Abstract
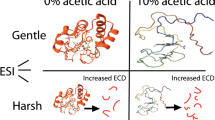
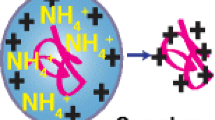
Nano-electrospray-ionization mass spectrometry (nano-ESI-MS) is employed here to describe equilibrium protein conformational transitions and to analyze the influence of instrumental settings, pH, and solvent surface tension on the charge-state distributions (CSD). A first set of experiments shows that high flow rates of N2 as curtain gas can induce unfolding of cytochrome c (cyt c) and myoglobin (Mb), under conditions in which the stability of the native protein structure has already been reduced by acidification. However, it is possible to identify conditions under which the instrumental settings are not limiting factors for the conformational stability of the protein inside ESI droplets. Under such conditions, equilibrium unfolding transitions described by ESI-MS are comparable with those obtained by other established biophysical methods. Experiments with the very stable proteins ubiquitin (Ubq) and lysozyme (Lyz) enable testing of the influence of extreme pH changes on the ESI process, uncoupled from acid-induced unfolding. When HCl is used for acidification, Ubq and Lyz mass spectra do not change between pH~7 and pH 2.2, indicating that the CSD is highly characteristic of a given protein conformation and not directly affected by even large pH changes. Use of formic or acetic acid for acidification of Ubq solutions results in major spectral changes that can be interpreted in terms of protein unfolding as a result of the increased hydrophobicity of the solvent. On the other hand, Lyz, cyt c, and Mb enable direct comparison of protein CSD (corresponding to either the folded or the unfolded protein) in HCl or acetic acid solutions at low pH. The values of surface tension for these solutions differ significantly. Confirming indications already present in the literature, we observe very similar CSD under these solvent conditions for several proteins in either compact or disordered conformations. The same is true for comparison between water and water–acetic acid for folded cyt c and Lyz. Thus, protein CSD from water–acetic solutions do not seem to be limited by the low surface tension of acetic acid as previously suggested. This result could reflect a general lack of dependence of protein CSD on the surface tension of the solvent. However, it is also possible that the effect of acetic acid on the precursor ESI droplets is smaller than generally assumed.






Similar content being viewed by others
References
Chowdhury SK, Katta V, Chait BT (1990) J Am Chem Soc 112:9012–9013
Katta V, Chait BT (1991) J Am Chem Soc 113:8534–8535
Grandori R (2003) Curr Org Chem 7:1–15
Mann M (1990) Org Mass Spectrom 25:575–587
Konermann L, Rosell FI, Mauk AG, Douglas DJ (1997) Biochemistry 36:6448–6454
Grandori R (2002) Protein Sci 11:453–458
Vis H, Heinemann U, Dobson CM, Robinson CV (1998) J Am Chem Soc 120:6427–6428
Simmons DA, Konermann L (2002) Biochemistry 41:1906–1914
Konermann L, Douglas DJ (1997) Biochemistry 36:12296–12302
Grandori R, Matečko I, Müller N (2001) J Mass Spectrom 37:191–196
Wang G, Cole RB (1997) in Electrospray Ionization Mass Spectrometry (Cole RB, Ed) John Wiley & Sons, New York p 137–174
Apostol I (1999) Anal Biochem 272:8–18
Mirza UA (1993) Anal Chem 65:1–6
Winston RL, Fitzgerald MC (1997) Mass Spectrom Rev 16:165–179
Fligge TA, Przybylski M, Quinn JP, Marshall AG (1998) Eur Mass Spectrom 4:401–404
Matečko I, Müller N, Grandori R (2002) Spectroscopy—An International Journal 16:361–371
