Abstract
Rationale
Anticipatory nausea (AN) is a poorly controlled side effect experienced by chemotherapy patients. Currently, pharmacotherapy is restricted to benzodiazepine anxiolytics, which have limited efficacy, have significant sedative effects and induce dependency. The non-psychoactive phytocannabinoid, cannabidiolic acid (CBDA), has shown considerable efficacy in pre-clinical AN models, however determination of its neuromotor tolerability profile is crucial to justify clinical investigation. Provisional evidence for appetite-stimulating properties also requires detailed investigation.
Objectives
This study aims to assess the tolerability of CBDA in locomotor activity, motor coordination and muscular strength tests, and additionally for ability to modulate feeding behaviours.
Methods
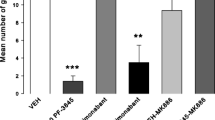
Male Lister Hooded rats administered CBDA (0.05–5 mg/kg; p.o.) were assessed in habituated open field (for locomotor activity), static beam and grip strength tests. A further study investigated whether these CBDA doses modulated normal feeding behaviour. Finally, evidence of anxiolytic-like effects in the habituated open field prompted testing of 5 mg/kg CBDA for anxiolytic-like activity in unhabituated open field, light/dark box and novelty-suppressed feeding (NSF) tests.
Results
CBDA had no adverse effects upon performance in any neuromotor tolerability test, however anxiolytic-like behaviour was observed in the habituated open field. Normal feeding behaviours were unaffected by any dose. CBDA (5 mg/kg) abolished the increased feeding latency in the NSF test induced by the 5-HT1AR antagonist, WAY-100,635, indicative of anxiolytic-like effects, but had no effect on anxiety-like behaviour in the novel open field or light/dark box.
Conclusions
CBDA is very well tolerated and devoid of the sedative side effect profile of benzodiazepines, justifying its clinical investigation as a novel AN treatment.




Similar content being viewed by others
Abbreviations
- CBDA:
-
Cannabidiolic acid
- CBD:
-
Cannabidiol
- pCB:
-
Phytocannabinoid
- CINV:
-
Chemotherapy-induced nausea and vomiting
- AN:
-
Anticipatory nausea
- CDP:
-
Chlordiazepoxide
- NK1:
-
Neurokinin 1
- 5-HT:
-
5-Hydroxytryptamine
- 5-HT1AR:
-
5-Hydroxytryptamine receptor type 1A
References
Ahlberg K, Ekman T, Gaston-Johansson F, Mock V (2003) Assessment and management of cancer-related fatigue in adults. Lancet 362:640–650. doi:10.1016/S0140-6736(03)14186-4
Basch E, Prestrud AA, Hesketh PJ et al (2012) Antiemetic use in oncology: updated guideline recommendations from ASCO. Am Soc Clin Oncol Educ Book 532:532–540. doi:10.14694/EdBook_AM.2012.32.532
Bolognini D, Rock EM, Cluny NL et al (2013) Cannabidiolic acid prevents vomiting in Suncus murinus and nausea-induced behaviour in rats by enhancing 5-HT1A receptor activation. Br J Pharmacol 168:1456–1470. doi:10.1111/bph.12043
Bourin M, Hascoët M (2003) The mouse light/dark box test. Eur J Pharmacol 463:55–65. doi:10.1016/S0014-2999(03)01274-3
Bovbjerg DH (2006) The continuing problem of post chemotherapy nausea and vomiting: contributions of classical conditioning. Auton Neurosci 129:92–98. doi:10.1016/j.autneu.2006.07.016
Britton DR, Britton KT (1981) A sensitive open field measure of anxiolytic drug activity. Pharmacol Biochem Behav 15:577–582. doi:10.1016/0091-3057(81)90212-4
Campos AC, Moreira FA, Gomes FV et al (2012) Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders. Philos Trans R Soc B Biol Sci 367:3364–3378. doi:10.1098/rstb.2011.0389
