Abstract
Sensitive, specific, simple, fast, and low-cost DNA detection methods are extremely important in clinical diagnostics, gene therapy, and a variety of biomedical studies. In this work, we developed a general method for the detection of specific DNA sequences from Mycobacterium tuberculosis (TB), hepatitis B virus (HBV), and myelocytomatosis viral oncogene (v-myc) using gold nanoparticles bifunctionalized with both a chemiluminescent (CL) reagent and a catalytic metal complex as signal reporters and a DNA strand complementary to the target as the capture probe. In this CL method, a biotinylated single-strand DNA capture probe was immobilized in a streptavidin-coated microwell. Upon the addition of the target single-strand DNA, the capture probe hybridized with the target DNA. After adding the bifunctionalized gold nanoparticles and H2O2, a well-defined CL signal was obtained, and the CL intensity was observed to change as the target DNA concentration was increased. It was possible to determine the concentration of the target TB single-strand DNA in the range 1.0 × 10−13–1.0 × 10−8 M with a detection limit of 4.8 × 10−14 M. HBV single-strand DNA and v-myc single-strand DNA could also be determined in the range 1.0 × 10−11–1.0 × 10−8 M with detection limits of 5.9 × 10−12 M and 8.0 × 10−12 M, respectively, using this CL technique. The method reported in this paper is the first label-free CL method for the determination of specific DNA sequences to utilize gold nanoparticles bifunctionalized with both a CL reagent and a catalytic metal complex. The sensitivity of this CL method is superior to those of most previously reported label-free methods. Compared with methods that use polymerase chain reaction amplification, this label-free CL method is much simpler, faster, and more economic. This work has thus demonstrated a simple and fast scanning strategy for the detection of specific DNA sequences related to diseases.

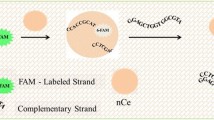
Schematic illustration of label-free CL method for detection of specific DNA sequences





Similar content being viewed by others
References
Yu X, Chai Y, Jiang J, Cui H. Sensitive ECL sensor for sequence-specific DNA from Mycobacterium tuberculosis based on N-(aminobutyl)-N-ethylisoluminol functionalized gold nanoparticles labeling. J Photochem Photobiol A Chem. 2012;241:45–51.
Daar AS, Thorsteinsdottir H, Martin DK, Smith AC, Nast S, Singer PA. Top ten biotechnologies for improving health in developing countries. Nat Genet. 2002;32(2):229–32.
Debouck C, Goodfellow PN. DNA microarrays in drug discovery and development. Nat Genet. 1999;21(1 Suppl):48–50.
Bailey RC, Nam JM, Mirkin CA, Hupp JT. Real-time multicolor DNA detection with chemoresponsive diffraction gratings and nanoparticle probes. J Am Chem Soc. 2003;125(44):13541–7.
Hong M, Zhou X, Lu Z, Zhu J. Nanoparticle-based, fluorous-tag-driven DNA detection. Angew Chem Int Ed. 2009;48(50):9503–6.
Nakayama S, Sintim HO. Colorimetric split G-quadruplex probes for nucleic acid sensing: improving reconstituted DNAzyme’s catalytic efficiency via probe remodeling. J Am Chem Soc. 2009;131(29):10320–33.
Zhang Y, Wang Y, Wang H, Jiang JH, Shen GL, Yu RQ, et al. Electrochemical DNA biosensor based on the proximity-dependent surface hybridization assay. Anal Chem. 2009;81(5):1982–7.
Fabris L, Dante M, Braun G, Seung JL, Reich NO, Moskovits M, et al. A heterogeneous PNA-based SERS method for DNA detection. J Am Chem Soc. 2007;129(19):6086–7.
Gardiner DF, Beavis KG. Laboratory diagnosis of mycobacterial infections. Semin Respir Infect. 2000;15(2):132–43.
Soini H, Musser JM. Molecular diagnosis of mycobacteria. Clin Chem. 2001;47(5):809–14.
Woods GL. Molecular methods in the detection and identification of mycobacterial infections. Arch Pathol Lab Med. 1999;123(11):1002–6.
Levidiotou S, Vrioni G, Galanakis E, Gesouli E, Pappa C, Stefanou D. Four-year experience of use of the Cobas Amplicor system for rapid detection of Mycobacterium tuberculosis complex in respiratory and nonrespiratory specimens in Greece. Eur J Clin Microbiol Infect Dis. 2003;22(6):349–56.
Lemaître N, Armand S, Vachée A, Capilliez O, Dumoulin C, Courcol RJ. Comparison of the real-time PCR method and the gen-probe amplified mycobacterium tuberculosis direct test for detection of mycobacterium tuberculosis in pulmonary and nonpulmonary specimens. J Clin Microbiol. 2004;42(9):4307–9.
Goessens WHF, de Man P, Koeleman JGM, Luijendijk A, te Witt R, Endtz HP, et al. Comparison of the COBAS AMPLICOR MTB and BDProbeTec ET assays for detection of Mycobacterium tuberculosis in respiratory specimens. J Clin Microbiol. 2005;43(6):2563–6.
