Abstract
Rationale
Prolonged use of nicotine appears to enhance incentive salience, a motivational-cognitive process that transforms an otherwise neutral stimulus into a “wanted” stimulus. It has been suggested that nicotinic enhancement of incentive salience contributes to the potential of relapse in individuals with tobacco addiction. However, there are two main limitations of prior research that caution this claim: (a) the use of passive experimentally delivered nicotine and (b) the use of sign-tracking as an index of incentive salience, without acknowledging the competing nature of goal- and sign-tracking responses.
Objectives
To determine whether nicotinic enhancement of incentive salience attributed to non-nicotinic stimuli occurs when rats self-administer nicotine, and whether it is facilitated by a prior history of nicotine self-administration.
Methods
Twenty-three male rats were trained daily, for 24 days, on a nicotine self-administration (SA) paradigm in the morning, and on a four-conditioned-stimuli Pavlovian conditioned approach (4-CS PCA) task in the afternoon. Self-administration was followed by extinction and cue reinstatement. A subcutaneous nicotine challenge was performed during the last 7 days of the study.
Results
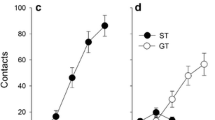
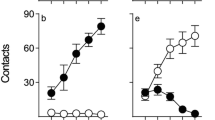
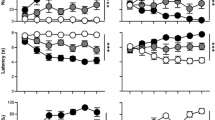
Nicotine self-administration selectively enhanced sign-tracking in the 4-CS PCA. Upon extinction, sign-tracking quickly declined to control levels. Experimenter-administered nicotine enhanced sign-tracking similarly regardless of nicotine history.
Conclusions
The results suggest that nicotinic enhancement of incentive salience is transient, and a previous history of nicotine use does not cause further sensitization. Taken together, these results suggest that nicotine enhances incentive salience, particularly—and perhaps exclusively—while onboard.






Similar content being viewed by others
References
Barrett ST, Odum AL (2011) The effects of repeated exposure on the reward-enhancing effects of nicotine. Behav Pharmacol 22(4):283–290. https://doi.org/10.1097/FBP.0b013e3283473c25
Beckmann JS, Chow JJ (2015) Isolating the incentive salience of reward-associated stimuli: value, choice, and persistence. Learn Mem 22(2):116–127. https://doi.org/10.1101/lm.037382.114
Benowitz NL (2010) Nicotine addiction. N Engl J Med 362(24):2295–2303. https://doi.org/10.1056/NEJMra0809890
Berridge KC (2007) The debate over dopamine’s role in reward: the case for incentive salience. Psychopharmacology 191(3):391–431. https://doi.org/10.1007/s00213-006-0578-x
Berridge KC (2012) From prediction error to incentive salience: mesolimbic computation of reward motivation. Eur J Neurosci 35(7):1124–1143. https://doi.org/10.1111/j.1460-9568.2012.07990.x
Berridge KC, Robinson TE (1998) What is the role of dopamine in reward: hedonic impact, reward learning, or incentive salience? Brain Res Rev 28(3):309–369. https://doi.org/10.1016/S0165-0173(98)00019-8
Berridge KC, Robinson TE (2016) Liking, wanting, and the incentive-sensitization theory of addiction. Am Psychol 71(8):670–679. https://doi.org/10.1037/amp0000059
Berridge KC, Robinson TE, Aldridge JW (2009) Dissecting components of reward: ‘liking’,‘wanting’, and learning. Curr Opin Pharmacol 9(1):65–73. https://doi.org/10.1016/j.coph.2008.12.014
Caggiula AR, Donny EC, Palmatier MI, Liu X, Chaudhri N, Sved AF (2008) The role of nicotine in smoking: a dual-reinforcement model. In: The motivational impact of nicotine and its role in tobacco use. Springer, New York, pp 91–109. https://doi.org/10.1007/978-0-387-78748-0_6
Changeux JP (2010) Nicotine addiction and nicotinic receptors: lessons from genetically modified mice. Nat Rev Neurosci 11(6):389–401. https://doi.org/10.1038/nrn2849
Chow JJ, Nickell JR, Darna M, Beckmann JS (2016) Toward isolating the role of dopamine in the acquisition of incentive salience attribution. Neuropharmacology 109:320–331. https://doi.org/10.1016/j.neuropharm.2016.06.028
Clemens KJ, Caillé S, Cador M (2010) The effects of response operandum and prior food training on intravenous nicotine self-administration in rats. Psychopharmacology 211(1):43–54. https://doi.org/10.1007/s00213-010-1866-z
Daniels CW, Sanabria F (2017) About bouts: A heterogeneous tandem schedule of reinforcement reveals dissociable components of operant behavior in Fischer rats. Journal of Experimental Psychology: Animal Learning and Cognition 43(3):280
