Abstract
Background
Despite evidence supporting the benefits of cannabinoids for symptom control across a wide range of medical conditions, concerns have been raised regarding the potential misuse and/or problematic use of cannabinoids (CBs).
Objective
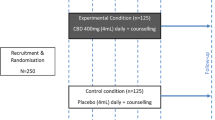
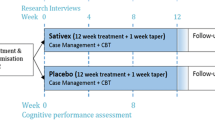
The first objective of this study was to examine the incidence of problematic prescription cannabinoid use (PPCBU) over a 12-month period among patients initiating cannabinoid therapy. The second objective was to examine the factors associated with PPCBU. A total of 265 patients who were prescribed oral cannabinoid therapy as part of usual medical practice were enrolled into this prospective observational study. Patients first completed a series of baseline questionnaires assessing demographic, clinical, and substance use variables. Three measures designed to assess PPCBU were then administered at 3, 6, and 12 months after initiation of cannabinoid therapy.
Results
At each of the follow-up assessment time points, a significantly greater number of patients scored below (vs above) cutoff scores on the three main PPCBU outcomes (all p’s < .001). At any follow-up time point, a maximum of roughly 25% of patients demonstrated PPCBU. Heightened odds of PPCBU were observed among patients with a history of psychiatric problems, tobacco smokers, and recreational cannabis users (all p’s < .05). Results indicated that past-year substance abuse, assessed using the DAST-20, was the strongest predictor of PPCBU (p < .005).
Conclusion
Findings from the present study could have implications for clinicians considering the use of cannabinoids for the management of patients with medical conditions. Although results indicated that the majority of patients included in this study did not reach cutoff scores on the three main PPCBU outcomes, our findings suggest that PPCBU should be routinely assessed and monitored over the course of cannabinoid therapy, particularly among patients with a history of psychiatric or substance use problems.
Similar content being viewed by others
References
Abrams DI, Jay CA, Shade SB, Vizoso H, Reda H, Press S, Kelly ME, Rowbotham MC, Petersen KL (2007) Cannabis in painful HIV-associated sensory neuropathy: a randomized placebo-controlled trial. Neurology 68(7):515–521. https://doi.org/10.1212/01.wnl.0000253187.66183.9c
Aggarwal SK (2013) Cannabinergic pain medicine: a concise clinical primer and survey of randomized-controlled trial results. Clin J Pain 29(2):162–171. https://doi.org/10.1097/AJP.0b013e31824c5e4c
Ballantyne JC, LaForge KS (2007) Opioid dependence and addiction during opioid treatment of chronic pain. Pain 129(3):235–255. https://doi.org/10.1016/j.pain.2007.03.028
Boscarino JA, Rukstalis M, Hoffman SN, Han JJ, Erlich PM, Gerhard GS, Stewart WF (2010) Risk factors for drug dependence among out-patients on opioid therapy in a large US health-care system. Addiction 105(10):1776–1782. https://doi.org/10.1111/j.1360-0443.2010.03052.x
Butler SF, Budman SH, Fernandez KC, Houle B, Benoit C, Katz N, Jamison RN (2007) Development and validation of the Current Opioid Misuse Measure. Pain 130(1):144–156. https://doi.org/10.1016/j.pain.2007.01.014
Butler SF, Budman SH, Fanciullo GJ, Jamison RN (2010) Cross validation of the current opioid misuse measure to monitor chronic pain patients on opioid therapy. Clin J Pain 26(9):770–776. https://doi.org/10.1097/AJP.0b013e3181f195ba
