Abstract
Rationale
Non-adherence with medication remains the major correctable cause of poor outcome in schizophrenia. However, few treatments have addressed this major determinant of outcome with novel long-term delivery systems.
Objectives
The aim of this study was to provide biological proof of concept for a long-term implantable antipsychotic delivery system in rodents and rabbits.
Materials and methods
Implantable formulations of haloperidol were created using biodegradable polymers. Implants were characterized for in vitro release and in vivo behavior using prepulse inhibition of startle in rats and mice, as well as pharmacokinetics in rabbits.
Results
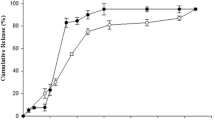
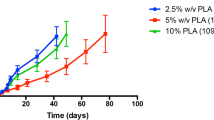
Behavioral measures demonstrate the effectiveness of haloperidol implants delivering 1 mg/kg in mice and 0.6 mg/kg in rats to block amphetamine (10 mg/kg) in mice or apomorphine (0.5 mg/kg) in rats. Additionally, we demonstrate the pattern of release from single polymer implants for 1 year in rabbits.
Conclusions
The current study suggests that implantable formulations are a viable approach to providing long-term delivery of antipsychotic medications in vivo using animal models of behavior and pharmacokinetics. In contrast to depot formulations, implantable formulations could last 6 months or longer. Additionally, implants can be removed throughout the delivery interval, offering a degree of reversibility not available with depot formulations.







Similar content being viewed by others
References
Adams CE, Fenton MK, Quraishi S, David AS (2001) Systematic meta-review of depot antipsychotic drugs for people with schizophrenia. Br J Psychiatry 179:290–299
Andrews L, Friedland G (2000) Progress in HIV therapeutics and the challenges of adherence to antiretroviral therapy. Infect Dis Clin North Am 14:901–928
Ayuso-Gutierrez JL, del Rio Vega JM (1997) Factors influencing relapse in the long-term course of schizophrenia. Schizophr Res 28:199–206
Boccuzzi SJ, Wogen J, Fox J, Sung JC, Shah AB, Kim J (2001) Utilization of oral hypoglycemic agents in a drug-insured U.S. population. Diabetes Care 24:1411–1415
Chui MA, Deer M, Bennett SJ, Tu W, Oury S, Brater DC, Murray MD (2003) Association between adherence to diuretic therapy and health care utilization in patients with heart failure. Pharmacotherapy 23:326–332
Corriss DJ, Smith TE, Hull JW, Lim RW, Pratt SI, Romanelli S (1999) Interactive risk factors for treatment adherence in a chronic psychotic disorders population. Psychiatry Res 89:269–274
Curtis H (1983) Biology, Fourth edn. Worth, New York
Dash AK, Cudworth GC, 2nd (1998) Therapeutic applications of implantable drug delivery systems. J Pharmacol Toxicol Methods 40:1–12
Davis JM, Chen N, Glick ID (2003) A meta-analysis of the efficacy of second-generation antipsychotics. Arch Gen Psychiatry 60:553–564
Fischel-Ghodsian F, Newton JM (1993) Analysis of drug release kinetics from degradable polymeric devices. J Drug Target 1:51–57
Gastpar M, Masiak M, Latif MA, Frazzingaro S, Medori R, Lombertie ER (2005) Sustained improvement of clinical outcome with risperidone long-acting injectable in psychotic patients previously treated with olanzapine. J Psychopharmacol 19:32–38
Geddes J, Freemantle N, Harrison P, Bebbington (2000) Atypical antipsychotics in the treatment of schizophrenia: systematic overview and meta-regression analysis. BMJ 321:1371–1376
Gould TJ, Bizily SP, Tokarczyk J, Kelly MP, Siegel SJ, Kanes SJ, Abel T (2004) Sensorimotor gating deficits in transgenic mice expressing a constitutively active form of Gs alpha. Neuropsychopharmacology 29:494–501
Harris NR, Lovell K, Day JC (2002) Consent and long-term neuroleptic treatment. J Psychiatr Ment Health Nurs 9:475–482
Irani F, Dankert M, Brensinger C, Bilker WB, Nair SR, Kohler CG, Kanes SJ, Turetsky BI, Moberg PJ, Ragland JD, Gur RC, Gur RE, Siegel SJ (2004) Patient attitudes towards surgically implantable, long-term delivery of psychiatric medicine. Neuropsychopharmacology 29:960–968
Kane JM, Aguglia E, Altamura AC, Ayuso Gutierrez JL, Brunello N, Fleischhacker WW, Gaebel W, Gerlach J, Guelfi JD, Kissling W, Lapierre YD, Lindstrom E, Mendlewicz J, Racagni G, Carulla LS, Schooler NR (1998) Guidelines for depot antipsychotic treatment in schizophrenia. European Neuropsychopharmacology Consensus Conference in Siena, Italy. Eur Neuropsychopharmacol 8:55–66
Kitchell JP, Wise DL (1985) Poly(lactic/glycolic acid) biodegradable drug-polymer matrix systems. Methods Enzymol 112:436–448
Lieberman JA, Stroup TS, McEvoy JP, Swartz MS, Rosenheck RA, Perkins DO, Keefe RS, Davis SM, Davis CE, Lebowitz BD, Severe J, Hsiao JK (2005) Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 353:1209–1223