Daniels CW, Watterson E, Garcia R, Mazur GJ, Brackney RJ, Sanabria F (2015) Revisiting the effect of nicotine on interval timing. Behav Brain Res 283:238–250. https://doi.org/10.1016/j.bbr.2015.01.027
Davey GC, Cleland GG, Oakley DA (1982) Applying Konorski’s model of classical conditioning to signal-centered behavior in the rat: some functional similarities between hunger CRs and sign-tracking. Learn Behav 10(2):257–262. https://doi.org/10.3758/BF03212279
Flagel SB, Watson SJ, Robinson TE, Akil H (2007) Individual differences in the propensity to approach signals vs goals promote different adaptations in the dopamine system of rats. Psychopharmacology 191(3):599–607. https://doi.org/10.1007/s00213-006-0535-8
Flagel SB, Akil H, Robinson TE (2009) Individual differences in the attribution of incentive salience to reward-related cues: implications for addiction. Neuropharmacology 56:139–148. https://doi.org/10.1016/j.neuropharm.2008.06.027
Flagel SB, Clark JJ, Robinson TE, Mayo L, Czuj A, Willuhn I, Akers CA, Clinton SM, Phillips PE, Akil H (2011) A selective role for dopamine in reward learning. Nature 469(7328):53–57. https://doi.org/10.1038/nature09588
Flores CM, Dávila-García MI, Ulrich YM, Kellar KJ (1997) Differential regulation of neuronal nicotinic receptor binding sites following chronic nicotine administration. J Neurochem 69(5):2216–2219
George TP, O’Malley SS (2004) Current pharmacological treatments for nicotine dependence. Trends Pharmacol Sci 25(1):42–48. https://doi.org/10.1016/j.tips.2003.11.003
Goeders JE, Murnane KS, Banks ML, Fantegrossi WE (2009) Escalation of food-maintained responding and sensitivity to the locomotor stimulant effects of cocaine in mice. Pharmacol Biochem Behav 93(1):67–74. https://doi.org/10.1016/j.pbb.2009.04.008
Guy EG, Fletcher PJ (2013) Nicotine-induced enhancement of responding for conditioned reinforcement in rats: role of prior nicotine exposure and α4β2 nicotinic receptors. Psychopharmacology 225(2):429–440. https://doi.org/10.1007/s00213-012-2832-8
Guy EG, Fletcher PJ (2014) The effects of nicotine exposure during Pavlovian conditioning in rats on several measures of incentive motivation for a conditioned stimulus paired with water. Psychopharmacology 231(11):2261–2271. https://doi.org/10.1007/s00213-013-3375-3
Holland PC, Asem JS, Galvin CP, Keeney CH, Hsu M, Miller A, Zhou V (2014) Blocking in autoshaped lever-pressing procedures with rats. Learn Behav 42(1):1–21. https://doi.org/10.3758/s13420-013-0120-z
Jarosz AF, Wiley J (2014) What are the odds? A practical guide to computing and reporting Bayes factors. J Probl Solving 7(1):2
JASP Team (2017) JASP (Version 0.8.1.1)[Computer software]
Kass RE, Raftery AE (1995) Bayes factors. J Am Stat Assoc 90(430):773–795. https://doi.org/10.1080/01621459.1995.10476572
Laviolette SR, Van Der Kooy D (2004) The neurobiology of nicotine addiction: bridging the gap from molecules to behaviour. Nat Rev Neurosci 5(1):55–65. https://doi.org/10.1038/nrn1298
Lopez JC, Karlsson RM, O'donnell P (2015) Dopamine D2 modulation of sign and goal tracking in rats. Neuropsychopharmacology 40(9):2096–2102. https://doi.org/10.1038/npp.2015.68
Marks MJ, Grady SR, Collins AC (1993) Downregulation of nicotinic receptor function after chronic nicotine infusion. J Pharmacol Exp Ther 266(3):1268–1276
McClure SM, Daw ND, Montague PR (2003) A computational substrate for incentive salience. Trends Neurosci 26(8):423–428. https://doi.org/10.1016/S0166-2236(03)00177-2
Moretti M, Mugnaini M, Tessari M, Zoli M, Gaimarri A, Manfredi I, Pistillo F, Clementi F, Gotti C (2010) A comparative study of the effects of the intravenous self-administration or subcutaneous minipump infusion of nicotine on the expression of brain neuronal nicotinic receptor subtypes. Mol Pharmacol 78(2):287–296. https://doi.org/10.1124/mol.110.064071
Olausson P, Jentsch JD, Taylor JR (2004) Nicotine enhances responding with conditioned reinforcement. Psychopharmacology 171(2):173–178. https://doi.org/10.1007/s00213-003-1575-y
Palmatier MI, Evans-Martin FF, Hoffman A, Caggiula AR, Chaudhri N, Donny EC, Liu X, Booth S, Gharib M, Craven L, Sved AF (2006) Dissociating the primary reinforcing and reinforcement-enhancing effects of nicotine using a rat self-administration paradigm with concurrently available drug and environmental reinforcers. Psychopharmacology 184(3–4):391–400. https://doi.org/10.1007/s00213-005-0183-4