Campos D, Pereira JR, Reinhardt RR et al (2001) Prevention of cisplatin-induced emesis by the oral neurokinin-1 antagonist, MK-869, in combination with granisetron and dexamethasone or with dexamethasone alone. J Clin Oncol 19:1759–1767
Cluny NL, Naylor RJ, Whittle BA, Javid FA (2011) The effects of cannabidiolic acid and cannabidiol on contractility of the gastrointestinal tract of Suncus murinus. Arch Pharm Res 34:1509–1517. doi:10.1007/s12272-011-0913-6
Curzon P, Zhang M, Radek R, Fox G (2009) The behavioral assessment of sensorimotor processes in the mouse: acoustic startle, sensory gating, locomotor activity, rotarod, and beam walking. Methods Behav. Anal, Neurosci
Dos Santos LV, Souza FH, Brunetto AT et al (2012) Neurokinin-1 receptor antagonists for chemotherapy-induced nausea and vomiting: a systematic review. J Natl Cancer Inst 104:1280–1292. doi:10.1093/jnci/djs335
Dulawa SC, Hen R (2005) Recent advances in animal models of chronic antidepressant effects: the novelty-induced hypophagia test. Neurosci Biobehav Rev 29:771–783. doi:10.1016/j.neubiorev.2005.03.017
Duvvuri V, Risbrough VB, Kaye WH, Geyer MA (2009) 5-HT1A receptor activation is necessary for 5-MeODMT-dependent potentiation of feeding inhibition. Pharmacol Biochem Behav 93:349–353. doi:10.1016/j.pbb.2009.05.014
Farrimond JA, Hill AJ, Whalley BJ, Williams CM (2010a) Cannabis constituents modulate δ9-tetrahydrocannabinol-induced hyperphagia in rats. Psychopharmacology (Berl) 210:97–106. doi:10.1007/s00213-010-1821-z
Farrimond JA, Whalley BJ, Williams CM (2012a) Non-Δ9tetrahydrocannabinol phytocannabinoids stimulate feeding in rats. Behav Pharmacol 23:113–117. doi:10.1097/FBP.0b013e32834ed832
Farrimond JA, Whalley BJ, Williams CM (2012b) Cannabinol and cannabidiol exert opposing effects on rat feeding patterns. Psychopharmacology (Berl) 223:117–129. doi:10.1007/s00213-012-2697-x
Farrimond JA, Whalley BJ, Williams CM (2010b) A low-Δ9tetrahydrocannabinol cannabis extract induces hyperphagia in rats. Behav Pharmacol. doi:10.1097/FBP.0b013e328340a062
Ferguson SA, Paule MG (1996) Effects of chlorpromazine and diazepam on time estimation behavior and motivation in rats. Pharmacol Biochem Behav 53:115–122. doi:10.1016/0091-3057(95)02002-0
Figueroa-Moseley C, Jean-Pierre P, Roscoe JA et al (2007) Behavioral interventions in treating anticipatory nausea and vomiting. J Natl Compr Canc Netw 5:44–50
Griebel G, Rodgers RJ, Perrault G, Sanger DJ (1999) Behavioural profiles in the mouse defence test battery suggest anxiolytic potential of 5-HT 1A receptor antagonists. Psychopharmacology (Berl) 144:121–130. doi:10.1007/s002130050984
Griebel G, Rodgers RJ, Perrault G, Sanger DJ (2000) The effects of compounds varying in selectivity as 5-HT1A receptor antagonists in three rat models of anxiety. Neuropharmacology 39:1848–1857. doi:10.1016/S0028-3908(00)00074-5
Gross C, Santarelli L, Brunner D et al (2000) Altered fear circuits in 5-HT(1A) receptor KO mice. Biol Psychiatry 48:1157–1163. doi:10.1016/S0006-3223(00)01041-6
Hainsworth JD, Hesketh PJ (1992) Single-dose ondansetron for the prevention of cisplatin-induced emesis: efficacy results. Semin Oncol 19:14–19
Hesketh PJ (2008) Chemotherapy-induced nausea and vomiting. N Engl J Med 358:2482–2494. doi:10.1056/NEJMra0706547
Hesketh PJ, Grunberg SM, Gralla RJ et al (2003) The oral neurokinin-1 antagonist aprepitant for the prevention of chemotherapy-induced nausea and vomiting: a multinational, randomized, double-blind, placebo-controlled trial in patients receiving high-dose cisplatin—the Aprepitant Protocol 052 Study Gr. J Clin Oncol 21:4112–4119. doi:10.1200/JCO.2003.01.095
