Abstract
Background
HDAC6 is a class IIB histone deacetylase expressed at many levels of the nociceptive pathway. This study tested the ability of novel and selective HDAC6 inhibitors to alleviate sensory hypersensitivity behaviors in mouse models of peripheral nerve injury and peripheral inflammation.
Methods
We utilized the murine spared nerve injury (SNI) model for peripheral nerve injury and the Complete Freund’s Adjuvant (CFA) model of peripheral inflammation. We applied the Von Frey assay to monitor mechanical allodynia.
Results
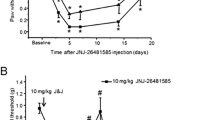
Using the SNI model, we demonstrate that daily administration of the brain-penetrant HDAC6 inhibitor, ACY-738, abolishes mechanical allodynia in male and in female mice. Importantly, there is no tolerance to the antiallodynic actions of these compounds as they produce a consistent increase in Von Frey thresholds for several weeks. We observed a similar antiallodynic effect when utilizing the HDAC6 inhibitor, ACY-257, which shows limited brain expression when administered systemically. We also demonstrate that ACY-738 and ACY-257 attenuate mechanical allodynia in the CFA model of peripheral inflammation.
Conclusions
Overall, our findings suggest that inhibition of HDAC6 provides a promising therapeutic avenue for the alleviation of mechanical allodynia associated with peripheral nerve injury and peripheral inflammation.




Similar content being viewed by others
References
Alba-Delgado C, Llorca-Torralba M, Mico JA, Berrocoso E (2018) The onset of treatment with the antidepressant desipramine is critical for the emotional consequences of neuropathic pain. Pain 159:2606–2619. https://doi.org/10.1097/j.pain.0000000000001372
Avrampou K et al (2019) RGS4 maintains chronic pain symptoms in rodent models. J Neurosci. https://doi.org/10.1523/JNEUROSCI.3154-18.2019
Bai G, Wei D, Zou S, Ren K, Dubner R (2010) Inhibition of class II histone deacetylases in the spinal cord attenuates inflammatory hyperalgesia. Mol Pain 6:51. https://doi.org/10.1186/1744-8069-6-51
Benoy V, Vanden Berghe P, Jarpe M, Van Damme P, Robberecht W, Van Den Bosch L (2017) Development of improved HDAC6 inhibitors as pharmacological therapy for axonal charcot-marie-tooth disease. Neurotherapeutics 14:417–428. https://doi.org/10.1007/s13311-016-0501-z
Bohren Y et al (2010) Mu-opioid receptors are not necessary for nortriptyline treatment of neuropathic allodynia. Eur J Pain 14:700–704. https://doi.org/10.1016/j.ejpain.2009.11.014
Bradner JE, West N, Grachan ML, Greenberg EF, Haggarty SJ, Warnow T, Mazitschek R (2010) Chemical phylogenetics of histone deacetylases. Nat Chem Biol 6:238–243. https://doi.org/10.1038/nchembio.313
Choucair-Jaafar N, Salvat E, Freund-Mercier MJ, Barrot M (2014) The antiallodynic action of nortriptyline and terbutaline is mediated by beta(2) adrenoceptors and delta opioid receptors in the ob/ob model of diabetic polyneuropathy. Brain Res 1546:18–26. https://doi.org/10.1016/j.brainres.2013.12.016
Cruccu G (2007) Treatment of painful neuropathy. Curr Opin Neurol 20:531–535. https://doi.org/10.1097/WCO.0b013e328285dfd6
D’Amour FE, Smith DL (1941) Amethos for determining loss of pain sensation. J Pharmacol Exp Ther:74–79
Denk F et al (2013) HDAC inhibitors attenuate the development of hypersensitivity in models of neuropathic. Pain Pain 154:1668–1679. https://doi.org/10.1016/j.pain.2013.05.021
Descalzi G, Ikegami D, Ushijima T, Nestler EJ, Zachariou V, Narita M (2015) Epigenetic mechanisms of chronic pain. Trends Neurosci 38:237–246. https://doi.org/10.1016/j.tins.2015.02.001
Descalzi G, Mitsi V, Purushothaman I, Gaspari S, Avrampou K, Loh YHE, Shen L, Zachariou V (2017) Neuropathic pain promotes adaptive changes in gene expression in brain networks involved in stress and depression. Sci Signal 10:10. https://doi.org/10.1126/scisignal.aaj1549