Martin SD, Libretto SE, Pratt DJ, Brewin JS, Huq ZU, Saleh BT (2003) Clinical experience with the long-acting injectable formulation of the atypical antipsychotic, risperidone. Curr Med Res Opin 19:298–305
McCombs JS, Nichol MB, Stimmel GL, Shi J, Smith RR (1999) Use patterns for antipsychotic medications in Medicaid patients with schizophrenia. J Clin Psychiatry 60(Suppl 19):5–11; discussion 12–3
Melbourne KM, Geletko SM, Brown SL, Willey-Lessne C, Chase S, Fisher A (1999) Medication adherence in patients with HIV infection: a comparison of two measurement methods. AIDS Read 9:329–338
Menzin J, Boulanger L, Friedman M, Mackell J, Lloyd JR (2003) Treatment adherence associated with conventional and atypical antipsychotics in a large state medicaid program. Psychiatr Serv 54:719–723
Natsugoe S, Kumanohoso T, Tokuda K, Shimada M, Mueller J, Nakamura K, Yamada K, Fukuzaki H, Aikou T (1997) Controlled release of cisplatin incorporated into biodegradable poly-d,l-lactic acid. Anticancer Res 17:1957–1960
Okada H, Toguchi H (1995) Biodegradable microspheres in drug delivery. Crit Rev Ther Drug Carr Syst 12:1–99
Olfson M, Mechanic D, Hansell S, Boyer CA, Walkup J, Weiden PJ (2000) Predicting medication noncompliance after hospital discharge among patients with schizophrenia. Psychiatr Serv 51:216–222
Regenthal R, Kunstler U, Junhold U, Preiss R (1997) Haloperidol serum concentrations and D2 dopamine receptor occupancy during low-dose treatment with haloperidol decanoate. Int Clin Psychopharmacol 12:255–261
Robinson DG, Woerner MG, Alvir JM, Bilder RM, Hinrichsen GA, Lieberman JA (2002) Predictors of medication discontinuation by patients with first-episode schizophrenia and schizoaffective disorder. Schizophr Res 57:209–219
Rosenheck R, Perlick D, Bingham S, Liu-Mares W, Collins J, Warren S, Leslie D, Allan E, Campbell EC, Caroff S, Corwin J, Davis L, Douyon R, Dunn L, Evans D, Frecska E, Grabowski J, Graeber D, Herz L, Kwon K, Lawson W, Mena F, Sheikh J, Smelson D, Smith-Gamble V (2003) Effectiveness and cost of olanzapine and haloperidol in the treatment of schizophrenia: a randomized controlled trial. Jama 290:2693–2702
Sabel BA, Dominiak P, Hauser W, During MJ, Freese A (1990) Levodopa delivery from controlled-release polymer matrix: delivery of more than 600 days in vitro and 225 days of elevated plasma levels after subcutaneous implantation in rats. J Pharmacol Exp Ther 255:914–922
Seeman MV (2001) Clinical trials in psychiatry: do results apply to practice? Can J Psychiatry 46:352–355
Sharon AC, Wise DL (1981) Development of drug delivery systems for use in treatment of narcotic addiction. NIDA Res Monogr 28:194–213
Siegel SJ, Kahn JB, Metzger K, Winey KI, Werner K, Dan N (2006) Effect of drug type on the degradation rate of PLGA matrices. Eur J Pharm Biopharm 64(3):287–293
Siegel SJ, Winey KI, Gur RE, Lenox RH, Bilker WB, Ikeda D, Gandhi N, Zhang WX (2002) Surgically implantable long-term antipsychotic delivery systems for the treatment of schizophrenia. Neuropsychopharmacology 26:817–823
Svarstad BL, Shireman TI, Sweeney JK (2001) Using drug claims data to assess the relationship of medication adherence with hospitalization and costs. Psychiatr Serv 52:805–811
Swerdlow NR, Geyer MA (1998) Using an animal model of deficient sensorimotor gating to study the pathophysiology and new treatments of schizophrenia. Schizophr Bull 24:285–301
Talbot K, Eidem WL, Tinsley CL, Benson MA, Thompson EW, Smith RJ, Hahn CG, Siegel SJ, Trojanowski JQ, Gur RE, Blake DJ, Arnold SE (2004) Dysbindin-1 is reduced in intrinsic, glutamatergic terminals of the hippocampal formation in schizophrenia. J Clin Invest 113:1353–1363
Tucker JS, Burnam MA, Sherbourne CD, Kung FY, Gifford AL (2003) Substance use and mental health correlates of nonadherence to antiretroviral medications in a sample of patients with human immunodeficiency virus infection. Am J Med 114:573–580
Velligan DI, Lam F, Ereshefsky L, Miller AL (2003) Psychopharmacology: perspectives on medication adherence and atypical antipsychotic medications. Psychiatr Serv 54:665–667
Acknowledgements
The Stanley Medical Research Institute funded this research. Intellectual property related to haloperidol implants developed at the University of Pennsylvania is owned by the University. Research by NRS and JMS supported by MH42228 and MH01436. All experiments comply with “Principles of Laboratory Animal Care” and conform to University of Pennsylvania IACUC standards.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Metzger, K.L., Shoemaker, J.M., Kahn, J.B. et al. Pharmacokinetic and behavioral characterization of a long-term antipsychotic delivery system in rodents and rabbits. Psychopharmacology 190, 201–211 (2007). https://doi.org/10.1007/s00213-006-0616-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0616-8