Hickok JT, Roscoe JA, Morrow GR et al (2003) Nausea and emesis remain significant problems of chemotherapy despite prophylaxis with 5-hydroxytryptamine-3 antiemetics: a University of Rochester James P. Wilmot Cancer Center Community Clinical Oncology Program Study of 360 cancer patients treated in treated in the community. Cancer 97:2880–2886. doi:10.1002/cncr.11408
Hill AJ, Mercier MS, Hill TDM et al (2012) Cannabidivarin is anticonvulsant in mouse and rat. Br J Pharmacol 167:1629–1642. doi:10.1111/j.1476-5381.2012.02207.x
Hill TDM, Cascio M-G, Romano B et al (2013) Cannabidivarin-rich cannabis extracts are anticonvulsant in mouse and rat via a CB1 receptor-independent mechanism. Br J Pharmacol 170:679–692. doi:10.1111/bph.12321
Janelsins MC, Tejani MA, Kamen C et al (2013) Current pharmacotherapy for chemotherapy-induced nausea and vomiting in cancer patients. Expert Opin Pharmacother 14:757–766. doi:10.1517/14656566.2013.776541
Kamen C, Tejani MA, Chandwani K et al (2014) Anticipatory nausea and vomiting due to chemotherapy. Eur J Pharmacol 722:172–179. doi:10.1016/j.ejphar.2013.09.071
Kilkenny C, Browne W, Cuthill IC et al (2010) Animal research: reporting in vivo experiments: the ARRIVE guidelines. Br J Pharmacol 160:1577–1579. doi:10.1111/j.1476-5381.2010.00872.x
Kris MG, Hesketh PJ, Somerfield MR et al (2006) American Society of Clinical Oncology guideline for antiemetics in oncology: update 2006. J Clin Oncol 24:2932–2947. doi:10.1200/JCO.2006.06.9591
Lotfi-Jam K, Carey M, Jefford M et al (2008) Nonpharmacologic strategies for managing common chemotherapy adverse effects: a systematic review. J Clin Oncol 26:5618–5629. doi:10.1200/JCO.2007.15.9053
Malik IA, Khan WA, Qazilbash M et al (1995) Clinical efficacy of lorazepam in prophylaxis of anticipatory, acute, and delayed nausea and vomiting induced by high doses of cisplatin. A prospective randomized trial. Am J Clin Oncol 18:170–175
Martin M (1996) The severity and pattern of emesis following different cytotoxic agents. Oncology 53(Suppl 1):26–31. doi:10.1159/000227637
Matteson S, Roscoe J, Hickok J, Morrow GR (2002) The role of behavioral conditioning in the development of nausea. Am J Obstet Gynecol 186:S239–S243. doi:10.1067/mob.2002.122597
McGrath JC, Drummond GB, McLachlan EM et al (2010) Guidelines for reporting experiments involving animals: the ARRIVE guidelines. Br J Pharmacol 160:1573–1576. doi:10.1111/j.1476-5381.2010.00873.x
Meyer OA, Tilson HA, Byrd WC, Riley MT (1979) A method for the routine assessment of fore- and hindlimb grip strength of rats and mice. Neurobehav Toxicol 1:233–236
Navari RM, Reinhardt RR, Gralla RJ et al (1999) Reduction of cisplatin-induced emesis by a selective neurokinin-1-receptor antagonist. L-754,030 Antiemetic Trials Group. N Engl J Med 340:190–195. doi:10.1056/NEJM199901213400304
Nesse RM, Carli T, Curtis GC, Kleinman PD (1980) Pretreatment nausea in cancer chemotherapy: a conditioned response? Psychosom Med 42:33–36
Prut L, Belzung C (2003) The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: a review. Eur J Pharmacol 463:3–33. doi:10.1016/S0014-2999(03)01272-X
Razavi D, Delvaux N, Farvacques C et al (1993) Prevention of adjustment disorders and anticipatory nausea secondary to adjuvant chemotherapy: a double-blind, placebo-controlled study assessing the usefulness of alprazolam. J Clin Oncol 11:1384–1390
Rex A, Voigt JP, Voits M, Fink H (1998) Pharmacological evaluation of a modified open-field test sensitive to anxiolytic drugs. Pharmacol Biochem Behav 59:677–683. doi:10.1016/S0091-3057(97)00461-9