Jiang J, Chai Y, Cui H. The electrogenerated chemiluminescence detection of IS6110 of Mycobacterium tuberculosis based on a luminol functionalized gold nanoprobe. RSC Advances. 2011;1(2):247–54.
Li F, Yu Y, Li Q, Zhou M, Cui H. A homogeneous signal-on strategy for the detection of rpoB genes of Mycobacterium tuberculosis based on electrochemiluminescent graphene oxide and ferrocene quenching. Anal Chem. 2014;86(3):1608–13.
Yu Y, Cao Q, Zhou M, Cui H. A novel homogeneous label-free aptasensor for 2,4,6-trinitrotoluene detection based on an assembly strategy of electrochemiluminescent graphene oxide with gold nanoparticles and aptamer. Biosens Bioelectron. 2013;43:137–42.
Li F, Cui H. A label-free electrochemiluminescence aptasensor for thrombin based on novel assembly strategy of oligonucleotide and luminol functionalized gold nanoparticles. Biosens Bioelectron. 2013;39(1):261–7.
He Y, Cui H. Label free and homogeneous histone sensing based on chemiluminescence resonance energy transfer between lucigenin and gold nanoparticles. Biosens Bioelectron. 2013;47:313–7.
Wang Z, Lee JH, Lu Y. Label-free colorimetric detection of lead ions with a nanomolar detection limit and tunable dynamic range by using gold nanoparticles and DNAzyme. Adv Mater. 2008;20(17):3263–7.
Thomas KG, Kamat PV. Chromophore-functionalized gold nanoparticles. Acc Chem Res. 2003;36(12):888–98.
Li S, Chen D, Zhou Q, Wang W, Gao L, Jiang J, et al. A general chemiluminescence strategy for measuring aptamer–target binding and target concentration. Anal Chem. 2014;86(11):5559–66.
Liu M, Zhang H, Shu J, Liu X, Li F, Cui H. Gold nanoparticles bifunctionalized by chemiluminescence reagent and catalyst metal complexes: synthesis and unique chemiluminescence property. Anal Chem. 2014;86(6):2857–61.
Wang F, Ma C, Zeng X, Li C, Deng Y, He N. Chemiluminescence molecular detection of sequence-specific HBV-DNA using magnetic nanoparticles. J Biomed Nanotechnol. 2012;8(5):786–90.
Hashimoto K, Ito K, Ishimori Y. Sequence-specific gene detection with a gold electrode modified with DNA probes and an electrochemically active dye. Anal Chem. 1994;66(21):3830–3.
Tian D, Zhang H, Chai Y, Cui H. Synthesis of N-(aminobutyl)-N-(ethylisoluminol) functionalized gold nanomaterials for chemiluminescent bio-probe. Chem Commun. 2011;47(17):4959–61.
Alric C, Taleb J, Duc GL, Mandon C, Billotey C, Meur-Herland AL, et al. Gadolinium chelate coated gold nanoparticles as contrast agents for both X-ray computed tomography and magnetic resonance imaging. J Am Chem Soc. 2008;130(18):5908–15.
Cui H, Shi M, Meng R, Zhou J, Lai C, Lin X. Effect of pH on inhibition and enhancement of luminol–H2O2–Co2+ chemiluminescence by phenolic compounds and amino acids. Photochem Photobiol. 2004;79(3):233–41.
Zhang Z, Cui H, Lai C, Liu L. Gold nanoparticle-catalyzed luminol chemiluminescence and its analytical applications. Anal Chem. 2005;77(10):3324–9.
Aoyagi S, Yamazaki M, Miyasaka T, Sakai K. Clarification of enhanced hydroxyl radical production in Fenton reaction with ATP/ADP based on luminol chemiluminescence. J Chem Eng Jpn. 2001;34(7):956–9.
Acknowledgments
The support of this research by the National Natural Science Foundation of China (grant nos. 21075115 and 21173201) and the Opening Fund of the State Key Laboratory of Electroanalytical Chemistry, Changchun Institute of Applied Chemistry, CAS (grant no. SKLEAC201408) is gratefully acknowledged.
Author contributions
All of the authors contrbuted to the writing of the manuscript, and they all approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The research was approved by the Ethical Committee of the University of Science and Technology China. All volunteers were informed of and agreed with the objectives of the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Published in the topical collection Highlights of Analytical Chemical Luminescence with guest editors Aldo Roda, Hua Cui, and Chao Lu.
Lingfeng Gao and Xiao He contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 726 kb)
Rights and permissions
About this article
Cite this article
Gao, L., He, X., Ju, L. et al. A label-free method for the detection of specific DNA sequences using gold nanoparticles bifunctionalized with a chemiluminescent reagent and a catalyst as signal reporters. Anal Bioanal Chem 408, 8747–8754 (2016). https://doi.org/10.1007/s00216-015-9244-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-9244-7