffinity column enzyme immunoassay for mycotoxin zearalenone in complicated samples. Plos One. 2014;9:e85606.
Li X, Li P, Lei J, Zhang Q, Zhang W, Li C. A simple strategy to obtain ultra-sensitive single-chain fragment variable antibodies for aflatoxin detection. RSC Adv. 2013;3:22367–72.
Ma F, Chen R, Li P, Zhang Q, Zhang W, Hu X. Preparation of an immunoaffinity column with amino-silica gel microparticles and its application in sample cleanup for aflatoxin detection in agri-products. Molecules. 2013;18:2222–35.
Beijing Products Quality Supervision and Inspection. GB/T 18979–2003: Determination of aflatoxins content in food. Cleanup by immunoaffinity chromatography and determination by high-performance liquid chromatography and fluorometer. Beijing: National Standardization Technical Committee on Food Industry; 2003.
Lee NA, Wang S, Allan RD, Kennedy IR. A rapid aflatoxin B1 ELISA: development and validation with reduced matrix effects for peanuts, corn, pistachio, and Soybeans. J Agri Food Chem. 2004;52:2746–55.
Wang Y, Wang H, Li P, Zhang Q, Kim HJ, Gee SJ, et al. Phage-displayed peptide that mimics aflatoxins and its application in immunoassay. J Agric Food Chem. 2013;61:2426–33.
Acknowledgement
This work was supported by the Special Fund for Agro-scientific Research in the Public Interest (201203094), the Project of National Science & Technology Pillar Plan (2012BAB19B09), the National Natural Science Foundation of China (31171702, 31501560), and Fundamental Research Funds for the Central Universities (Z109021566).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no competing interest.
Additional information
Published in the topical collection Immunoanalysis for Environmental Monitoring and Human Health with guest editors Shirley J. Gee, Ivan R. Kennedy, Alice Lee, Hideo Ohkawa, Tippawan Prapamontol, and Ting Xu.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 29 kb)
Rights and permissions
About this article
Cite this article
Wang, Y., Li, P., Zhang, Q. et al. A toxin-free enzyme-linked immunosorbent assay for the analysis of aflatoxins based on a VHH surrogate standard. Anal Bioanal Chem 408, 6019–6026 (2016). https://doi.org/10.1007/s00216-016-9370-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9370-x