Rock E, Parker L (2013a) Effect of low doses of cannabidiolic acid and ondansetron on LiCl-induced conditioned gaping (a model of nausea-induced behaviour) in rats. Br J Pharmacol. doi:10.1111/bph.12162
Rock EM, Limebeer CL, Navaratnam R et al (2014a) A comparison of cannabidiolic acid with other treatments for anticipatory nausea using a rat model of contextually elicited conditioned gaping. Psychopharmacology (Berl). doi:10.1007/s00213-014-3498-1
Rock EM, Limebeer CL, Parker LA (2014b) Anticipatory nausea in animal models: a review of potential novel therapeutic treatments. Exp Brain Res. doi:10.1007/s00221-014-3942-9
Rock EM, Parker LA (2013b) Suppression of lithium chloride-induced conditioned gaping (a model of nausea-induced behaviour) in rats (using the taste reactivity test) with metoclopramide is enhanced by cannabidiolic acid. Pharmacol Biochem Behav 111:84–89. doi:10.1016/j.pbb.2013.08.012
Roila F, Herrstedt J, Aapro M et al (2010) Guideline update for MASCC and ESMO in the prevention of chemotherapy- and radiotherapy-induced nausea and vomiting: results of the Perugia consensus conference. Ann Oncol 21(Suppl 5):v232–v243. doi:10.1093/annonc/mdq194
Roscoe JA, Morrow GR, Aapro MS et al (2011) Anticipatory nausea and vomiting. Support Care Cancer 19:1533–1538. doi:10.1007/s00520-010-0980-0
Sánchez C (1996) 5-HT(1A) receptors play an important role in modulation of behavior of rats in a two-compartment black and white box. Behav Pharmacol 7:788–797
de Schier ARM, Ribeiro NP, Silva AC et al (2012) Cannabidiol, a Cannabis sativa constituent, as an anxiolytic drug. Rev Bras Psiquiatr 34(Suppl 1):S104–S110. doi:10.1590/S1516-44462012000500008
Stanley JL, Lincoln RJ, Brown TA et al (2005) The mouse beam walking assay offers improved sensitivity over the mouse rotarod in determining motor coordination deficits induced by benzodiazepines. J Psychopharmacol 19:221–227. doi:10.1177/0269881105051524
Stephens N, Fearon K (2008) Anorexia, cachexia and nutrition. Medicine (Baltimore) 36:78–81. doi:10.1016/j.mpmed.2007.11.004
Williams CM, Kirkham TC (2002) Observational analysis of feeding induced by Delta9-THC and anandamide. Physiol Behav 76:241–250. doi:10.1016/S0031-9384(02)00725-4
Williams CM, Rogers PJ, Kirkham TC (1998) Hyperphagia in pre-fed rats following oral delta9-THC. Physiol Behav 65:343–346. doi:10.1016/S0031-9384(98)00170-X
Zhang J, Huang X-Y, Ye M-L et al (2010) Neuronal nitric oxide synthase alteration accounts for the role of 5-HT1A receptor in modulating anxiety-related behaviors. J Neurosci 30:2433–2441. doi:10.1523/JNEUROSCI.5880-09.2010
Acknowledgments
This research was supported by grants to CMW and BJW by GW Pharmaceuticals and Otsuka Pharmaceuticals, and in part by the University of Reading Research Endowment Trust Fund to DIB.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The work reported was funded in part by grants to BJW and CMW from GW Pharmaceuticals and Otsuka Pharmaceuticals. The original study concept was discussed with the sponsor (GW Pharmaceuticals) although all subsequent study design, data collection, analysis and interpretation were conducted independently by the authors. The report was approved by the sponsor company prior to submission, and the authors retain full control of all primary data.
Rights and permissions
About this article
Cite this article
Brierley, D.I., Samuels, J., Duncan, M. et al. Neuromotor tolerability and behavioural characterisation of cannabidiolic acid, a phytocannabinoid with therapeutic potential for anticipatory nausea. Psychopharmacology 233, 243–254 (2016). https://doi.org/10.1007/s00213-015-4100-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-015-4100-1