Carliner H, Brown QL, Sarvet AL, Hasin DS (2017) Cannabis use, attitudes, and legal status in the U.S.: a review. Prev Med. https://doi.org/10.1016/j.ypmed.2017.07.008
Chabal C, Erjavec MK, Jacobson L, Mariano A, Chaney E (1997) Prescription opiate abuse in chronic pain patients: clinical criteria, incidence, and predictors. Clin J Pain 13(2):150–155. https://doi.org/10.1097/00002508-199706000-00009
Chou R, Fanciullo GJ, Fine PG, Adler JA, Ballantyne JC, Davies P, Donovan MI, Fishbain DA, Foley KM, Fudin J, Gilson AM, Kelter A, Mauskop A, O'Connor PG, Passik SD, Pasternak GW, Portenoy RK, Rich BA, Roberts RG, Todd KH, Miaskowski C (2009) Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain. J Pain 10(2):113–130. https://doi.org/10.1016/j.jpain.2008.10.008
Collin C, Davies P, Mutiboko IK, Ratcliffe S (2007) Randomized controlled trial of cannabis-based medicine in spasticity caused by multiple sclerosis. Eur J Neurol 14(3):290–296. https://doi.org/10.1111/j.1468-1331.2006.01639.x
Collin C, Ehler E, Waberzinek G, Alsindi Z, Davies P, Powell K, Notcutt W, O’Leary C, Ratcliffe S, Novakova I, Zapletalova O, Pikova J, Ambler Z (2010) A double-blind, randomized, placebo-controlled, parallel-group study of Sativex, in subjects with symptoms of spasticity due to multiple sclerosis. Neurol Res 32(5):451–459. https://doi.org/10.1179/016164109X12590518685660
Compton WM, Grant BF, Colliver JD, Glantz MD, Stinson FS (2004) Prevalence of marijuana use disorders in the United States: 1991–1992 and 2001–2002. JAMA 291(17):2114–2121. https://doi.org/10.1001/jama.291.17.2114
Compton WM, Han B, Hughes A, Jones CM, Blanco C (2017) Use of marijuana for medical purposes among adults in the United States. JAMA 317(2):209–211. https://doi.org/10.1001/jama.2016.18900
Corroon JM Jr, Mischley LK, Sexton M (2017) Cannabis as a substitute for prescription drugs—a cross-sectional study. J Pain Res 10:989–998. https://doi.org/10.2147/JPR.S134330
Edlund MJ, Steffick D, Hudson T, Harris KM, Sullivan M (2007) Risk factors for clinically recognized opioid abuse and dependence among veterans using opioids for chronic non-cancer pain. Pain 129(3):355–362. https://doi.org/10.1016/j.pain.2007.02.014
Edlund MJ, Sullivan MD, Han X, Booth BM (2013) Days with pain and substance use disorders: is there an association? Clin J Pain 29(8):689–695. https://doi.org/10.1097/AJP.0b013e318270fa77
Furlan AD, Reardon R, Weppler C (2010) Opioids for chronic noncancer pain: a new Canadian practice guideline. CMAJ 182(9):923–930. https://doi.org/10.1503/cmaj.100187
Garland EL, Brown SM, Howard MO (2016) Thought suppression as a mediator of the association between depressed mood and prescription opioid craving among chronic pain patients. J Behav Med 39(1):128–138. https://doi.org/10.1007/s10865-015-9675-9
Grattan A, Sullivan MD, Saunders KW, Campbell CI, Von Korff MR (2012) Depression and prescription opioid misuse among chronic opioid therapy recipients with no history of substance abuse. Ann Fam Med 10(4):304–311. https://doi.org/10.1370/afm.1371
Guy W (1976) Assessment manual for psychopharmacology—revised. Department of Health, Education, and Welfare Public Health Service, Alcohol, Drug Abuse, and Mental Health Administration, NIMH Psychopharmacology Research Branch, Division of Extramural Research Programs, Rockville, MD, pp 218–222