Palmatier MI, Matteson GL, Black JJ, Liu X, Caggiula AR, Craven L, Donny EC, Sved AF (2007) The reinforcement enhancing effects of nicotine depend on the incentive value of non-drug reinforcers and increase with repeated drug injections. Drug Alcohol Depend 89(1):52–59. https://doi.org/10.1016/j.drugalcdep.2006.11.020
Palmatier MI, Marks KR, Jones SA, Freeman KS, Wissman KM, Sheppard AB (2013) The effect of nicotine on sign-tracking and goal-tracking in a Pavlovian conditioned approach paradigm in rats. Psychopharmacology 226(2):247–259. https://doi.org/10.1007/s00213-012-2892-9
Palmatier MI, Kellicut MR, Sheppard AB, Brown RW, Robinson DL (2014) The incentive amplifying effects of nicotine are reduced by selective and non-selective dopamine antagonists in rats. Pharmacol Biochem Behav 126:50–62. https://doi.org/10.1016/j.pbb.2014.08.012
Panlilio LV, Hogarth L, Shoaib M (2015) Concurrent access to nicotine and sucrose in rats. Psychopharmacology 232(8):1451–1460. https://doi.org/10.1007/s00213-014-3787-8
Peartree NA, Sanabria F, Thiel KJ, Weber SM, Cheung TH, Neisewander JL (2012) A new criterion for acquisition of nicotine self-administration in rats. Drug Alcohol Depend 124(1):63–69. https://doi.org/10.1016/j.drugalcdep.2011.12.011
Picciotto MR (2003) Nicotine as a modulator of behavior: beyond the inverted U. Trends Pharmacol Sci 24(9):493–499. https://doi.org/10.1016/S0165-6147(03)00230-X
Raiff BR, Dallery J (2008) The generality of nicotine as a reinforcer enhancer in rats: effects on responding maintained by primary and conditioned reinforcers and resistance to extinction. Psychopharmacology 201(2):305–314. https://doi.org/10.1007/s00213-008-1282-9
Robinson TE, Berridge KC (1993) The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Rev 18(3):247–291. https://doi.org/10.1016/0165-0173(93)90013-P
Robinson TE, Berridge KC (2008) The incentive sensitization theory of addiction: some current issues. Phil Trans R Soc Lond B Biol Sci 363(1507):3137–3146. https://doi.org/10.1098/rstb.2008.0093
Stringfield SJ, Palmatier MI, Boettiger CA, Robinson DL (2017) Orbitofrontal participation in sign-and goal-tracking conditioned responses: effects of nicotine. Neuropharmacology 116:208–223
Tomie A, Grimes KL, Pohorecky LA (2008) Behavioral characteristics and neurobiological substrates shared by Pavlovian sign-tracking and drug abuse. Brain Res Rev 58(1):121–135. https://doi.org/10.1016/j.brainresrev.2007.12.003
Versaggi CL, King CP, Meyer PJ (2016) The tendency to sign-track predicts cue-induced reinstatement during nicotine self-administration, and is enhanced by nicotine but not ethanol. Psychopharmacology 233(15–16):2985–2997. https://doi.org/10.1007/s00213-016-4341-7
Wonnacott S, Sidhpura N, Balfour DJ (2005) Nicotine: from molecular mechanisms to behaviour. Curr Opin Pharmacol 5(1):53–59. https://doi.org/10.1016/j.coph.2004.12.002
Yager LM, Robinson TE (2015) Individual variation in the motivational properties of a nicotine cue: sign-trackers vs. goal-trackers. Psychopharmacology 232(17):3149–3160. https://doi.org/10.1007/s00213-015-3962-6
Zhang J, Berridge KC, Tindell AJ, Smith KS, Aldridge JW (2009) A neural computational model of incentive salience. PLoS Comput Biol 5(7):e1000437. https://doi.org/10.1371/journal.pcbi.1000437
Acknowledgements
Portions of this research were presented at the 2017 meeting of the Society for the Quantitative Analysis of Behavior in Denver, CO. We would like to thank Dr. Joshua Beckmann for his helpful insight and discussions pertaining to this project, Dr. Foster Olive for use of his equipment, and Korinna Romero and Ryan Becker for their help with data collection. Armani Del Franco is now a graduate student at the University of Minnesota.
Funding
This research was supported by a grant from the National Institutes of Health (5R00DA036569-04) awarded to Dr. Cassandra Gipson, funds from Barrett, The Honors College, awarded to Paula F. Overby, and by the Western Alliance to Expand Student Opportunities (WAESO).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Overby, P.F., Daniels, C.W., Del Franco, A. et al. Effects of nicotine self-administration on incentive salience in male Sprague Dawley rats. Psychopharmacology 235, 1121–1130 (2018). https://doi.org/10.1007/s00213-018-4829-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-4829-4