Falkenberg KJ, Johnstone RW (2014) Histone deacetylases and their inhibitors in cancer, neurological diseases and immune disorders. Nat Rev Drug Discov 13:673–691. https://doi.org/10.1038/nrd4360
Fukada M et al (2012) Loss of deacetylation activity of Hdac6 affects emotional behavior in mice. PLoS One 7:e30924. https://doi.org/10.1371/journal.pone.0030924
Gaspari S et al (2018) Suppression of RGSz1 function optimizes the actions of opioid analgesics by mechanisms that involve the Wnt/beta-catenin pathway. Proc Natl Acad Sci USA 115:E2085–E2094. https://doi.org/10.1073/pnas.1707887115
Haggarty SJ, Koeller KM, Wong JC, Grozinger CM, Schreiber SL (2003) Domain-selective small-molecule inhibitor of histone deacetylase 6 (HDAC6)-mediated tubulin deacetylation. Proc Natl Acad Sci USA 100:4389–4394. https://doi.org/10.1073/pnas.0430973100
Hubbert C et al (2002) HDAC6 is a microtubule-associated deacetylase. Nature 417:455–458. https://doi.org/10.1038/417455a
Jamison RN, Mao J (2015) Opioid analgesics. Mayo Clin Proc 90:957–968. https://doi.org/10.1016/j.mayocp.2015.04.010
Jochems J et al (2014) Antidepressant-like properties of novel HDAC6-selective inhibitors with improved brain bioavailability. Neuropsychopharmacology 39:389–400. https://doi.org/10.1038/npp.2013.207
Jochems J et al (2015) Enhancement of stress resilience through histone deacetylase 6-mediated regulation of glucocorticoid receptor chaperone dynamics. Biol Psychiatry 77:345–355. https://doi.org/10.1016/j.biopsych.2014.07.036
Jones SS, Jarpe MB (2013) Specific regulation of cytokine levels by HDAC6 inhibitors. United States Patent (US 2013/0225543 A1)
Kremer M, Salvat E, Muller A, Yalcin I, Barrot M (2016) Antidepressants and gabapentinoids in neuropathic pain: mechanistic insights. Neuroscience 338:183–206. https://doi.org/10.1016/j.neuroscience.2016.06.057
Krukowski K et al (2017) HDAC6 inhibition effectively reverses chemotherapy-induced peripheral neuropathy. Pain 158:1126–1137. https://doi.org/10.1097/j.pain.0000000000000893
Ling W, Mooney L, Hillhouse M (2011) Prescription opioid abuse, pain and addiction: clinical issues and implications. Drug Alcohol Rev 30:300–305. https://doi.org/10.1111/j.1465-3362.2010.00271.x
Ma J, Huo X, Jarpe MB, Kavelaars A, Heijnen CJ (2018) Pharmacological inhibition of HDAC6 reverses cognitive impairment and tau pathology as a result of cisplatin treatment. Acta Neuropathol Commun 6:103. https://doi.org/10.1186/s40478-018-0604-3
Ma J, Trinh RT, Mahant ID, Peng B, Matthias P, Heijnen CJ, Kavelaars A (2019) Cell-specific role of histone deacetylase 6 in chemotherapy-induced mechanical allodynia and loss of intraepidermal nerve fibers. Pain 160:2877–2890. https://doi.org/10.1097/j.pain.0000000000001667
Magni G (1991) The use of antidepressants in the treatment of chronic pain. A review of the current evidence. Drugs 42:730–748. https://doi.org/10.2165/00003495-199142050-00002
Mao Y, Zhou J, Liu X, Gu E, Zhang Z, Tao W (2019) Comparison of different histone deacetylase inhibitors in attenuating inflammatory pain in rats pain. Res Manag 2019:1648919. https://doi.org/10.1155/2019/1648919
Matsushita Y, Araki K, Omotuyi O, Mukae T, Ueda H (2013) HDAC inhibitors restore C-fibre sensitivity in experimental neuropathic pain model. Br J Pharmacol 170:991–998. https://doi.org/10.1111/bph.12366
Matthes HW et al (1996) Loss of morphine-induced analgesia, reward effect and withdrawal symptoms in mice lacking the mu-opioid-receptor gene. Nature 383:819–823. https://doi.org/10.1038/383819a0
Megat S et al (2015) Kappa-opioid receptors are not necessary for the antidepressant treatment of neuropathic pain. Br J Pharmacol 172:1034–1044. https://doi.org/10.1111/bph.12963
Mitsi V, Zachariou V (2016) Modulation of pain, nociception, and analgesia by the brain reward center. Neuroscience 338:81–92. https://doi.org/10.1016/j.neuroscience.2016.05.017