Haney M, Rabkin J, Gunderson E, Foltin RW (2005) Dronabinol and marijuana in HIV(+) marijuana smokers: acute effects on caloric intake and mood. Psychopharmacology 181(1):170–178. https://doi.org/10.1007/s00213-005-2242-2
Haney M, Gunderson EW, Rabkin J, Hart CL, Vosburg SK, Comer SD, Foltin RW (2007) Dronabinol and marijuana in HIV-positive marijuana smokers. Caloric intake, mood, and sleep. J Acquir Immune Defic Syndr (1999) 45(5):545–554. https://doi.org/10.1097/QAI.0b013e31811ed205
Hasin DS (2017) US Epidemiology of Cannabis Use and Associated Problems. Neuropsychopharmacology 43(1):195-212
Hasin DS, Saha TD, Kerridge BT, Goldstein RB, Chou SP, Zhang H, Jung J, Pickering RP, Ruan WJ, Smith SM, Huang B, Grant BF (2015) Prevalence of marijuana use disorders in the United States between 2001–2002 and 2012–2013. JAMA Psychiatry 72(12):1235–1242. https://doi.org/10.1001/jamapsychiatry.2015.1858
Hasin DS, Kerridge BT, Saha TD, Huang B, Pickering R, Smith SM, Jung J, Zhang H, Grant BF (2016) Prevalence and correlates of DSM-5 cannabis use disorder, 2012-2013: findings from the National Epidemiologic Survey on Alcohol and Related Conditions-III. Am J Psychiatry 173(6):588–599. https://doi.org/10.1176/appi.ajp.2015.15070907
Hurst H, Bolton J (2004) Assessing the clinical significance of change scores recorded on subjective outcome measures. J Manip Physiol Ther 27(1):26–35. https://doi.org/10.1016/j.jmpt.2003.11.003
Ives TJ, Chelminski PR, Hammett-Stabler CA, Malone RM, Perhac JS, Potisek NM, Shilliday BB, DeWalt DA, Pignone MP (2006) Predictors of opioid misuse in patients with chronic pain: a prospective cohort study. BMC Health Serv Res 6(1):46. https://doi.org/10.1186/1472-6963-6-46
Jamison RN, Serraillier J, Michna E (2011) Assessment and treatment of abuse risk in opioid prescribing for chronic pain. Pain Res Treat 2011:941808
Johnson JR, Burnell-Nugent M, Lossignol D, Ganae-Motan ED, Potts R, Fallon MT (2010) Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J Pain Symptom Manag 39(2):167–179. https://doi.org/10.1016/j.jpainsymman.2009.06.008
Kahan M, Srivastava A (2007) Is there a role for marijuana in medical practice? No. Can Fam Physician Medecin de famille canadien 53(1):22–25
Kalant H (2004) Adverse effects of cannabis on health: an update of the literature since 1996. Prog Neuro-Psychopharmacol Biol Psychiatry 28(5):849–863. https://doi.org/10.1016/j.pnpbp.2004.05.027
Kirsh KL, Jass C, Bennett DS, Hagen JE, Passik SD (2007) Initial development of a survey tool to detect issues of chemical coping in chronic pain patients. Palliat Support Care 5(3):219–226
Kouyanou K, Pither CE, Wessely S (1997) Medication misuse, abuse and dependence in chronic pain patients. J Psychosom Res 43(5):497–504. https://doi.org/10.1016/S0022-3999(97)00171-2
Liebschutz JM, Saitz R, Weiss RD, Averbuch T, Schwartz S, Meltzer EC, Claggett-Borne E, Cabral H, Samet JH (2010) Clinical factors associated with prescription drug use disorder in urban primary care patients with chronic pain. J Pain 11(11):1047–1055. https://doi.org/10.1016/j.jpain.2009.10.012
Martel MO, Dolman AJ, Edwards RR, Jamison RN, Wasan AD (2014) The association between negative affect and prescription opioid misuse in patients with chronic pain: the mediating role of opioid craving. J Pain 15(1):90–100. https://doi.org/10.1016/j.jpain.2013.09.014