Fenn JB (1993) J Am Soc Mass Spectrom 4:524–535
Konishi Y, Feng R (1994) Biochemistry 33:9706–9711
Kebarle P, Ho Y (1997) in Electrospray Ionization Mass Spectrometry (Cole RB, Ed) John Wiley & Sons, New York p 3–63
Dobo A, Kaltashov IA (2001) Anal Chem 73:4763–4773
Babu KR, Douglas DJ (2000) Biochemistry 39:14702–14710
Konermann L, Silva EA, Sogbein OF (2001) Anal Chem 73:4836–4844
Gatlin CL, Tureček (1994) Anal Chem 66:712–718
de la Mora JF, Van Berkel GJ, Enke CG, Cole RB, Martinez-Sanchez M, Fenn JB (2000) J Mass Spectrom 35:939–952
Van Berkel GJ, Zhou F (1995) Anal Chem 67:2916–2923
Grandori R, Matečko I, Mayr P, Müller N (2001) J Mass Spectrom 36:918–922
Peschke M, Blades A, Kebarle P (2002) J Am Chem Soc 124:11519–11530
Iavarone AT, Williams ER (2003) J Am Chem Soc 125:2319–2327
de la Mora JF (2000) Anal Chim Acta 406:93–104
Dole M, Mack LL, Hines RL, Mobley RC, Ferguson LD, Alice MB (1968) J Chem Phys 49:2240–2249
Iribarne JV, Thomson BA (1976) J Chem Phys 64:2287–2294
Goto Y, Hagihara Y, Hamada D, Hoshino M, Nishii I (1993) Biochemistry 32:11878–11885
Pan XM, Sheng XR, Zhou JM (1997) FEBS Letters 402:25–27
Konermann L, Douglas DJ (1998) J Am Soc Mass Spectrom 9:1248–1254
Loladze VV, Makhatadze GI (2002) Protein Sci 11:174–177
Sasahara K, Demura M, Nitta K (2002) Proteins 49:472–482
Kamatari YO, Konno T, Kataoka M, Akasaka K (1998) Protein Sci 7:681–688
Mao D, Babu KR, Chen Y-L, Douglas DJ (2003) Anal Chem 75:1325–1330
Goto Y, Fink AL (1990) J Mol Biol 214:803–805
Mirza UA, Chait BT (1994) Anal Chem 66:2898–2904
Takáts Z, Drahos L, Schlosser G, Vékey K (2002) Anal Chem 74:6427–6429
Le Blanc JCY, Beuchemin D, Siu KWM, Guevremont R, Berman SS (1991) Org Mass Spectrom 26:831–839
Babu KR, Moradian A, Douglas DJ (2001) J Am Soc Mass Spectrom 12:317–328
Konermann L, Douglas DJ (1998) Rapid Commun Mass Spectrom 12:435–442
Grandori R (2003) J Mass Spectrom 38:11–15
Gandini D, Gogioso L, Bolognesi M, Bordo D (1996) Proteins 24:439–449
Laurents DV, Huyghues-Despointes BMP, Bruix M, Thurlkill RL, Schell D, Newsom S, Grimsley GR, Shaw KL, Treviño S, Rico M, Briggs JM, Antosiewicz JM, Scholtz JM, Pace CN (2003) J Mol Biol 325:1077–1092
Shaw KL, Grimsley GR, Yakovlev GI, Makarov AA, Pace N (2001) Protein Sci 10:1206–1215
Huyghues-Despointes BMP, Thurlkill RL, Daily MD, Schell D, Briggs JM, Antosiewicz JM, Pace CN, Scholtz JM (2003) J Mol Biol 325:1093–1105
Yang AS, Honig B (1994) J Mol Biol 237:602–614
Yang A-S, Honig B (1993) J Mol Biol 231:459–474
Anthonsen HW, Baptista A, Drabløs F, Martel P, Petersen SB (1994) J Biotechnol 36:185–220
Sundd M, Iverson N, Ibarra-Molero B, Sanches-Ruiz JM, Robertson AD (2002) Biochemistry 41:7586–7596
Šamalikova M, Grandori R (2003) J Mass Spectrom in press
Creighton TE (1993) Proteins, structure and molecular properties 2nd ed W H Freeman and Company, New York
Shortle D, Ackerman MS (2001) Science 293:487–489
Plaxco KW, Gross M (2001) Nature Struct Biol 8:659–660
Vazquez G, Alvarez E, Navaza JM (1995) J Chem Eng Data 40:611–614
Šamalikova M, Grandori R (2003) J Am Chem Soc, 125:13352–13353
Acknowledgments
This work was supported by the grants P13906, T135 and P13511 of the Austrian Science Foundation (FWF).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Šamalikova, M., Matečko, I., Müller, N. et al. Interpreting conformational effects in protein nano-ESI-MS spectra. Anal Bioanal Chem 378, 1112–1123 (2004). https://doi.org/10.1007/s00216-003-2339-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-003-2339-6