Mitsi V et al (2015) RGS9-2--controlled adaptations in the striatum determine the onset of action and efficacy of antidepressants in neuropathic pain states. Proc Natl Acad Sci USA 112:E5088–E5097. https://doi.org/10.1073/pnas.1504283112
Morris MJ, Monteggia LM (2013) Unique functional roles for class I and class II histone deacetylases in central nervous system development and function. Int J Dev Neurosci 31:370–381. https://doi.org/10.1016/j.ijdevneu.2013.02.005
Ossipov MH, Lai J, Malan TP Jr, Porreca F (2000) Spinal and supraspinal mechanisms of neuropathic pain. Ann N Y Acad Sci 909:12–24. https://doi.org/10.1111/j.1749-6632.2000.tb06673.x
Reisner L (2003) Antidepressants for chronic neuropathic pain. Curr Pain Headache Rep 7:24–33
Russo SJ, Nestler EJ (2013) The brain reward circuitry in mood disorders. Nat Rev Neurosci 14:609–625. https://doi.org/10.1038/nrn3381
Schloesser RJ, Martinowich K, Manji HK (2012) Mood-stabilizing drugs: mechanisms of action. Trends Neurosci 35:36–46. https://doi.org/10.1016/j.tins.2011.11.009
Seidel C, Schnekenburger M, Dicato M, Diederich M (2015) Histone deacetylase 6 in health and disease. Epigenomics 7:103–118. https://doi.org/10.2217/epi.14.69
Shields SD, Eckert WA 3rd, Basbaum AI (2003) Spared nerve injury model of neuropathic pain in the mouse: a behavioral and anatomic analysis. J Pain 4:465–470
Sultan A, Gaskell H, Derry S, Moore RA (2008) Duloxetine for painful diabetic neuropathy and fibromyalgia pain: systematic review of randomised trials. BMC Neurol 8:29. https://doi.org/10.1186/1471-2377-8-29
Suzuki R, Dickenson A (2005) Spinal and supraspinal contributions to central sensitization in peripheral neuropathy. Neurosignals 14:175–181. https://doi.org/10.1159/000087656
Tao W, Chen Q, Wang L, Zhou W, Wang Y, Zhang Z (2015) Brainstem brain-derived neurotrophic factor signaling is required for histone deacetylase inhibitor-induced pain relief. Mol Pharmacol 87:1035–1041. https://doi.org/10.1124/mol.115.098186
Van Helleputte L et al (2018) Inhibition of histone deacetylase 6 (HDAC6) protects against vincristine-induced peripheral neuropathies and inhibits tumor growth. Neurobiol Dis 111:59–69. https://doi.org/10.1016/j.nbd.2017.11.011
Volkow ND, Jones EB, Einstein EB, Wargo EM (2019) Prevention and treatment of opioid misuse and addiction: a review. JAMA Psychiatry 76:208–216. https://doi.org/10.1001/jamapsychiatry.2018.3126
Webster LR (2017) Risk factors for opioid-use disorder and overdose. Anesth Analg 125:1741–1748. https://doi.org/10.1213/ANE.0000000000002496
Yalcin I et al (2009) Beta(2)-adrenoceptors are critical for antidepressant treatment of neuropathic pain. Ann Neurol 65:218–225. https://doi.org/10.1002/ana.21542
Zammataro M, Sortino MA, Parenti C, Gereau RW, Chiechio S (2014) HDAC and HAT inhibitors differently affect analgesia mediated by group II metabotropic glutamate receptors. Mol Pain 10:68. https://doi.org/10.1186/1744-8069-10-68
Ziegler D, Fonseca V (2015) From guideline to patient: a review of recent recommendations for pharmacotherapy of painful diabetic neuropathy. J Diabetes Complicat 29:146–156. https://doi.org/10.1016/j.jdiacomp.2014.08.008
Funding
This study was supported by National Institute of Neurological Disorders and Stroke NS086444 (V.Z.), NS093537 (V.Z.), and National Institute on Drug Abuse PPG- POIDAO8227 (V.Z.), NIH T32 5T32DA007135-34 (KP) NIH T32 GM007280 (RAS). Randal A. Serafini is a recipient of the Irene and Eric Simon Brain Research Foundation Summer Fellowship (2019). K. Avrampou is a graduate student in the Molecular Biology and Biomedine program of the University of Crete, Greece.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sakloth, F., Manouras, L., Avrampou, K. et al. HDAC6-selective inhibitors decrease nerve-injury and inflammation-associated mechanical hypersensitivity in mice. Psychopharmacology 237, 2139–2149 (2020). https://doi.org/10.1007/s00213-020-05525-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05525-9