Martel MO, Finan PH, McHugh RK, Issa M, Edwards RR, Jamison RN, Wasan AD (2016) Day-to-day pain symptoms are only weakly associated with opioid craving among patients with chronic pain prescribed opioid therapy. Drug Alcohol Depend 162:130–136. https://doi.org/10.1016/j.drugalcdep.2016.02.047
Michna E, Ross EL, Hynes WL, Nedeljkovic SS, Soumekh S, Janfaza D, Palombi D, Jamison RN (2004) Predicting aberrant drug behavior in patients treated for chronic pain: importance of abuse history. J Pain Symptom Manag 28(3):250–258. https://doi.org/10.1016/j.jpainsymman.2004.04.007
Narang S, Gibson D, Wasan AD, Ross EL, Michna E, Nedeljkovic SS, Jamison RN (2008) Efficacy of dronabinol as an adjuvant treatment for chronic pain patients on opioid therapy. J Pain 9(3):254–264. https://doi.org/10.1016/j.jpain.2007.10.018
Nielsen S, Lintzeris N, Bruno R, Campbell G, Larance B, Hall W, Hoban B, Cohen ML, Degenhardt L (2015) Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med (Malden, Mass) 16(2):356–366. https://doi.org/10.1111/pme.12594
Notcutt W, Langford R, Davies P, Ratcliffe S, Potts R (2012) A placebo-controlled, parallel-group, randomized withdrawal study of subjects with symptoms of spasticity due to multiple sclerosis who are receiving long-term Sativex(R) (nabiximols). Mult Scler (Houndmills, Basingstoke, England) 18(2):219–228. https://doi.org/10.1177/1352458511419700
Osborn LA, Lauritsen KJ, Cross N, Davis AK, Rosenberg H, Bonadio F, Lang B (2015) Self-medication of somatic and psychiatric conditions using botanical marijuana. J Psychoactive Drugs 47(5):345–350. https://doi.org/10.1080/02791072.2015.1096433
Passik SD, Lowery A (2011) Psychological variables potentially implicated in opioid-related mortality as observed in clinical practice. Pain Med (Malden, Mass) 12(Suppl 2):S36–S42. https://doi.org/10.1111/j.1526-4637.2011.01130.x
Portenoy RK, Ganae-Motan ED, Allende S, Yanagihara R, Shaiova L, Weinstein S, McQuade R, Wright S, Fallon MT (2012) Nabiximols for opioid-treated cancer patients with poorly-controlled chronic pain: a randomized, placebo-controlled, graded-dose trial. J Pain 13(5):438–449. https://doi.org/10.1016/j.jpain.2012.01.003
Saunders KW, Von Korff M, Campbell CI, Banta-Green CJ, Sullivan MD, Merrill JO, Weisner C (2012) Concurrent use of alcohol and sedatives among persons prescribed chronic opioid therapy: prevalence and risk factors. J Pain 13(3):266–275. https://doi.org/10.1016/j.jpain.2011.11.004
Savage SR (2008) The language of pain and addiction. In: Smith HS, Passik SD (eds) Pain and chemical dependency. Oxford, pp 9–13
Savage SR, Romero-Sandoval A, Schatman M, Wallace M, Fanciullo G, McCarberg B, Ware M (2016) Cannabis in pain treatment: clinical and research considerations. J Pain 17(6):654–668. https://doi.org/10.1016/j.jpain.2016.02.007
Schieffer BM, Pham Q, Labus J, Baria A, Van Vort W, Davis P, Davis F, Naliboff BD (2005) Pain medication beliefs and medication misuse in chronic pain. J Pain 6(9):620–629. https://doi.org/10.1016/j.jpain.2005.04.004
Skinner HA (1982) The drug abuse screening test. Addict Behav 7(4):363–371. https://doi.org/10.1016/0306-4603(82)90005-3
Smith HS, Passik SD (2008) Pain and chemical dependency. Oxford University Press, New York
Spearing MK, Post RM, Leverich GS, Brandt D, Nolen W (1997) Modification of the Clinical Global Impressions (CGI) Scale for use in bipolar illness (BP): the CGI-BP. Psychiatry Res 73(3):159–171. https://doi.org/10.1016/S0165-1781(97)00123-6
Toth C, Mawani S, Brady S, Chan C, Liu C, Mehina E, Garven A, Bestard J, Korngut L (2012) An enriched-enrolment, randomized withdrawal, flexible-dose, double-blind, placebo-controlled, parallel assignment efficacy study of nabilone as adjuvant in the treatment of diabetic peripheral neuropathic pain. Pain 153(10):2073–2082. https://doi.org/10.1016/j.pain.2012.06.024
Wade DT, Makela P, Robson P, House H, Bateman C (2004) Do cannabis-based medicinal extracts have general or specific effects on symptoms in multiple sclerosis? A double-blind, randomized, placebo-controlled study on 160 patients. Mult Scler (Houndmills, Basingstoke, England) 10(4):434–441. https://doi.org/10.1191/1352458504ms1082oa
Wade DT, Makela PM, House H, Bateman C, Robson P (2006) Long-term use of a cannabis-based medicine in the treatment of spasticity and other symptoms in multiple sclerosis. Mult Scler (Houndmills, Basingstoke, England) 12(5):639–645. https://doi.org/10.1177/1352458505070618
Ware MA, Fitzcharles MA, Joseph L, Shir Y (2010) The effects of nabilone on sleep in fibromyalgia: results of a randomized controlled trial. Anesth Analg 110(2):604–610. https://doi.org/10.1213/ANE.0b013e3181c76f70
Ware MA, Wang T, Shapiro S, Collet JP (2015) Cannabis for the management of pain: assessment of safety study (COMPASS). J Pain 16(12):1233–1242. https://doi.org/10.1016/j.jpain.2015.07.014
Wasan AD, Butler SF, Budman SH, Benoit C, Fernandez K, Jamison RN (2007) Psychiatric history and psychologic adjustment as risk factors for aberrant drug-related behavior among patients with chronic pain. Clin J Pain 23(4):307–315. https://doi.org/10.1097/AJP.0b013e3180330dc5
Wu SA, Compton P, Bolus R, Schieffer B, Pham Q, Baria A, Van Vort W, Davis F, Shekelle P, Naliboff BD (2006) The addiction behaviors checklist: validation of a new clinician-based measure of inappropriate opioid use in chronic pain. J Pain Symptom Manag 32:342–351
Acknowledgements
The authors wish to acknowledge all the additional clinical site investigators who enrolled subjects for the study, including Dr. Gordon Ko, Dr. Peter Blecher, Dr. Aline Boulanger, and Dr. May Ong-Lam. We also wish to acknowledge the advice of Dr. Kenneth Kirsch, the study management team at AXON for study coordination and data management, and medical writer Mark E. Rose, MA, for putting together the early draft of the paper.
Funding
The study was supported by an unrestricted educational grant from Valeant Pharmaceuticals (Canada). Valeant markets nabilone under the brand name Cesamet®.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
MAW discloses the following relationships: CanniMed, Green Sky Labs (grant to institution), CHI Inc., Zynerba, and CannaRoyalty (consultant). RJ discloses the following relationships: Astra Zeneca, Knight, Paladin, and Purdue Pharma (speakers’ bureau, consultant). MM and JS declare that they have no conflict of interest.
Electronic supplementary material
Supplementary Table 1
(DOCX 17 kb)
Supplementary Table 2
(DOCX 16 kb)
Supplementary Table 3
(DOCX 17 kb)
Supplementary Table 4
(DOCX 14 kb)
Supplementary Table 5
(DOCX 12 kb)
Supplementary Table 6
(DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Ware, M.A., Martel, M.O., Jovey, R. et al. A prospective observational study of problematic oral cannabinoid use. Psychopharmacology 235, 409–417 (2018). https://doi.org/10.1007/s00213-017-4811-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4811-6